Levorphanol
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]; Associate Editor(s)-in-Chief: Chetan Lokhande, M.B.B.S [2]
WikiDoc MAKES NO GUARANTEE OF VALIDITY. WikiDoc is not a professional health care provider, nor is it a suitable replacement for a licensed healthcare provider. WikiDoc is intended to be an educational tool, not a tool for any form of healthcare delivery. The educational content on WikiDoc drug pages is based upon the FDA package insert, National Library of Medicine content and practice guidelines / consensus statements. WikiDoc does not promote the administration of any medication or device that is not consistent with its labeling. Please read our full disclaimer here.
Overview
Levorphanol is an analgesic opioid that is FDA approved for the {{{indicationType}}} of chronic pain, pain (moderate to severe) and premedication for anesthetic procedure - Surgical procedure. Common adverse reactions include cardiovascular: hypotension, dermatologic: pruritus, gastrointestinal: constipation, nausea, vomitingpsychiatric: altered mental status, disturbance in mood.
Adult Indications and Dosage
FDA-Labeled Indications and Dosage (Adult)
- Dosage should be individualized based on degree of pain, clinical setting, physical condition of the patient, and kind and dose of concurrent medication
- Pain, chronic: initial, total daily dose 1/15 to 1/12 of total daily dose of oral morphine; wait 72 h between dose adjustments
- Pain (Moderate to Severe): up to 1 mg IV injection in divided doses, may repeat in 3 to 6 h as needed; max 4 to 8 mg daily
- Pain (Moderate to Severe): 1 to 2 mg IM/SC, may repeat in 6 to 8 h as needed; max 3 to 8 mg IM daily
- Pain (Moderate to Severe): 2 mg orally, may repeat in 6 to 8 h as needed; may increase dose up to 3 mg every 6 to 8 h if needed; max 6 to 12 mg daily
- Premedication for anesthetic procedure - surgical procedure: 1 to 2 mg IM/SC 60 to 90 min before surgery
Off-Label Use and Dosage (Adult)
Guideline-Supported Use
There is limited information about Off-Label Guideline-Supported Use of Levorphanol in adult patients.
Non–Guideline-Supported Use
There is limited information about Off-Label Non–Guideline-Supported Use of Levorphanol in adult patients.
Pediatric Indications and Dosage
FDA-Labeled Indications and Dosage (Pediatric)
- Safety and effectiveness in pediatric patients have not been established
Off-Label Use and Dosage (Pediatric)
Guideline-Supported Use
There is limited information about Off-Label Guideline-Supported Use of Levorphanol in pediatric patients.
Non–Guideline-Supported Use
There is limited information about Off-Label Non–Guideline-Supported Use of Levorphanol in pediatric patients.
Contraindications
- Levorphanol is contraindicated in patients hypersensitive to levorphanol tartrate.
Warnings
Respiratory Depression
- Levorphanol, like morphine, may be expected to produce serious or potentially fatal respiratory depression if given in an excessive dose, too frequently, or if given in full dosage to compromised or vulnerable patients. This is because the doses required to produce analgesia in the general clinical population may cause serious respiratory depression in vulnerable patients. Safe usage of this potent opioid requires that the dose and dosage interval be individualized to each patient based on the severity of the pain, weight, age, diagnosis and physical status of the patient, and the kind and dose of concurrently administered medication.
- The initial dose of Levorphanol should be reduced by 50% or more when the drug is given to patients with any condition affecting respiratory reserve or in conjunction with other drugs affecting the respiratory center. Subsequent doses should then be individually titrated according to the patient’s response. Respiratory depression produced by levorphanol tartrate can be reversed by naloxone, a specific antagonist (see Overdosage).
Preexisting Pulmonary Disease
- Because Levorphanol causes respiratory depression, it should be administered with caution to patients with impaired respiratory reserve or respiratory depression from some other cause (eg, from other medication, uremia, severe infection, obstructive respiratory conditions, restrictive respiratory diseases, intrapulmonary shunting or chronic bronchial asthma). As with other strong opioids, use of Levorphanol in acute or severe bronchial asthma is not recommended (see Respiratory Depression).
Head Injury and Increased Intracranial Pressure
- The respiratory depressant effects of Levorphanol with carbon dioxide retention and secondary elevation of cerebral spinal fluid pressure may be markedly exaggerated in the presence of head injury, other intracranial lesions or pre-existing increase in intracranial pressure. Opioids, including Levorphanol, produce effects that may obscure neurological signs of further increase in pressure in patients with head injuries. In addition, Levorphanol may affect level of consciousness that may complicate neurological evaluation.
