Hepatic failure
| Hepatic failure | |
| ICD-10 | K72.9 |
|---|---|
| DiseasesDB | 5728 |
| MeSH | D017093 |
|
WikiDoc Resources for Hepatic failure |
|
Articles |
|---|
|
Most recent articles on Hepatic failure Most cited articles on Hepatic failure |
|
Media |
|
Powerpoint slides on Hepatic failure |
|
Evidence Based Medicine |
|
Clinical Trials |
|
Ongoing Trials on Hepatic failure at Clinical Trials.gov Trial results on Hepatic failure Clinical Trials on Hepatic failure at Google
|
|
Guidelines / Policies / Govt |
|
US National Guidelines Clearinghouse on Hepatic failure NICE Guidance on Hepatic failure
|
|
Books |
|
News |
|
Commentary |
|
Definitions |
|
Patient Resources / Community |
|
Patient resources on Hepatic failure Discussion groups on Hepatic failure Patient Handouts on Hepatic failure Directions to Hospitals Treating Hepatic failure Risk calculators and risk factors for Hepatic failure
|
|
Healthcare Provider Resources |
|
Causes & Risk Factors for Hepatic failure |
|
Continuing Medical Education (CME) |
|
International |
|
|
|
Business |
|
Experimental / Informatics |
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1] Associate Editor(s)-in-Chief: Roghayeh Marandi[2]
Synonyms and keywords: Liver failure; fulminating hepatic failure
Overview
Liver failure is the inability of the liver to perform its normal synthetic and metabolic function as part of normal physiology.
Historical Perspective
- The original definition of Acute liver failure by Trey and Davidson was in 1959.[1]
- In the late 1980s and early 1990s, more terminologies of Acute liver failure proposed.[2][3]
- Term of Acute-on-chronic Liver failure suggested by Jalan and willimas in 2002.[4]
Classification
Three forms are recognized:
- Acute liver failure -when liver failure occurs rapidly. The most reliable signs of severe acute liver injury are prolonged international normalized ratio ([INR] ≥ 1.5) and any degree of hepatic encephalopathy, in patients without preexisting liver disease and the illness duration is less than 26 weeks.[5][6]
- Acute liver failure was further classified depending on the interval between the onset of jaundice and the onset of encephalopathy into: "Hyperacute " (interval0-7 days), "Acute" (interval 8-28days), "sub-acute" (interval 29days to 12 weeks), "late -onset" (interval 12 -26 weeks).[3][7]
- Chronic liver failure - When liver failure occurs as a result of cirrhosis. It usually means that the liver has been failing gradually for some time (more than 26 weeks), possibly for years. This is called chronic liver failure or End-stage Liver Disease (ESLD).
- Acute on Chronic Liver Failure - when acute hepatic decompensation observed in patients with preexisting chronic liver disease within 4 weeks, characterized by one or more extrahepatic organ failures and with a significantly increased risk of death.[8]
Pathophysiology
- Hepatic injury results in hepatocyte necrosis which occurs due to ATP depletion causing cellular swelling and cell membrane disruptions. Most cases of Acute liver failure (except acute fatty liver of pregnancy and Reye syndrome) will have massive hepatocyte necrosis and/or apoptosis which ultimately result in liver failure. Secondary multi-organ failure occurs after hepatic failure.
The pathophysiology of ALF can be divided into cause‐specific liver injury pathophysiologies and pathophysiology related to the occurrence of secondary multi-organ failure.
- Cause-specific pathophysiologies:[9]
- Immune-mediated hepatocellular injury: :
- Direct hepatocellular injury:
- Toxic metabolites: Acetaminophen, metabolic disorders
- Acetaminophen is predominantly metabolized in the liver through glucuronidation and sulfation, with a small amount metabolized by the cytochrome P450 system. Its metabolite, N-acetyl-p-benzoquinone imine (NAPQI), generated via the P450 pathway is subsequently conjugated by glutathione. In the setting of acetaminophen overdose, glutathione stores may become depleted, resulting in direct hepatocyte injury via NAPQI
- Toxins of Amanita phalloides(amatoxins, phallotoxins, and virotoxins) which inhibit RNA polymerases, cause necrosis of the liver, also partly in the kidney, with the cellular changes causing the fragmentation and segregation of all nuclear components.[10]
- Toxic metabolites: Acetaminophen, metabolic disorders
- Ischemic hepatocellular injury:
- Secondary multiorgan failure is often a result of the initial massive proinflammatory response in reaction to pathogen‐specific molecular patterns (PAMPs) from heterotropic viruses as well as a response to damage‐associated molecular patterns (DAMPs), such as histones, DNA, and high mobility group box‐1 proteins released from injured cells upon hepatocyte death secondary to toxic etiologies which generating a systemic inflammatory response syndrome( SIRS) followed by a compensatory anti‐inflammatory response leading to immune cell dysfunction and sepsis. Dysregulation of systemic vascular tone leads to low systemic vascular resistance, causing hypotension and peripheral vasodilation results in poor pulmonary oxygen exchange, impaired tissue oxygen delivery, and lactic acidosis[11]. Cerebrovascular and renovascular tone are most affected, resulting in cerebral hyperperfusion and hepatic encephalopathy along with functional renal failure. In the brain, altered blood-brain barrier occurs secondary to inflammatory mediators leading to microglial activation, accumulation of glutamine secondary to ammonia crossing the BBB, and subsequent oxidative stress leading to depletion of adenosine triphosphate (ATP) and guanosine triphosphate (GTP). This ultimately leads to astrocyte swelling and cerebral edema and hepatic encephalopathy.
- To put it in the short statement,Acute liver failure results in hemodynamic instability. It is initially associated with hypovolemia due to a combination of poor oral intake and increased fluid loss. As Acute liver failure progresses, the release of circulatory cytokines and inflammatory mediators cause systemic vasodilation and worsens hypotension. The end results are low systemic vascular resistance, systemic hypotension, and increased cardiac output resembling septic shock. These hemodynamic changes lead to decreased peripheral tissue oxygenation and eventually multiorgan failure.
- Chronic liver failure is the result of Cirrhosis which is is an advanced stage of liver fibrosis that is accompanied by distortion of the hepatic vasculature.[12]
- The pathogenesis of Acute-on-chronic liver failure is unclear but many theories are proposed in such as neutrophilic dysfunction that increases the risk of infections, circulating changes, oxidative stress, and toxin hypothesis.
