Dehydroepiandrosterone
 | |
| Clinical data | |
|---|---|
| Routes of administration | Oral |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Metabolism | Hepatic |
| Elimination half-life | 12 hours |
| Excretion | Urinary:?% |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| E number | {{#property:P628}} |
| ECHA InfoCard | {{#property:P2566}}Lua error in Module:EditAtWikidata at line 36: attempt to index field 'wikibase' (a nil value). |
| Chemical and physical data | |
| Formula | C19H28O2 |
| Molar mass | 288.43 |
| Melting point | 148.5 °C (299.3 °F) |
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [2]; Associate Editor(s)-in-Chief: Dayana Davidis, M.D. [3]
Synonyms and keywords: Dehydro-3-epiandrosterone, DHEA, Dehydroisoandrosterone, 3β-Hydroxy-5-androsten-17-one, 3β-Hydroxyandrost-5-en-17-one, Androstenol, Androstenolone, Hydroxyandrost-5-en-17-one, Prasterone, trans-Dehydroandrosterone
Overview
Dehydroepiandrosterone is a natural steroid prohormone produced from cholesterol by the adrenal glands, the gonads, adipose tissue, brain and in the skin (by an autocrine mechanism). DHEA is the precursor of androstenedione, which can undergo further conversion to produce the androgen testosterone and the estrogens estrone and estradiol. The physiologic significance of DHEA is not yet proven and supplementation is not yet approved by FDA.
Brand Names
Brand names for DHEA include Prastera® and Fidelin®.
DHEAS (Dehydroepiandrosterone sulfate)
Dehydroepiandrosterone sulfate (DHEAS, Template:PubChem) is the sulfated version of DHEA. This conversion is reversibly catalyzed by sulfotransferase (SULT2A1) primarily in the adrenals, the liver, and small intestine. In the blood, most DHEA is found as DHEAS with levels that are about 300 times higher than free DHEA. Orally ingested DHEA is converted to its sulfate when passing through intestines and liver. While DHEA levels naturally reach their peak in the early morning hours, DHEAS levels show no diurnal variation.
From a practical point of view, measurement of DHEAS is preferable to DHEA as levels are more stable.
Physiology
Production
Dehydroepiandrosterone or DHEA is secreted by the adrenal glands, which convert it from the hormone pregnenolone, a hormone secreted in the brain that is converted from cholesterol. DHEA is the basic hormone that the adrenal cortex uses to synthesise all the sex hormones including testosterone, progesterone and oestrogen and the stress hormone cortisol. It is the most abundant hormone in the body, however by age 40 its production deceases by half its peak age, 20, by age 75 it drops to 10 percent its levels at peak age.
All studies to date were not conclusive enough to prove that supplementation with DHEA would help increase its production in humans, animals studies have shown promise however humans metabolise DHEA differently than animals
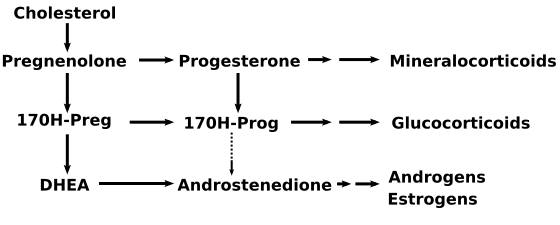
DHEA is produced from cholesterol through two cytochrome P450 enzymes. Cholesterol is converted to pregnenolone by the enzyme P450 scc (side chain cleavage); then another enzyme, CYP17A1, converts pregnenolone to 17α-Hydroxypregnenolone and then to DHEA. In humans, DHEA is the dominant steroid hormone and precursor of all sex steroids.
DHEA is converted to testosterone in women and to esterogen in men.
Role
DHEA can be understood as a prohormone for the sex steroids. DHEAS may be viewed as buffer and reservoir. Its production in the brain suggests that it also has a role as a neurosteroid. As most DHEA is produced by the zona reticularis of the adrenal, it is argued that there is a role in the immune and stress response.
As almost all DHEA is derived from the adrenal glands, blood measurements of DHEAS/DHEA are useful to detect excess adrenal activity as seen in adrenal cancer or hyperplasia, including certain forms of congenital adrenal hyperplasia. Women with polycystic ovary syndrome tend to have elevated levels of DHEAS.
A number of ongoing studies are conducted to link low levels of DHEA to premature ageing, low immunity, low and weak muscle mass, joint pain and arthritis pain, low energy, low stamina and even to fertility problems in women, impaired thinking in elderly and progression of Alzheimer's.
Claims have been found to link low levels of DHEA to obesity, metabolic syndrome, abnormal cholesterol levels and increased waistline.
Effects and uses
Studies have shown that DHEA is useful in patients with systemic lupus erythematosus. An application of the evidence was reviewed by the U.S. Food and Drug Administration in 2001 and is available online.[1] This review also shows that cholesterol and other serum lipids decrease with the use of DHEA (mainly a decrease in HDL-C and triglycerides can be expected in women, p110).