Cardiovascular Effects
- The use of Levorphanol in acute myocardial infarction or in cardiac patients with myocardial dysfunction or coronary insufficiency should be limited because the effects of levorphanol on the work of the heart are unknown.
Hypotensive Effect
- The administration of Levorphanol may result in severe hypotension in the postoperative patient or in any individual whose ability to maintain blood pressure has been compromised by a depleted blood volume or by administration of drugs, such as phenothiazines or general anesthetics. Opioids may produce orthostatic hypotension in ambulatory patients.
Use in Liver Disease
- Levorphanol should be administered with caution to patients with extensive liver disease who may be vulnerable to excessive sedation due to increased pharmacodynamic sensitivity or impaired metabolism of the drug.
Biliary Surgery
- Levorphanol has been shown to cause moderate to marked rises in pressure in the common bile duct when given in analgesic doses. It is not recommended for use in biliary surgery.
Use in Alcoholism or Drug Dependence
- Levorphanol has an abuse potential as great as morphine, and the prescription of this drug must always balance the prospective benefits against the risk of abuse and dependence. The use of levorphanol in patients with a history of alcohol or other drug dependence, either active or in remission, has not been specifically studied (see drug abuse and dependence).
Adverse Reactions
Clinical Trials Experience
- In approximately 1400 patients treated with Levorphanol in controlled clinical trials, the type and incidence of side effects were those expected of an opioid analgesic, and no unforeseen or unusual toxicity was reported.
- Drugs of this type are expected to produce a cluster of typical opioid effects in addition to analgesia, consisting of nausea, vomiting, altered mood and mentation, pruritus, flushing, difficulties in urination, constipation and biliary spasm. The frequency and intensity of these effects appears to be dose related. Although listed as adverse events these are expected pharmacologic actions of these drugs and should be interpreted as such by the clinician.
- The following adverse events have been reported with the use of Levorphanol:
- Body as a Whole: abdominal pain, dry mouth, sweating
- Cardiovascular System: cardiac arrest, shock, hypotension, arrhythmias including bradycardia and tachycardia, palpitations, extra-systoles
- Digestive System: nausea, vomiting, dyspepsia, biliary tract spasm
- Nervous System: coma, suicide attempt, convulsions, depression, dizziness, confusion, lethargy, abnormal dreams, abnormal thinking, nervousness, drug withdrawal, hypokinesia, dyskinesia, hyperkinesia, CNS stimulation, personality disorder, amnesia, insomnia
- Respiratory System: apnea, cyanosis, hypoventilation
- Skin & Appendages: pruritus, urticaria, rash, injection site reaction
- Special Senses: abnormal vision, pupillary disorder, diplopia
- Urogenital System: kidney failure, urinary retention, difficulty urinating
Postmarketing Experience
There is limited information regarding Levorphanol Postmarketing Experience in the drug label.
Drug Interactions
- Interactions with Other CNS Agents: Concurrent use of Levorphanol with all central nervous system depressants (eg, alcohol, sedatives, hypnotics, other opioids, general anesthetics, barbitu-rates, tricyclic antidepressants, phenothiazines, tranquilizers, skeletal muscle relaxants and antihistamines) may result in additive central nervous system depressant effects. Respiratory depression, hypotension, and profound sedation or coma may occur. When such combined therapy is contemplated, the dose of one or both agents should be reduced. Although no interaction between MAO inhibitors and Levorphanol has been observed, it is not recommended for use with MAO inhibitors.
- Most cases of serious or fatal adverse events involving Levorphanol reported to the manufacturer or the FDA have involved either the administration of large initial doses or too frequent doses of the drug to nonopioid tolerant patients, or the simultaneous administration of levorphanol with other drugs affecting respiration (see INDIVIDUALIZATION OF DOSAGE and WARNINGS). The initial dose of levorphanol should be reduced by approximately 50% or more when it is given to patients along with another drug affecting respiration.
- Interactions with Mixed Agonist/Antagonist Opioid Analgesics: Agonist/antagonist analgesics (eg, pentazocine, nalbuphine, butorphanol, dezocine and buprenorphine) should NOT be administered to a patient who has received or is receiving a course of therapy with a pure agonist opioid analgesic such as Levorphanol. In opioid-dependent patients, mixed agonist/antagonist analgesics may precipitate withdrawal symptoms.