Causes
Causes for Acute liver failure[13][14]:
| Category | Etiology of Acute liver failure |
|---|---|
| Viruses | |
| Drugs |
|
| Metabolic diseases | |
| Toxins |
|
| Vascular diseases | |
| Malignant Infiltration |
|
| Autoimmune disease | |
| Indeterminate |
|
- Acetaminophen hepatotoxicity is the most common cause of ALF in the U.S. and Europe. It results from excessive ingestion of acetaminophen either from suicidal ideations or inadvertent use of supratherapeutic doses for pain control. Increased production of the toxic metabolite N-acetyl-p-benzoquinone imine causes hepatic injury. Acetaminophen toxicity is dose-related with typically at least 10 gram/day required to cause ALF.
- Drug-induced liver injury is the cause of about 50% of ALF cases in the U.S. Many over-the-counter medications, weight loss medications, and prescription medications can lead to acute liver injury. Liver injury from drugs could be dose-dependent and predictable (acetaminophen toxicity) or idiosyncratic and unpredictable (carbamazepine, valproate).
- Viral hepatitis is the most common cause of acute liver failure worldwide and is the predominant cause of ALF in developing countries. The most common viruses are hepatitis A, B, and E infections as well as other rare viral causes including herpes simplex virus, epstein-barr virus, cytomegalovirus, and parvoviruses.
- Amanita phalloides is the most common mushroom to cause hepatotoxicity. History of recent mushroom ingestion should be obtained in patients who present with severe gastrointestinal (GI) symptoms such as nausea, vomiting, abdominal cramping, and diarrhea. Symptoms usually start within 6 to 12 hours of mushroom ingestion. The diagnosis of mushroom poisoning is made clinically because no blood test is available to confirm the diagnosis.
Causes of chronic liver failure
- Chronic liver failure usually occurs in the context of cirrhosis. Cirrhosis must be differentiated from other causes of abnormal liver function tests. In patients with stable cirrhosis, decompensation may occur due to various causes:
- Constipation
- Infection
- Increased alcohol intake
- Medications
- Bleeding from esophageal varices or dehydration
Differential Diagnosis
- Acute liver failure is a distinctive syndrome that is not confused with other conditions. The major differential diagnosis is in the cause of acute liver failure, whether viral (hepatitis A, B, C, D or E), autoimmune, metabolic (Wilson disease), drug-induced or idiopathic.[15][16]
Epidemiology and Demographics
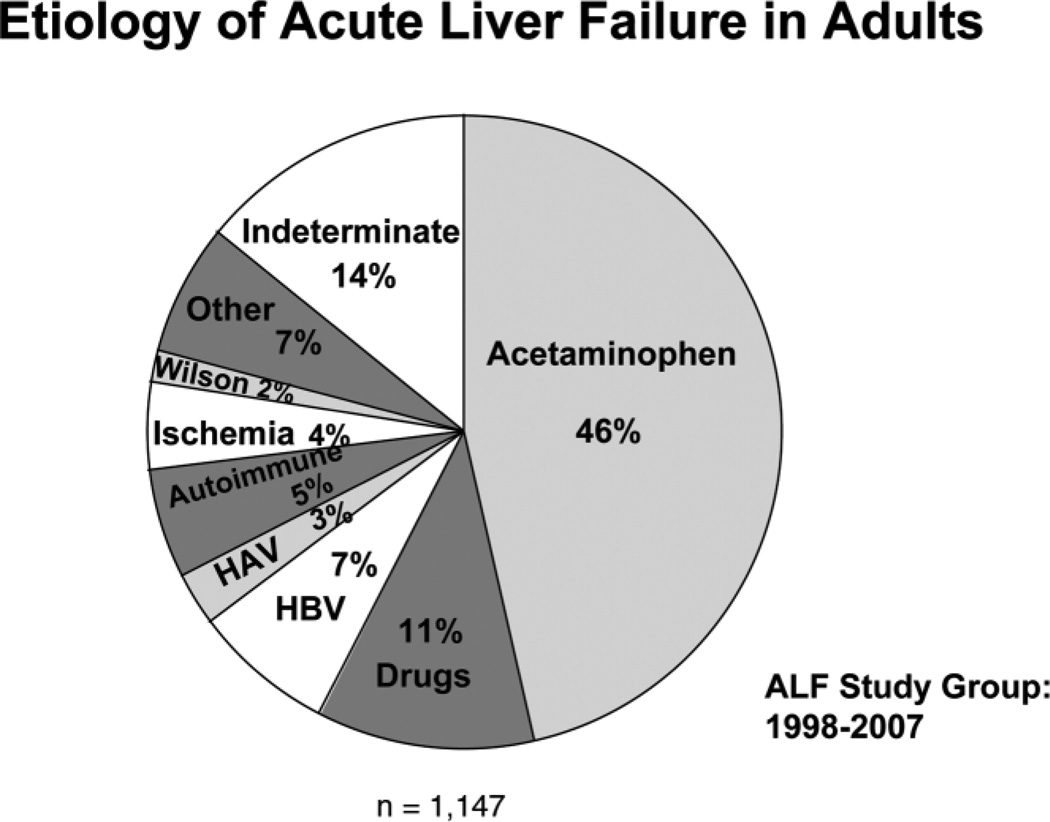
- Acute liver failure affects approximately 2,000–3,000 Americans each year.Acute liver failure was responsible for 3.3% of US liver transplants in 2017.[17]Drug-induced hepatotoxicity occurs for more than 50% of acute liver failure cases including Acetaminophen toxicity (42%) and idiosyncratic drug reactions. Nearly 15% of cases remain of indeterminate etiology.[13][18][19]
- Chronic liver disease and Cirrhosis are the 12(th) leading cause of death in the United States.[20]
Risk factors
- Risk factors for acute liver failure
- Chronic alcohol abuse
- Poor nutritional status
- Female sex
- Age >40 years
- Pregnancy
- Chronic hepatitis B
- Chronic pain and narcotic use
- Complementary and alternative medicine hepatotoxicity
- Acetaminophen and antidepressant therapy
- Chronic hepatitis C
- HIV and hepatitis C coinfection
Screening
Not applicable
Natural History, Complications and Prognosis
Natural History
- Patients who develop severe acute liver injury without preexisting chronic liver disease, often demonstrate significant liver dysfunction with coagulopathy (defined as an international normalized ratio (INR)≥1.5) and are designated as acute liver failure (ALF) when any degree of hepatic encephalopathy (HE) is present.[21]
Clinical features in Hepatic failure and complications
Brain
Liver
- Loss of metabolic function
- Hypoglycemia
- Hyperammonemia
- Coagulopathy
- Lactic acidosis
Kidney and adrenal
System
Heart
- High output state
- Subclinical myocardial injury
- Hepatocardiac syndrome
Pancreas
Lungs
- Acute lung injury
- Acute respiratory distress syndrome(ARDS)
- Hepatopulmonary syndrome
Bone marrow
- Frequent suppression
- Anemia
- Thrombocytopenia
Prognosis
prognosis of Acute liver failure
- The prognosis in patients with acute liver failure is highly variable and depends on the etiology, subtypes (hyperacute, acute,...), age, and the degree of coagulopathy. Determining the prognosis for these patients is vital. The overall mortality of ALF is currently between 30% to 40%.[13] Liver transplantation has dramatically improved short-term survival in patients with acute liver failure. Still, 25% to 45% of patients will survive with medical treatment.[22]
- Identification of patients who will eventually require liver transplantation should be addressed through continuous medical assessment. The most widely accepted prognostic tool for patients who present with ALF is King's College Criteria (KCC) .Although his scoring system is generally quite accurate in predicting poor prognosis and, along with clinical judgment, is useful for ensuring timely transfer to a liver transplant center in adults, but data suggest they may not reliably predict outcomes in the pediatric population.[23][19][24][22]
Prognosis of chronic liver failure
- Patients with compensated cirrhosis have a median survival of 6–12 years. Decompensation and end stage liver disease occurs in 5%–7% annually; median survival then declines to 2 years.[25]
- Several retrospectives studies have reported in patients with end-stage liver disease, the MELD score to have similar predictive value to the king's college criteria for mortality associated with ALF.