DHEA supplementation has been studied as a treatment for Alzheimer's disease, but was found to be ineffective.[2] Some small placebo-controlled randomized clinical trial studies have found long-term supplementation to improve mood and relieve depression[3][4] or to decrease insulin resistance.[5] However, a larger placebo-controlled randomized clinical trial reported in the New England Journal of Medicine in 2006 found that DHEA supplementation in elderly men and women had no beneficial effects on body composition, physical performance, insulin sensitivity, or quality of life.[6]
DHEA supplements are sometimes used as muscle-building or performance-enhancing drugs by athletes. However, a randomized placebo-controlled trial found that DHEA supplementation had no effect on lean body mass, strength, or testosterone levels.[7]
A 1986 study found that a higher level of endogenous DHEA, as determined by a single measurement, correlated with a lower risk of death or cardiovascular disease.[8] However, a more recent 2006 study found no correlation between DHEA levels and risk of cardiovascular disease or death in men.[9]
Some in vitro studies have found DHEA to have an anti-proliferative or apoptotic effect on cancer cell lines.[10][11][12] The clinical significance of these findings, if any, is unknown. Higher levels of DHEA, in fact, have been correlated with an increased risk of developing breast cancer in both pre- and postmenopausal women.[13][14]
A 2002 review found that DHEA was difficult to study in an animal model. The authors concluded that there was no evidence that DHEA was beneficial for any of the conditions for which it had been studied to that point, that it was associated with significant side effects, and that based on these findings, "there is currently no scientific reason to prescribe DHEA for any purpose whatsoever."[15]
Disputed Effects
In the United States, dietary supplements containing DHEA or DHEAS have been advertised with claims that they may be beneficial for a wide variety of ailments. DHEA and DHEAS are readily available in the United States, where they are regulated as foods rather than as medications. Given the lack of any proven benefit from DHEA supplementation, a 2004 review in the American Journal of Sports Medicine concluded that "The marketing of this supplement's effectiveness far exceeds its science."[16]
Causes of Alterations in Concentrations of DHEA
Decreased
Increased
- Adrenal cortex tumors
- Adrenogenital Syndrome
- Androgen-producing tumor
- Hirsutism
- Hyperprolactinemia
- Ovarian cancer
- Polycystic ovaries [17] [18]
Precautions
Some assert that DHEA should not be supplemented outside specialist centres under careful observation of experts in the field of endocrinology.
Side effects may include:
- Stunted growth in teens who have not reached their height potential.
- Palpitations and other arrhythmias
- Extensive growth of body hair, or hirsutism
- Hair loss, especially male pattern baldness
- Acne
Contraindication
As DHEAS and DHEA are converted to sex steroids, their use is contraindicated in patients with any cancer that is estrogen- or testosterone-dependent.
Increasing Endogenous Production
Regular exercise is known to increase DHEA production in the body.[19][20][21] Caloric restriction has also been shown to increase DHEA in primates.[22] Some theorize that the increase in endogenous DHEA brought about by caloric restriction is partially responsible for the longer life known to be associated with caloric restriction.[23]
Legality
A bill has been introduced on in the U.S. Senate (S. 762) that attempts to classify DHEA as a controlled substance under the category of anabolic steroids. The sponsor is Charles Grassley (R-IA). The cosponsors are Richard Durbin (D-IL), and John McCain (R-AZ).[24] In Canada, a prescription is required to buy DHEA.[25]
References
- ↑ FDA document regading DHEA and SLE
- ↑ Wolkowitz OM, Kramer JH, Reus VI; et al. (2003). "DHEA treatment of Alzheimer's disease: a randomized, double-blind, placebo-controlled study". Neurology. 60 (7): 1071–6. PMID 12682308.
- ↑ Wolkowitz OM, Reus VI, Keebler A; et al. (1999). "Double-blind treatment of major depression with dehydroepiandrosterone". The American journal of psychiatry. 156 (4): 646–9. PMID 10200751.
- ↑ Schmidt PJ, Daly RC, Bloch M; et al. (2005). "Dehydroepiandrosterone monotherapy in midlife-onset major and minor depression". Arch. Gen. Psychiatry. 62 (2): 154–62. doi:10.1001/archpsyc.62.2.154. PMID 15699292.
- ↑ Kawano H, Yasue H, Kitagawa A; et al. (2003). "Dehydroepiandrosterone supplementation improves endothelial function and insulin sensitivity in men". J. Clin. Endocrinol. Metab. 88 (7): 3190–5. PMID 12843164.
- ↑ Nair KS, Rizza RA, O'Brien P; et al. (2006). "DHEA in elderly women and DHEA or testosterone in elderly men". N. Engl. J. Med. 355 (16): 1647–59. doi:10.1056/NEJMoa054629. PMID 17050889.