Use in Specific Populations
Pregnancy
Teratogenic Effects
- Pregnancy Category C. Levorphanol has been shown to be teratogenic in mice when given at a single oral dose of 25 mg/kg. The tested dose caused a near 50% mortality of the mouse embryos. There are no adequate and well-controlled studies in pregnant women. Levorphanol should be used in pregnancy only if the potential benefit justifies the potential risk to the fetus.
Nonteratogenic Effects
- Babies born to mothers who have been taking opioids regularly prior to delivery may be physically dependent.
- A study in rabbits has demonstrated that at doses of 1.5 to 20 mg/kg, Levorphanol administered intravenously crosses the placental barrier and depresses fetal respiration.
Pregnancy Category (AUS):
There is no Australian Drug Evaluation Committee (ADEC) guidance on usage of Levorphanol in women who are pregnant.
Labor and Delivery
- The use of Levorphanol in labor and delivery in humans has not been studied. However, as with other opioids, administration of Levorphanol to the mother during labor and delivery may result in respiratory depression in the newborn. Therefore, its use during labor and delivery is not recommended.
Nursing Mothers
- Studies of levorphanol concentrations in breast milk have not been performed. However, morphine, which is structurally similar to levorphanol, is excreted in human milk. Because of the potential for serious adverse reactions from Levorphanol in nursing infants, a decision should be made whether to discontinue nursing or to discontinue the drug, taking into account the importance of the drug to the mother.
Pediatric Use
- Levorphanol is not recommended in children under the age of 18 years as the safety and efficacy of the drug in this population has not been established.
Geriatic Use
- The initial dose of Levorphanol should be reduced by 50% or more in the infirm elderly patient, even though there have been no reports of unexpected adverse events in older populations. All drugs of this class may be associated with a profound or prolonged effect in elderly patients for both pharmacokinetic and pharmacodynamic reasons and caution is indicated.
Gender
There is no FDA guidance on the use of Levorphanol with respect to specific gender populations.
Race
There is no FDA guidance on the use of Levorphanol with respect to specific racial populations.
Renal Impairment
There is no FDA guidance on the use of Levorphanol in patients with renal impairment.
Hepatic Impairment
There is no FDA guidance on the use of Levorphanol in patients with hepatic impairment.
Females of Reproductive Potential and Males
There is no FDA guidance on the use of Levorphanol in women of reproductive potentials and males.
Immunocompromised Patients
There is no FDA guidance one the use of Levorphanol in patients who are immunocompromised.
Administration and Monitoring
Administration
There is limited information regarding Levorphanol Administration in the drug label.
Monitoring
There is limited information regarding Levorphanol Monitoring in the drug label.
IV Compatibility
There is limited information regarding the compatibility of Levorphanol and IV administrations.
Overdosage
- Most reports of overdosage known to the manufacturer and to the FDA involve three clinical situations. These are: 1. the use of larger than recommended doses or too frequent doses, 2. administration of the drug to children or small adults without any reduction in dosage, and 3. the use of the drug in ordinary dosage in patients compromised by concurrent illness.
- As with all opioids, overdose can occur due to accidental or intentional misuse of this product, especially in infants and children who may gain access to the drug in the home. Based on its pharmacology, levorphanol overdosage would be expected to produce signs of respiratory depression, cardiovascular failure (especially in predisposed patients) and/or central nervous system depression. Serious overdosage with Levorphanol is characterized by respiratory depression (a decrease in respiratory rate and/or tidal volume, periodic breathing, cyanosis), extreme somnolence progressing to stupor or coma, skeletal muscle flaccidity, cold and clammy skin, constricted pupils, and sometimes bradycardia and hypotension. In severe overdosage, apnea, circulatory collapse, cardiac arrest and death may occur.
- Treatment: The specific treatment of suspected levorphanol tartrate overdosage is immediate establishment of an adequate airway and ventilation, followed (if necessary) by intravenous naloxone. The respiratory and cardiac status of the patient should be continuously monitored and appropriate supportive measures instituted, such as oxygen, intravenous fluids and/or vasopressors, if required. Physicians are reminded that the duration of levorphanol action far exceeds the duration of action of naloxone, and repeated dosing with naloxone may be required. Naloxone should be administered cautiously to persons known or suspected to be physically dependent on Levorphanol. In such cases an abrupt and complete reversal of opioid effects may precipitate an acute abstinence syndrome. If necessary to administer naloxone to the physically dependent patient, the antagonist should be administered with extreme care and by titration with smaller than usual doses of the antagonist.