Diagnosis
History and Symptoms | Physical Examination | Laboratory Findings | Electrocardiogram | Chest X Ray | CT | MRI | Echocardiography or Ultrasound | Other Imaging Findings | Other Diagnostic Studies | Clinical prediction rules
History and symptoms
Social history
- History of alcohol use:
- Amount
- Duration
- History of illicit drug use
- History of unprotected sexual intercourse
- History of recent travel
Past Medical history
- History of infections:[26]
- Hepatitis B
- Hepatitis C
- History of autoimmune disorders:
- History of blood transfusions
- History of current or prior depression (including assessment of suicidality), anxiety, psychosis, or other mental illness.
Menstrual history
- History of menstrual irregularities
Family history
- Family history of liver disease:
Medication history
- History of use of all medications used over the last 6 months, including prescription medications, over-the-counter agents, herbal supplements, wild mushrooms, or other alternatives/complementary therapies;
Symptoms
- Liver failure may present with[27]
- Jaundice
- Increase in abdominal girth due to ascites
- Pruritus
- Signs of upper gastrointestinal bleeding:
- Symptoms due to hepatic encephalopathy:
- Altered mental status
- Confusion
- Sleep disturbances
- Muscle cramps due to reduction in effective circulating plasma volume
- Lower extremity edema
Physical Examination
- Complete physical examination should be performed.
Appearance of the patient
Vital signs
In patients with hepatic failure vital signs include:[30]
- Blood pressure may be normal, low or high.
- Hypothermia or hyperthermia may be present
- Tachycardia with regular or iregularpulse may be present
- Tachypnea
Skin
Signs of liver disease may be seen, such as:[31]
- Yellow skin
- Protracted and disabling pruritus
- Spider angiomas
- Palmar erythema
HEENT
- Icteric sclera[31]
- Dilated pupils, sluggishly responsive to light[32]
- Ophthalmoscopic exam may be abnormal with findings of papilledema[33]
Neck
Physical examination of neck is usually normal but in Hepatic failure due to CHF,Jugular vein distention may be seen.
Lungs
Heart
Physical examination of heart is usually normal unless the cause of hepatic failure is Heart disease such as Right heart failure.
Abdomen
Signs of liver disease may be seen such as:
- Fluid collection in the abdomen (ascites)[34]
- Abdominal tenderness[35]
- A palpable liver in the epigastrium (hepatomegaly- feel with inspiration, relocate during expiration)[36]
Back
Physical examination of back is usually normal
Genitourinary
- Small testicles
- Oliguria
- signs of renal dysfunction
Neuromuscular
- Jerking movement of the limbs (asterixis) is highly suggestive of hepatic encephalopathy[37]{{#ev:youtube|Or65nOrcz1A}}
- Attention deficit and slow information processing[38]
- Abnormal paper and pencil tests[38]
- Altered mental status may be seen[39]
- Scoring on Glasgow coma scale may be low
- Clonus may be present[32]
- Hyporeflexia[40]
- Positive (abnormal) Babinski reflex[41]
- Bilaterally muscle weakness may be seen[28]
- Examination cranial nerves is usually normal, but features of raised ICP(unequal and fixed pupils) may be seen.
Extremities
- Athetosis (writhing, sinuous movements of the digits and extremities) may be present
Laboratory Findings
- Prolonged prothrombin time(INR>1.5)
- Elevated aminotransferase level
- Elevated bilirubin level
- low platelet count
- Elevated ammonia level
- Elevated serum Cr, BUN
- Elevated amylase and lipase
- Hypoglycemia
- Hypokalemia
- Hypophosphatemia
- Acidosis or alkalosis
- Elevated LDH level
- laboratory tests are recommended for establishing an etiology and determining the prognosis of Acute liver failure:
|
Imaging
- Imaging is not required for diagnosis but it is useful in the correct clinical context, for example:
- Abdominal ultrasound with Doppler to confirm portal and hepatic vein patency
- CXR for evaluation of lungs
- Non-contrast computed tomography (CT) scan of the head for patients with Hepatic encephalopathy
Treatment
The most important part of the management of hepatic failure involves the timely diagnosis of it. Making a timely diagnosis in a patient who presents with liver dysfunction and an altered mental state remains the single most important management step for the clinician, as a delay can lead to substantial morbidity and mortality. Although there is no proven therapy for ALF, understanding the progression of ALF, from loss of hepatocytes to the development of multiorgan failure, helps the clinician in disease-specific complication management.
The management of Hepatic failure should involve
- Specific Treatment
- Supportive and symptomatic management
- Management of complications
- Emergency therapies
- Liver transplantation
1. Specific treatment:
- Identification of the etiology and initiation of specific treatment.
