Levomethadone
 | |
| Clinical data | |
|---|---|
| AHFS/Drugs.com | International Drug Names |
| Routes of administration | Oral, IV, IM, SC, IT[1] |
| ATC code | |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Bioavailability | High[1] |
| Protein binding | 60-90%[1] |
| Elimination half-life | ~18 hours[1] |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| E number | {{#property:P628}} |
| ECHA InfoCard | {{#property:P2566}}Lua error in Module:EditAtWikidata at line 36: attempt to index field 'wikibase' (a nil value). |
| Chemical and physical data | |
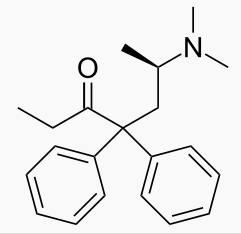
| Formula | C21H27NO |
| Molar mass | 309.445 g/mol |
| 3D model (JSmol) | |
| |
| |
|
WikiDoc Resources for Levomethadone |
|
Articles |
|---|
|
Most recent articles on Levomethadone Most cited articles on Levomethadone |
|
Media |
|
Powerpoint slides on Levomethadone |
|
Evidence Based Medicine |
|
Clinical Trials |
|
Ongoing Trials on Levomethadone at Clinical Trials.gov Trial results on Levomethadone Clinical Trials on Levomethadone at Google
|
|
Guidelines / Policies / Govt |
|
US National Guidelines Clearinghouse on Levomethadone NICE Guidance on Levomethadone
|
|
Books |
|
News |
|
Commentary |
|
Definitions |
|
Patient Resources / Community |
|
Patient resources on Levomethadone Discussion groups on Levomethadone Patient Handouts on Levomethadone Directions to Hospitals Treating Levomethadone Risk calculators and risk factors for Levomethadone
|
|
Healthcare Provider Resources |
|
Causes & Risk Factors for Levomethadone |
|
Continuing Medical Education (CME) |
|
International |
|
|
|
Business |
|
Experimental / Informatics |
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]
Overview
Levomethadone (INN; L-Polamidon, L-Polamivet, Levadone, Levothyl), or levamethadone, is a synthetic opioid analgesic and antitussive which is marketed in Europe and is used for pain management and in opioid maintenance therapy.[1][2][3] In addition to being used as a pharmaceutical drug itself, levomethadone, or R-(−)-methadone, is the active enantiomer of methadone,[2] having approximately 50x the potency of the S-(+)-enantiomer as well as greater μ-opioid receptor selectivity.[1][4] Accordingly, it is about twice as potent as methadone by weight and its effects are virtually identical in comparison.[5][6] In addition to its activity at the opioid receptors, levomethadone has been found to act as a weak competitive antagonist of the N-methyl-D-aspartate (NMDA) receptor complex[7] and as a potent noncompetitive antagonist of the α3β4 nicotinic acetylcholine (nACh) receptor.[8]
There is now an asymmetric synthesis[9] available to prepare both levomethadone [R-(−)-methadone] and dextromethadone [S-(+)-methadone].[10]
The separation of the stereoisomers is one of the easier in organic chemistry and is described in the original patent. It involves "treatment of racemic methadone base with d-(+)-tartaric acid in an acetone/water mixture [which] precipitates almost solely the dextro-methadone levo-tartrate, and the more potent levo-methadone can easily be retrieved from the mother liquor in a high state of optical purity" [11]
Regulatory status
Levomethadone is listed under the Single Convention On Narcotic Drugs 1961 and is a Schedule II Narcotic controlled substance in the US as an isomer of methadone (ACSCN 9250) and is not listed separately, nor is dextromethadone.[12] It is similarly controlled under the German Betäubungsmittelgesetz and similar laws in practically every other country.
See also
References
- ↑ 1.0 1.1 1.2 1.3 1.4 1.5 Helmut Buschmann (20 December 2002). Analgesics: From Chemistry and Pharmacology to Clinical Application. Wiley-VCH. p. 196. ISBN 978-3-527-30403-5. Retrieved 17 May 2012.
- ↑ 2.0 2.1 F.. Macdonald (1997). Dictionary of Pharmacological Agents. CRC Press. p. 1294. ISBN 978-0-412-46630-4. Retrieved 17 May 2012.
- ↑ Index Nominum 2000: International Drug Directory. Taylor & Francis US. 2000. p. 605. ISBN 978-3-88763-075-1. Retrieved 17 May 2012.
- ↑ Renate Förch; Holger Schönherr; A. Tobias A. Jenkins (11 August 2009). Surface Design: Applications in Bioscience and Nanotechnology. Wiley-VCH. p. 193. ISBN 978-3-527-40789-7. Retrieved 17 May 2012.
- ↑ Eduardo Bruera; Sriram Yennurajalingam (16 August 2011). Oxford American Handbook of Hospice and Palliative Medicine. Oxford University Press. p. 43. ISBN 978-0-19-538015-6. Retrieved 17 May 2012.
- ↑ Verthein U, Ullmann R, Lachmann A; et al. (November 2005). "The effects of racemic D,L-methadone and L-methadone in substituted patients--a randomized controlled study". Drug and Alcohol Dependence. 80 (2): 267–71. doi:10.1016/j.drugalcdep.2005.04.007. PMID 15916866.
- ↑ Eric C. Strain; Maxine L. Stitzer (4 November 2005). The Treatment of Opioid Dependence. JHU Press. p. 63. ISBN 978-0-8018-8303-3. Retrieved 19 May 2012.
- ↑ Xiao Y, Smith RD, Caruso FS, Kellar KJ (October 2001). "Blockade of rat alpha3beta4 nicotinic receptor function by methadone, its metabolites, and structural analogs". The Journal of Pharmacology and Experimental Therapeutics. 299 (1): 366–71. PMID 11561100.
- ↑ Hull JD, Scheinmann F, Turner NJ, (March 2003). "Synthesis of optically active methadones, LAAM and bufuralol by lipase-catalysed acylations". Tetrahedron: Asymmetry. 14 (5): 567–576. doi:10.1016/S0957-4166(03)00019-3.
- ↑ US patent 6143933
- ↑ https://www.erowid.org/archive/rhodium/chemistry/methadone.html
- ↑ http://www.deadiversion.usdoj.gov/quotas/conv_factor/index.html
- Pages with script errors
- CS1 maint: Explicit use of et al.
- CS1 maint: Multiple names: authors list
- Template:drugs.com link with non-standard subpage
- E number from Wikidata
- ECHA InfoCard ID from Wikidata
- Chemical articles with unknown parameter in Infobox drug
- Articles without EBI source
- Chemical pages without DrugBank identifier
- Articles without KEGG source
- Articles without UNII source
- Articles containing unverified chemical infoboxes
- Drug
- Amines
- Analgesics
- Antitussives
- Drug rehabilitation
- Ketones
- Mu-opioid agonists
- Nicotinic antagonists
- NMDA receptor antagonists
- Synthetic opioids
- Glycine receptor antagonists