Pharmacology
| |
| |
Levorphanol
| |
| Systematic (IUPAC) name | |
| 17-methylmorphinan-3-ol | |
| Identifiers | |
| CAS number | |
| ATC code | None |
| PubChem | |
| DrugBank | |
| Chemical data | |
| Formula | Template:OrganicBox atomTemplate:OrganicBox atomTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBox atomTemplate:OrganicBoxTemplate:OrganicBox atomTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBox |
| Mol. mass | 257.371 g/mol |
| SMILES | & |
| Pharmacokinetic data | |
| Bioavailability | 70% (oral); 100% (IV) |
| Protein binding | 40% |
| Metabolism | Hepatic |
| Half life | 11-16 hours |
| Excretion | ? |
| Therapeutic considerations | |
| Pregnancy cat. |
C(US) |
| Legal status |
Controlled (S8)(AU) Schedule I(CA) Schedule II(US) Class A (UK) |
| Dependence Liability | High |
| Routes | oral, intravenous, subcutaneous, intramuscular |
Mechanism of Action
- There is limited information about the mechanism of action of Levorphanol.
Structure
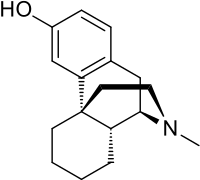
- Levorphanol (levorphanol tartrate) is a potent opioid analgesic with empirical formula C17H23NO•C4H6O6•2H2O and molecular weight 443.5. Each mg of levorphanol tartrate is equivalent to 0.58 mg levorphanol base. Chemically levorphanol is levo-3-hydroxy-N-methylmorphinan. The USP nomenclature is 17-methylmorphinan 3-ol tartrate (1:1)(Salt) dihydrate. The material has 3 asymmetric carbon atoms. The chemical structure is:

- Levorphanol tartrate is a white crystalline powder, soluble in water and ether but insoluble in chloroform.
- Each 1-mL ampul contains 2 mg levorphanol tartrate, 1.8 mg methyl paraben preservative, 0.2 mg propyl paraben preservative, sodium hydroxide to adjust pH to approximately 4.3 and Water for Injection.
- Each milliliter in the 10 mL vials contains 2 mg levorphanol tartrate, 4.5 mg phenol preservative, sodium hydroxide to adjust pH to approximately 4.3 and Water for Injection.
- Each tablet contains 2 mg levorphanol tartrate, lactose, corn starch, stearic acid and talc.
Pharmacodynamics
- Levorphanol is a potent synthetic opioid similar to morphine in its actions. Like other mu-agonist opioids it is believed to act at receptors in the periventricular and periaqueductal gray matter in both the brain and spinal cord to alter the transmission and perception of pain. Onset of analgesia and peak analgesic effect following administration of levorphanol are similar to morphine when administered at equianalgesic doses.
- Levorphanol produces a degree of respiratory depression similar to that produced by morphine at equianalgesic doses, and like many mu-opioid drugs, levorphanol produces euphoria or has a positive effect on mood in many individuals. Two mg of intramuscular levorphanol tartrate depresses respiration to a degree approximately equivalent to that produced by 10 to 15 mg of intramuscular morphine in man. The hemodynamic changes after intravenous administration of levorphanol have not been studied in man but are expected to clinically resemble those seen after morphine.
- As with other opioids, the blood levels required for analgesia are determined by the opioid tolerance of the patient and are likely to rise with chronic use. The rate of development of tolerance is highly variable and is determined by the dose, dosing interval, age, use of concomitant drugs and physical status of the patient. While blood levels of opioid drugs may be helpful in assessing individual cases, dosage is usually adjusted by careful clinical observation of the patient.
Pharmacokinetics
- The pharmacokinetics of levorphanol have been studied in a limited number of cancer patients following intravenous (IV), intramuscular (IM) and oral (PO) administration. Following IV administration, plasma concentrations of levorphanol decline in a triexponential manner with a terminal half-life of approximately 11 to 16 hours and a clearance of 0.78 to 1.1 L/kg/hr. Based on terminal half-life, steady-state plasma concentrations should be achieved by the third day of dosing. Levorphanol is rapidly distributed (<1 hr) and redistributed (1 to 2 hours) following IV administration and has a steady-state volume of distribution of 10 to 13 L/kg. In vitro studies of protein binding indicate that levorphanol is only 40% bound to plasma proteins.
- No pharmacokinetic studies of the absorption of IM levorphanol are available, but clinical data suggests that absorption is rapid with onset of effects within 15 to 30 minutes of administration.