Acetaminophen intoxication
- Oral NAC: 140 mg/kg loading dose, then 70 mg/kg every 4 hours until discontinued by hepatology or transplantation surgery attending physician
- Or
- IV NAC: 150 mg/kg loading dose, then 50 mg/kg IV over 4 hours, then 100 mg/kg IV over 16 hours as a continuous infusion until discontinued by hepatology or transplantation surgery attending physician
- Charcoal: via NGT every 4 hours alternating with silymarin
- Penicillin G: 1 g/kg/day IV and
- NAC (Dosing as for acetaminophen overdose.),
- Silymarin: 300 mg PO/NGT every 12 hours,
- Legalon-SIL: 5 mg/kg/day IV (given in 4 divided doses) or 5 mg/kg IV loading dose followed by 20 mg/kg/day via continuous infusion
- Acyclovir: 10 mg/kg IV every 8 hours (using IBW) adjusted for kidney function
- Ganciclovir: 5 mg/kg IV every 12 hours (using IBW) adjusted for kidney function
Hepatitis B virus infection
- Delivery of the fetus
2. Supportive and symptomatic management
- Timely transfer to the critical care unit
- Check clinical status continuously
- Fluid restriction
- IV H2 Blockers or PPI
- Antibiotics
- Prevention of hypoglycemia
- Avoid sedation
- 12 hourly electrolytes and coagulation studies
3. Management of Complications:
Treatment of Hepatic encephalopathy:
Even minimal hepatic encephalopathy may benefit from treatment.[43]
- Reduce protein intake:
Traditionally it has been presumed that excessive protein intake leads to increased generation of ammonia, which, in the setting of severe liver impairment, will accumulate and worsen the hepatic encephalopathy. While very large protein loads (such as gastrointestinal hemorrhage, because blood is rich in protein) are known to precipitate encephalopathy, the need for patients with chronic liver disease patients to be protein restricted has been disproven.[44] Indeed, because chronic liver disease is a catabolic state, a protein restricted diet would lead to protein malnutrition and a negative nitrogen balance.
- Correction of hypokalemia:
Concomitant hypokalemia should be corrected as hypokalemia increases renal ammonia production and may promote conversion of ammonium into ammonia which can cross the blood-brain barrier.[45]
Lactulose is a compound that will cause osmotic diarrhea, thus lessening the time available for intestinal bacteria to metabolize protein into ammonia within the bowel. Further, it acidifies the environment in the lumen of the bowel. This promotes the conversion of lumenal ammonia (NH3) to ammonium (NH4+) which, by which virtue of its net charge, should be less readily absorbed into the bloodstream from the bowel lumen. Despite this theoretical and appealing mechanism, a meta-analysis of randomized controlled trials by the international Cochrane Collaboration found benefit, but suggests there is little evidence for its preferred use to treat hepatic encephalopathy.[46] Indeed, any drug (laxative) which speeds up transit through the bowel thereby lessening the time available for bacteria to metabolize protein into ammonia, works just as well.
- Lactulose can be given rectally for patients who cannot take oral medications.[47][48][49] One regimen is 300 mL (200 gm) of lactulose syrup (10 gm/15 ml) in 1 L of water which is retained for 1 hour, with the patient in the Trendelenburg position.[50]
Antibiotics may be given to kill bacteria present in the bowel thereby decreasing bacterial conversion of protein to ammonia (and other toxic substances) there. Although effective, neomycin, a non-absorbable aminoglycoside antibiotic, is essentially contraindicated; it has been found that a proportion of the ingested dose is indeed absorbed due to increased gut permeability, thus increasing the risk of renal failure and hearing loss (i.e. two of the potential side effects of neomycin). The former side-effect, in particular, is especially worrisome given the already increased likelihood of renal failure in cirrhosis and portal hypertension (i.e. hepatorenal syndrome). Metronidazole has also been studied.[51]
Rifaximin , receieved orphan drug status in 2005 for the treatment of hepatic encephalopathy. In contrast to neomycin, its tolerability profile is comparable to placebo.[52] Multiple clinical trials have demonstrated that rifaximin at a dose of 400 mg taken orally 3 times a day was as effective as lactulose or lactilol at improving hepatic encephalopathy symptoms.[53] Similarly, rifaximin was as effective as neomycin and paromomycin.[54] Rifaximin was better tolerated than both the cathartics and the other nonabsorbable antibiotics. A number of concerns remain regarding rifaximin's role in the treatment of hepatic encephalopathy. It remains to be determined if rifaximin can improve severe encephalopathy symptoms as rapidly as lactulose. There are also concerns regarding the cost-effectiveness of the medication.
- Benzodiazepine receptor antagonists:
A meta-analysis of randomized controlled trials by the international cochrane collaboration found benefit from flumazenil.[55] The doses of flumazenil varied around a median of 2 milligrams over 10 minutes: flumazenil was given as a continuous infusion (12 trials), preceded by bolus injections in two trials. One trial used only bolus injections. Patients received flumazenil at a total dose ranging from 0.2 to 19.5 milligram (median 2 milligram). The median duration of treatment was 10 minutes (range one minute to 72 hours)'. However, the benefit was short.
L-ornithine-L-aspartate stimulates the urea cycle, and has shown encouraging results in randomized controlled trials.[56][57][58]
Contraindicated medications
Hepatic coma is considered an absolute contraindication to the use of the following medications:
Treatment of cerebral edema:
The goal in the management of ICH is to lower the ICP to less than 20 to 25 mm Hg and maintain the cerebral perfusion pressure above 50 to 60 mm Hg. This is mainly performed by increasing the mean arterial pressure (MAP) and decreasing the ICP by the methods mentioned below:[59]
- IV mannitol at the dose of 0.5 to 1.0 g/kg
- Hypertonic saline delayed the development of ICH
- Hyperventilation decreases ICP
- Hypothermia decreases ICP
- Seizure control
- Avoid hyperthermia
Treatment of hepatorenal syndrome:
Because of the high mortality associated with hepatorenal syndrome, emphasis is on prevention in patients who are at risk for the condition. Strategies for avoiding hepatorenal syndrome include appropriate and non-aggressive use of diuretics, identification and early treatment of infection and hemorrhage, and avoidance of other toxins that can affect both the liver and kidney.[60]
The definitive treatment for hepatorenal syndrome is liver transplantation, and all other therapies can best be described as bridges to transplantation. These treatment strategies include the following:[61]
Albumin
All major studies showing improvement in renal function in patients with hepatorenal syndrome have involved expansion of the volume of the plasma with albumin given intravenously [62][63] One regimen is 1 gm albumin per kg of body weight intravenously on day one followed by followed by 20-40 grams daily.[64]
Midodrine and octreotide
Midodrine is an alpha-agonist and octreotide is an analog of somatostatin. The medications are respectively systemic vasoconstrictors and inhibitors of vasodilators, and were not found to be useful when used individually in the treatment of the hepatorenal syndrome.[65] However, one study of 13 patients with hepatorenal syndrome showed significant improvement when the two were used together (with midodrine given orally, octreotide given subcutaneously and both dosed according to blood pressure), with three patients surviving to discharge.[66] A nonrandomized, observational study used "100 μg subcutaneously TID, with the goal to increase the dose to 200 μg subcutaneous TID" and "midodrine administration started at 5, 7.5, or 10 mg TID orally, with the goal to increase the dose to 12.5 or 15 mg if necessary" and found that "octreotide/midodrine treatment appears to improve 30-day survival".[67]
Vasopressin analogues
The vasopressin analogue ornipressin was found in a number of studies to be useful in improvement of renal function in patients with hepatorenal syndrome,[62][68] but has been limited by ischemic complications[62]. Terlipressin is a vasopressin analogue that has been found in one study to be useful for improving renal function in patients with hepatorenal syndrome with a lesser incidence of ischemia.[63] Neither medication is available for use in North America.