- ↑ Wallace MB, Lim J, Cutler A, Bucci L (1999). "Effects of dehydroepiandrosterone vs androstenedione supplementation in men". Medicine and science in sports and exercise. 31 (12): 1788–92. PMID 10613429.
- ↑ Barrett-Connor E, Khaw KT, Yen SS (1986). "A prospective study of dehydroepiandrosterone sulfate, mortality, and cardiovascular disease". N. Engl. J. Med. 315 (24): 1519–24. PMID 2946952.
- ↑ Arnlöv J, Pencina MJ, Amin S; et al. (2006). "Endogenous sex hormones and cardiovascular disease incidence in men". Ann. Intern. Med. 145 (3): 176–84. PMID 16880459.
- ↑ Yang NC, Jeng KC, Ho WM, Hu ML (2002). "ATP depletion is an important factor in DHEA-induced growth inhibition and apoptosis in BV-2 cells". Life Sci. 70 (17): 1979–88. PMID 12148690.
- ↑ Schulz S, Klann RC, Schönfeld S, Nyce JW (1992). "Mechanisms of cell growth inhibition and cell cycle arrest in human colonic adenocarcinoma cells by dehydroepiandrosterone: role of isoprenoid biosynthesis". Cancer Res. 52 (5): 1372–6. PMID 1531325.
- ↑ Loria RM (2002). "Immune up-regulation and tumor apoptosis by androstene steroids". Steroids. 67 (12): 953–66. PMID 12398992.
- ↑ Tworoger SS, Missmer SA, Eliassen AH; et al. (2006). "The association of plasma DHEA and DHEA sulfate with breast cancer risk in predominantly premenopausal women". Cancer Epidemiol. Biomarkers Prev. 15 (5): 967–71. doi:10.1158/1055-9965.EPI-05-0976. PMID 16702378.
- ↑ Key T, Appleby P, Barnes I, Reeves G (2002). "Endogenous sex hormones and breast cancer in postmenopausal women: reanalysis of nine prospective studies". J. Natl. Cancer Inst. 94 (8): 606–16. PMID 11959894.
- ↑ "DHEA: the last elixir". Prescrire international. 11 (60): 118–23. 2002. PMID 12199273.
- ↑ Tokish JM, Kocher MS, Hawkins RJ (2004). "Ergogenic aids: a review of basic science, performance, side effects, and status in sports". The American journal of sports medicine. 32 (6): 1543–53. doi:10.1177/0363546504268041. PMID 15310585.
- ↑ Sailer, Christian, Wasner, Susanne. Differential Diagnosis Pocket. Hermosa Beach, CA: Borm Bruckmeir Publishing LLC, 2002:77 ISBN 1591032016
- ↑ Kahan, Scott, Smith, Ellen G. In A Page: Signs and Symptoms. Malden, Massachusetts: Blackwell Publishing, 2004:68 ISBN 140510368X
- ↑ Eur J Appl Physiol Occup Physiol 1998 Oct;78(5):466-71
- ↑ Eur J Appl Physiol. 2001 Jul;85(1- 2):177-84
- ↑ J Gerontol A Biol Sci Med Sci. 2002 Apr;57(4):B158-65
- ↑ Exp Gerontol. 2003 Jan-Feb; 38(1-2):35-46
- ↑ Roberts E. The importance of dehydroepiandrosterone sulfate in the blood of primates: a longer and healthier life? Biochem Pharmacol. 1999 Feb 15;57(4):329-46.
- ↑ S. 762: A bill to include dehydroepiandrosterone as an anabolic steroid, from Govtrack.us. Accessed May 9, 2007.
- ↑ Dr. Michael Colgin. The Deal With D.H.E.A. Vista Magazine Online. www.vistamag.com [1]
External links
- Information on DHEA from the Mayo Clinic
- DHEA, from the Skeptic's Dictionary
- DHEA in elderly women and DHEA or testosterone in elderly men, published in the New England Journal of Medicine in 2006. "Neither DHEA nor low-dose testosterone replacement in elderly people has physiologically relevant beneficial effects on body composition, physical performance, insulin sensitivity, or quality of life."
- Pages with script errors
- CS1 maint: Explicit use of et al.
- CS1 maint: Multiple names: authors list
- Drugs with non-standard legal status
- E number from Wikidata
- ECHA InfoCard ID from Wikidata
- Chemical articles with unknown parameter in Infobox drug
- Articles without EBI source
- Chemical pages without ChemSpiderID
- Chemical pages without DrugBank identifier
- Articles without KEGG source
- Articles without InChI source
- Articles without UNII source
- Articles containing unverified chemical infoboxes
- Androgens
- Neurosteroids
- Dietary supplements
- Endocrinology