- Levorphanol is well absorbed after PO administration with peak plasma concentrations occurring approximately 1 hour after dosing. The bioavailability of levorphanol tablets compared to IM or IV administration is not known.
- Plasma concentrations of levorphanol following chronic administration in patients with cancer increased with the dose, but the analgesic effect was dependent on the degree of opioid tolerance of the patient. Expected steady-state plasma concentrations for a 6-hour dosing interval can reach 2 to 5 times those following a single dose, depending on the patient’s individual clearance of the drug. Very high plasma concentrations of levorphanol can be reached in patients on chronic therapy due to the long half-life of the drug. One study in 11 patients using the drug for control of cancer pain reported plasma concentrations from 5 to 10 ng/mL after a single 2-mg dose up to 50 to 100 ng/mL after repeated oral doses of 20 to 50 mg/day.
- Animal studies suggest that levorphanol is extensively metabolized in the liver and is eliminated as the glucuronide metabolite. This renally excreted inactive glucuronide metabolite accumulates with chronic dosing in plasma at concentrations that reach fivefold that of the parent compound.
- The effects of age, gender, hepatic and renal disease on the pharmacokinetics of levorphanol are not known. As with all drugs of this class, patients at the extremes of age are expected to be more susceptible to adverse effects because of a greater pharmacodynamic sensitivity and probable increased variability in pharmacokinetics due to age or disease.
Nonclinical Toxicology
There is limited information regarding Levorphanol Nonclinical Toxicology in the drug label.
Clinical Studies
There is limited information regarding Levorphanol Clinical Studies in the drug label.
How Supplied
- Ampuls: 1 mL, 2 mg/mL levorphanol tartrate – boxes of 10 (NDC 0187-3072-10).
- Multiple-Dose Vials: 10 mL, 2 mg/mL levorphanol tartrate – boxes of 1 (NDC 0187-3074-20).
- Scored Oral Tablets: 2 mg round, white, flat beveled edge tablets in bottles of 100 (NDC 0187-3251-10); with LEVO engraved on one side and 3251 and full bisect scored on the other side.
Storage
- Storage: Tablets should be stored at 25°C (77°F); excursions permitted to 15°C- 30°C (59°F - 86°F).
- Dispense in tight containers as defined in USP/NF.
- Parenteral dosage forms should be stored at 25°C (77°F); excursions permitted to 15°C - 30°C (59°F - 86°F).
- DEA Order Form Required.
- Manufactured for:
- Valeant Pharmaceuticals International
- Costa Mesa, CA 92626
- Valeant Pharmaceuticals International
- 3300 Hyland Ave., Costa Mesa, CA 92626 U.S.A.
- 714-545-0100
- 3325197EX03
- Rev. April 2004
Images
Drug Images
{{#ask: Page Name::Levorphanol |?Pill Name |?Drug Name |?Pill Ingred |?Pill Imprint |?Pill Dosage |?Pill Color |?Pill Shape |?Pill Size (mm) |?Pill Scoring |?NDC |?Drug Author |format=template |template=DrugPageImages |mainlabel=- |sort=Pill Name }}
Package and Label Display Panel
{{#ask: Label Page::Levorphanol |?Label Name |format=template |template=DrugLabelImages |mainlabel=- |sort=Label Page }}
Patient Counseling Information
There is limited information regarding Levorphanol Patient Counseling Information in the drug label.
Precautions with Alcohol
Alcohol-Levorphanol interaction has not been established. Talk to your doctor about the effects of taking alcohol with this medication.
Brand Names
There is limited information regarding Levorphanol Brand Names in the drug label.
Look-Alike Drug Names
There is limited information regarding Levorphanol Look-Alike Drug Names in the drug label.
Price
References
The contents of this FDA label are provided by the National Library of Medicine.
{{#subobject:
|Page Name=Levorphanol |Pill Name=Levorphanol pill identifier.jpeg |Drug Name=L-Dromoran |Pill Ingred=Lactose, corn starch, stearic acid, talc|+sep=; |Pill Imprint=LEVO;3251 |Pill Dosage=2 mg |Pill Color=White|+sep=; |Pill Shape=Round |Pill Size (mm)=6.00 |Pill Scoring=2 |Pill Image= |Drug Author=Valeant Pharmaceuticals International |NDC=0187-3072-10
}}
{{#subobject:
|Label Page=Levorphanol |Label Name=Levorphanol label.png
}}