Transjugular intrahepatic portosystemic shunt

Transjugular intrahepatic portosystemic shunts (TIPS) involve decompression of the high pressures in the portal circulation by placing a small stent between a portal and hepatic vein. They have also been shown to improve renal function in patients with hepatorenal syndrome.[69][70]
Liver dialysis
Liver dialysis involves extracorporeal dialysis to remove toxins from the circulation. The molecular adsorbents recirculation system (MARS) has shown some utility as a bridge to transplantation in patients with hepatorenal syndrome.[71]
Hemodialysis
Renal replacement therapy may be required to 'bridge' the patient to liver transplantation, although the condition of the patient may dictate the modality used.[72]
Other medications
Other agents used in treatment include
Management of Coagulopathy[5]
- Routine correction of thrombocytopenia or elevated INR by plasma infusion, in the absence of bleeding, is not indicated in acute liver failure, because of the low incidence of bleeding manifestations in ALF and the risk of volume expansion with plasma replacement. In addition, INR is an important prognostic indicator in ALF, correction of coagulopathy would alter the INR and interfere in the assessment of prognosis.
- Routine administration of vitamin K (5 to10 mg subcutaneously)
- plasma or clotting factor replacement therapy in clinically significant bleeding or the need for a procedure with a high bleeding risk such as ICP monitor insertion. Recombinant activated factor VII is used if INR is still high to correct coagulopathy in these patients.
- Patients with thrombocytopenia with platelet count less than 50,000 cells/mm3 with clinically significant bleeding should receive platelet transfusion. In the absence of bleeding, there is no need to initiate platelet transfusion.[5]
Management of metabolic abnormalities
- Intravenous glucose should be administered for the prophylaxis and treatment of hypoglycemia.
- Electrolyte abnormalities should be promptly identified and corrected urgently.
- Enteral feeding should be started at the earliest in patients who are unlikely to resume oral nutrition within 5 days. Parenteral feeding should be considered when enteral feeding cannot be instituted or is contraindicated, though parenteral feeding is associated with an increased risk of infections. Both of them reduce stress-related gastric ulcers.[59]
- Serum ammonia levels should be monitored and protein load should be adjusted accordingly.
Treatment of hemodynamic instabilty
- Invasive hemodynamic monitoring
- Colloid, crystalloid fluids, and blood products
- Vassopressor
4. Emergency therapies
- Extracorporeal system to bridge liver transplantation or regenerating of the native liver and removing toxic substances:
- Artificial support systems: are extracorporeal devices that have either charcoal or other adherent particles in an extracorporeal circuit to help with detoxification. Unfortunately, randomized control trials have not shown conclusive benefits with these devices in patients and further study is needed.
- Bioartificial systems use cryopreserved cells and they are able to not only detoxify but also perform synthetic liver functions.
5. Liver transplantation
- Early consultation with liver transplant specialists and transfer of patients to a liver transplant center when necessary
Contraindicated medications
Severe hepatic failure is considered an absolute contraindication to the use of the following medications:
- Carvedilol
- Conjugated estrogens/bazedoxifene
- Diclofenamide
- Dronedarone
- Nebivolol
- Rosuvastatin
- Simvastatin
- Spironolactone
- Sulfamethoxazole/Trimethoprim
- Doxorubicin Hydrochloride
- Tipranavir
The ALFSG index is a newer option that may be more accurate.[75]
References
- ↑ Riordan SM, Williams R (May 2008). "Perspectives on liver failure: past and future". Semin. Liver Dis. 28 (2): 137–41. doi:10.1055/s-2008-1073113. PMID 18452113.
- ↑ Bernuau J, Rueff B, Benhamou JP (May 1986). "Fulminant and subfulminant liver failure: definitions and causes". Semin. Liver Dis. 6 (2): 97–106. doi:10.1055/s-2008-1040593. PMID 3529410.
- ↑ 3.0 3.1 O'Grady JG, Schalm SW, Williams R (July 1993). "Acute liver failure: redefining the syndromes". Lancet. 342 (8866): 273–5. doi:10.1016/0140-6736(93)91818-7. PMID 8101303.
- ↑ Jalan R, Williams R (2002). "Acute-on-chronic liver failure: pathophysiological basis of therapeutic options". Blood Purif. 20 (3): 252–61. doi:10.1159/000047017. PMID 11867872.
- ↑ 5.0 5.1 5.2 Stravitz RT, Lee WM (September 2019). "Acute liver failure". Lancet. 394 (10201): 869–881. doi:10.1016/S0140-6736(19)31894-X. PMID 31498101.
- ↑ Rajaram P, Subramanian R (October 2018). "Acute Liver Failure". Semin Respir Crit Care Med. 39 (5): 513–522. doi:10.1055/s-0038-1673372. PMID 30485882.
- ↑ Gimson AE, O'Grady J, Ede RJ, Portmann B, Williams R (1986). "Late onset hepatic failure: clinical, serological and histological features". Hepatology. 6 (2): 288–94. doi:10.1002/hep.1840060222. PMID 3082735.
- ↑ Moreau R, Jalan R, Gines P, Pavesi M, Angeli P, Cordoba J, Durand F, Gustot T, Saliba F, Domenicali M, Gerbes A, Wendon J, Alessandria C, Laleman W, Zeuzem S, Trebicka J, Bernardi M, Arroyo V (June 2013). "Acute-on-chronic liver failure is a distinct syndrome that develops in patients with acute decompensation of cirrhosis". Gastroenterology. 144 (7): 1426–37, 1437.e1–9. doi:10.1053/j.gastro.2013.02.042. PMID 23474284.
- ↑ Dong V, Nanchal R, Karvellas CJ (February 2020). "Pathophysiology of Acute Liver Failure". Nutr Clin Pract. 35 (1): 24–29. doi:10.1002/ncp.10459. PMID 31840297.
- ↑ Vetter J (January 1998). "Toxins of Amanita phalloides". Toxicon. 36 (1): 13–24. doi:10.1016/s0041-0101(97)00074-3. PMID 9604278.
- ↑ Chung RT, Stravitz RT, Fontana RJ, Schiodt FV, Mehal WZ, Reddy KR, Lee WM (September 2012). "Pathogenesis of liver injury in acute liver failure". Gastroenterology. 143 (3): e1–e7. doi:10.1053/j.gastro.2012.07.011. PMC 3641754. PMID 22796239.
- ↑ Schuppan D, Afdhal NH (March 2008). "Liver cirrhosis". Lancet. 371 (9615): 838–51. doi:10.1016/S0140-6736(08)60383-9. PMC 2271178. PMID 18328931.
- ↑ 13.0 13.1 13.2 Lee WM, Squires RH, Nyberg SL, Doo E, Hoofnagle JH (April 2008). "Acute liver failure: Summary of a workshop". Hepatology. 47 (4): 1401–15. doi:10.1002/hep.22177. PMC 3381946. PMID 18318440.
- ↑ Watkins PB, Kaplowitz N, Slattery JT, Colonese CR, Colucci SV, Stewart PW, Harris SC (July 2006). "Aminotransferase elevations in healthy adults receiving 4 grams of acetaminophen daily: a randomized controlled trial". JAMA. 296 (1): 87–93. doi:10.1001/jama.296.1.87. PMID 16820551.
- ↑ Chayanupatkul M, Schiano TD (February 2020). "Acute Liver Failure Secondary to Drug-Induced Liver Injury". Clin Liver Dis. 24 (1): 75–87. doi:10.1016/j.cld.2019.09.005. PMID 31753252.
- ↑ Murray KF, Hadzic N, Wirth S, Bassett M, Kelly D (October 2008). "Drug-related hepatotoxicity and acute liver failure". J. Pediatr. Gastroenterol. Nutr. 47 (4): 395–405. doi:10.1097/MPG.0b013e3181709464. PMID 18852631.
- ↑ Kim WR, Lake JR, Smith JM, Schladt DP, Skeans MA, Noreen SM, Robinson AM, Miller E, Snyder JJ, Israni AK, Kasiske BL (February 2019). "OPTN/SRTR 2017 Annual Data Report: Liver". Am. J. Transplant. 19 Suppl 2: 184–283. doi:10.1111/ajt.15276. PMID 30811890.
- ↑ Larson AM, Polson J, Fontana RJ, Davern TJ, Lalani E, Hynan LS, Reisch JS, Schiødt FV, Ostapowicz G, Shakil AO, Lee WM (December 2005). "Acetaminophen-induced acute liver failure: results of a United States multicenter, prospective study". Hepatology. 42 (6): 1364–72. doi:10.1002/hep.20948. PMID 16317692.
- ↑ 19.0 19.1 Patton H, Misel M, Gish RG (March 2012). "Acute liver failure in adults: an evidence-based management protocol for clinicians". Gastroenterol Hepatol (N Y). 8 (3): 161–212. PMC 3365519. PMID 22675278.
- ↑ Asrani SK, Larson JJ, Yawn B, Therneau TM, Kim WR (August 2013). "Underestimation of liver-related mortality in the United States". Gastroenterology. 145 (2): 375–82.e1–2. doi:10.1053/j.gastro.2013.04.005. PMC 3890240. PMID 23583430.
- ↑ Koch DG, Speiser JL, Durkalski V, Fontana RJ, Davern T, McGuire B, Stravitz RT, Larson AM, Liou I, Fix O, Schilsky ML, McCashland T, Hay JE, Murray N, Shaikh OS, Ganger D, Zaman A, Han SB, Chung RT, Brown RS, Munoz S, Reddy KR, Rossaro L, Satyanarayana R, Hanje AJ, Olson J, Subramanian RM, Karvellas C, Hameed B, Sherker AH, Lee WM, Reuben A (September 2017). "The Natural History of Severe Acute Liver Injury". Am. J. Gastroenterol. 112 (9): 1389–1396. doi:10.1038/ajg.2017.98. PMC 5587371. PMID 28440304.
- ↑ 22.0 22.1 Mendizabal M, Silva MO (January 2016). "Liver transplantation in acute liver failure: A challenging scenario". World J. Gastroenterol. 22 (4): 1523–31. doi:10.3748/wjg.v22.i4.1523. PMC 4721985. PMID 26819519.
- ↑ Sundaram V, Shneider BL, Dhawan A, Ng VL, Im K, Belle S, Squires RH (February 2013). "King's College Hospital Criteria for non-acetaminophen induced acute liver failure in an international cohort of children". J. Pediatr. 162 (2): 319–23.e1. doi:10.1016/j.jpeds.2012.07.002. PMC 3504621. PMID 22906509.
- ↑ McDowell Torres D, Stevens RD, Gurakar A (July 2010). "Acute liver failure: a management challenge for the practicing gastroenterologist". Gastroenterol Hepatol (N Y). 6 (7): 444–50. PMC 2933761. PMID 20827368.
- ↑ Potosek J, Curry M, Buss M, Chittenden E (November 2014). "Integration of palliative care in end-stage liver disease and liver transplantation". J Palliat Med. 17 (11): 1271–7. doi:10.1089/jpm.2013.0167. PMC 4229716. PMID 25390468.
- ↑ Flores YN, Lang CM, Salmerón J, Bastani R (2012). "Risk factors for liver disease and associated knowledge and practices among Mexican adults in the US and Mexico". J Community Health. 37 (2): 403–11. doi:10.1007/s10900-011-9457-4. PMID 21877109.
- ↑ Bloom S, Kemp W, Lubel J (2015). "Portal hypertension: pathophysiology, diagnosis and management". Intern Med J. 45 (1): 16–26. doi:10.1111/imj.12590. PMID 25230084.
- ↑ 28.0 28.1 Bleibel W, Al-Osaimi AM (2012). "Hepatic encephalopathy". Saudi J Gastroenterol. 18 (5): 301–9. doi:10.4103/1319-3767.101123. PMC 3500018. PMID 23006457.
- ↑ Salam M, Matherly S, Farooq IS, Stravitz RT, Sterling RK, Sanyal AJ; et al. (2012). "Modified-orientation log to assess hepatic encephalopathy". Aliment Pharmacol Ther. 35 (8): 913–20. doi:10.1111/j.1365-2036.2012.05038.x. PMC 3616137. PMID 22348593.
- ↑ Wannhoff A, Nusshag C, Stremmel W, Merle U (2017). "Slow ventricular tachycardia presenting with acute liver failure". SAGE Open Med Case Rep. 5: 2050313X17718100. doi:10.1177/2050313X17718100. PMC 5528920. PMID 28835821.
- ↑ 31.0 31.1 Dogra S, Jindal R (2011). "Cutaneous manifestations of common liver diseases". J Clin Exp Hepatol. 1 (3): 177–84. doi:10.1016/S0973-6883(11)60235-1. PMC 3940632. PMID 25755383.
- ↑ 32.0 32.1 Shawcross DL, Wendon JA (2012). "The neurological manifestations of acute liver failure". Neurochem Int. 60 (7): 662–71. doi:10.1016/j.neuint.2011.10.006. PMID 22067133.
- ↑ Crippin JS, Gross JB, Lindor KD (1992). "Increased intracranial pressure and hepatic encephalopathy in chronic liver disease". Am J Gastroenterol. 87 (7): 879–82. PMID 1615943.
- ↑ Losowsky MS, Scott BB (1973). "Ascites and oedema in liver disease". Br Med J. 3 (5875): 336–8. PMC 1586440. PMID 4579401.
- ↑ Riley TR, Koch K (2003). "Characteristics of upper abdominal pain in those with chronic liver disease". Dig Dis Sci. 48 (10): 1914–8. PMID 14627332.
- ↑ McCormick PA, Nolan N (2004). "Palpable epigastric liver as a physical sign of cirrhosis: a prospective study". Eur J Gastroenterol Hepatol. 16 (12): 1331–4. PMID 15618841.
- ↑ Invalid
<ref>tag; no text was provided for refs namedpmid27089111 - ↑ 38.0 38.1 Nabi E, Bajaj JS (2014). "Useful tests for hepatic encephalopathy in clinical practice". Curr Gastroenterol Rep. 16 (1): 362. doi:10.1007/s11894-013-0362-0. PMC 3918211. PMID 24357348.
- ↑ Rahimi RS, Elliott AC, Rockey DC (2013). "Altered mental status in cirrhosis: etiologies and outcomes". J Investig Med. 61 (4): 695–700. doi:10.2310/JIM.0b013e318289e254. PMID 23474970.
- ↑ ADAMS RD, FOLEY JM (1953). "The neurological disorder associated with liver disease". Res Publ Assoc Res Nerv Ment Dis. 32: 198–237. PMID 13134644.
- ↑ Djiambou-Nganjeu H (2017). "Hepatic Encephalopathy in Liver Cirrhosis". J Transl Int Med. 5 (1): 64–67. doi:10.1515/jtim-2017-0013. PMC 5490964. PMID 28680841.
- ↑ Izumi S, Langley PG, Wendon J, Ellis AJ, Pernambuco RB, Hughes RD, Williams R (June 1996). "Coagulation factor V levels as a prognostic indicator in fulminant hepatic failure". Hepatology. 23 (6): 1507–11. doi:10.1002/hep.510230630. PMID 8675171.
- ↑ Prasad S, Dhiman RK, Duseja A, Chawla YK, Sharma A, Agarwal R (2007). "Lactulose improves cognitive functions and health-related quality of life in patients with cirrhosis who have minimal hepatic encephalopathy". Hepatology. 45 (3): 549–59. doi:10.1002/hep.21533. PMID 17326150.
- ↑ Córdoba J, López-Hellín J, Planas M; et al. (2004). "Normal protein diet for episodic hepatic encephalopathy: results of a randomized study". J. Hepatol. 41 (1): 38–43. doi:10.1016/j.jhep.2004.03.023. PMID 15246205.
- ↑ Artz SA, Paes IC, Faloon WW (1966). "Hypokalemia-induced hepatic coma in cirrhosis. Occurrence despite neomycin therapy". Gastroenterology. 51 (6): 1046–53. PMID 5958605.
- ↑ Als-Nielsen B, Gluud L, Gluud C. "Nonabsorbable disaccharides for hepatic encephalopathy". Cochrane Database Syst Rev: CD003044. PMID 15106187.
- ↑ Kersh ES, Rifkin H (1973). "Lactulose enemas". Ann. Intern. Med. 78 (1): 81–4. PMID 4682313.
- ↑ Ratnaike RN, Hicks EP, Hislop IG (1975). "The rectal administration of lactulose". Australian and New Zealand journal of medicine. 5 (2): 137–40. PMID 240347.
- ↑ Uribe M, Campollo O, Vargas F; et al. (1987). "Acidifying enemas (lactitol and lactose) vs. nonacidifying enemas (tap water) to treat acute portal-systemic encephalopathy: a double-blind, randomized clinical trial". Hepatology. 7 (4): 639–43. PMID 3301614.
- ↑ Blei AT, Córdoba J (2001). "Hepatic Encephalopathy". Am. J. Gastroenterol. 96 (7): 1968–76. doi:10.1111/j.1572-0241.2001.03964.x. PMID 11467622.
- ↑ Morgan MH, Read AE, Speller DC (1982). "Treatment of hepatic encephalopathy with metronidazole". Gut. 23 (1): 1–7. PMID 7035298.
- ↑ Williams R, James OF, Warnes TW, Morgan MY (2000). "Evaluation of the efficacy and safety of rifaximin in the treatment of hepatic encephalopathy: a double-blind, randomized, dose-finding multi-centre study". European journal of gastroenterology & hepatology. 12 (2): 203–8. PMID 10741936.
- ↑ Bucci L, Palmieri GC (1993). "Double-blind, double-dummy comparison between treatment with rifaximin and lactulose in patients with medium to severe degree hepatic encephalopathy". Current medical research and opinion. 13 (2): 109–18. PMID 8325041.
- ↑ Pedretti G, Calzetti C, Missale G, Fiaccadori F (1991). "Rifaximin versus neomycin on hyperammoniemia in chronic portal systemic encephalopathy of cirrhotics. A double-blind, randomized trial". The Italian journal of gastroenterology. 23 (4): 175–8. PMID 1751811.
- ↑ Als-Nielsen B, Gluud LL, Gluud C (2004). "Benzodiazepine receptor antagonists for hepatic encephalopathy". Cochrane database of systematic reviews (Online) (2): CD002798. doi:10.1002/14651858.CD002798.pub2. PMID 15106178.
- ↑ Poo J, Góngora J, Sánchez-Avila F, Aguilar-Castillo S, García-Ramos G, Fernández-Zertuche M, Rodríguez-Fragoso L, Uribe M (2006). "Efficacy of oral L-ornithine-L-aspartate in cirrhotic patients with hyperammonemic hepatic encephalopathy. Results of a randomized, lactulose-controlled study". Ann Hepatol. 5 (4): 281–8. PMID 17151582.
- ↑ Poo JL, Góngora J, Sánchez-Avila F; et al. (2006). "Efficacy of oral L-ornithine-L-aspartate in cirrhotic patients with hyperammonemic hepatic encephalopathy. Results of a randomized, lactulose-controlled study". Annals of hepatology : official journal of the Mexican Association of Hepatology. 5 (4): 281–8. PMID 17151582.
- ↑ Stauch S, Kircheis G, Adler G; et al. (1998). "Oral L-ornithine-L-aspartate therapy of chronic hepatic encephalopathy: results of a placebo-controlled double-blind study". J. Hepatol. 28 (5): 856–64. PMID 9625322.
- ↑ 59.0 59.1 Plauth M, Schuetz T (November 2009). "Hepatology - Guidelines on Parenteral Nutrition, Chapter 16". Ger Med Sci. 7: Doc12. doi:10.3205/000071. PMC 2795384. PMID 20049084.
- ↑ Cavallin M, Fasolato S, Marenco S, Piano S, Tonon M, Angeli P (2015). "The Treatment of Hepatorenal Syndrome". Dig Dis. 33 (4): 548–54. doi:10.1159/000375346. PMID 26159272.
- ↑ Fabrizi F, Aghemo A, Messa P (2013). "Hepatorenal syndrome and novel advances in its management". Kidney Blood Press Res. 37 (6): 588–601. doi:10.1159/000355739. PMID 24356549.
- ↑ 62.0 62.1 62.2 Guevara M, Gines P, Fernandez-Esparrach G, Sort P, Salmeron JM, Jimenez W, Arroyo V, Rodes J. Reversibility of hepatorenal syndrome by prolonged administration of ornipressin and plasma volume expansion. Hepatology 1998 Jan;27(1):35-41. PMID 9425914
- ↑ 63.0 63.1 Ortega R, Gines P, Uriz J, Cardenas A, Calahorra B, De Las Heras D, Guevara M, Bataller R, Jimenez W, Arroyo V, Rodes J. Terlipressin therapy with and without albumin for patients with hepatorenal syndrome: results of a prospective, nonrandomized study. Hepatology 2002 Oct;36 (4 Pt 1):941-8. PMID 12297842
- ↑ Ginès P, Cárdenas A, Arroyo V, Rodés J (2004). "Management of cirrhosis and ascites". N. Engl. J. Med. 350 (16): 1646–54. doi:10.1056/NEJMra035021. PMID 15084697.
- ↑ Pomier-Layrargues G, Paquin SC, Hassoun Z, Lafortune M, Tran A. Octreotide in hepatorenal syndrome: a randomized, double-blind, placebo-controlled, crossover study. Hepatology 2003 Jul;38(1):238-43.
- ↑ Angeli P, Volpin R, Gerunda G, Craighero R, Roner P, Merenda R, Amodio P, Sticca A, Caregaro L, Maffei-Faccioli A, Gatta A. Reversal of type 1 hepatorenal syndrome with the administration of midodrine and octreotide. Hepatology 1999 Jun;29(6):1690-7. PMID 10347109
- ↑ Esrailian E, Pantangco ER, Kyulo NL, Hu KQ, Runyon BA (2007). "Octreotide/Midodrine therapy significantly improves renal function and 30-day survival in patients with type 1 hepatorenal syndrome". Dig. Dis. Sci. 52 (3): 742–8. doi:10.1007/s10620-006-9312-0. PMID 17235705.
- ↑ Gulberg V, Bilzer M, Gerbes AL. Long-term therapy and retreatment of hepatorenal syndrome type 1 with ornipressin and dopamine. Hepatology 1999 Oct;30(4):870-5. PMID 10498636
- ↑ Wong F, Pantea L, Sniderman K. Midodrine, octreotide, albumin, and TIPS in selected patients with cirrhosis and type 1 hepatorenal syndrome. Hepatology. 2004 Jul;40(1):55-64. PMID 15239086.
- ↑ Guevara M, Rodes J. Hepatorenal syndrome. Int J Biochem Cell Biol. 2005 Jan;37(1):22-6. PMID 15381144.
- ↑ Mitzner SR, Stange J, Klammt S, Risler T, Erley CM, Bader BD, Berger ED, Lauchart W, Peszynski P, Freytag J, Hickstein H, Loock J, Lohr JM, Liebe S, Emmrich J, Korten G, Schmidt R. Improvement of hepatorenal syndrome with extracorporeal albumin dialysis MARS: results of a prospective, randomized, controlled clinical trial. Liver Transpl. 2000 May;6(3):277-86. PMID 10827226.
- ↑ Witzke O, Baumann M, Patschan D, Patschan S, Mitchell A, Treichel U, Gerken G, Philipp T, Kribben A. Which patients benefit from hemodialysis therapy in hepatorenal syndrome? J Gastroenterol Hepatol. 2004 Dec;19(12):1369-73. PMID 15610310
- ↑ Holt S, Goodier D, Marley R, Patch D, Burroughs A, Fernando B, Harry D, Moore K. Improvement in renal function in hepatorenal syndrome with N-acetylcysteine. Lancet. 1999 Jan 23;353(9149):294-5. PMID 9929029
- ↑ Clewell JD, Walker-Renard P. Prostaglandins for the treatment of hepatorenal syndrome. Ann Pharmacother. 1994 Jan;28(1):54-5. PMID 8123962
- ↑ Rutherford A, King LY, Hynan LS, Vedvyas C, Lin W, Lee WM; et al. (2012). "Development of an accurate index for predicting outcomes of patients with acute liver failure". Gastroenterology. 143 (5): 1237–43. doi:10.1053/j.gastro.2012.07.113. PMC 3480539. PMID 22885329.