Valsartan
{{DrugProjectFormSinglePage |authorTag=Sheng Shi, M.D. [1], Rabin Bista, M.B.B.S. [2] |genericName=Valsartan |aOrAn=an |drugClass=Angiotensin 2 Receptor Blocker |indicationType=treatment |indication=hypertension, heart failure |hasBlackBoxWarning=Yes |adverseReactions=hypotension, dizziness, headache, raised serum blood urea nitrogen , raised serum creatinine , cough |blackBoxWarningTitle=WARNING: FETAL TOXICITY |blackBoxWarningBody=
- When pregnancy is detected, discontinue valsartan as soon as possible.
- Drugs that act directly on the renin-angiotensin system can cause injury and death to the developing fetus.
|fdaLIADAdult=
Hypertension
- Dosing information
- Recommended starting dosage: 80 mg or 160 mg PO qd when used as monotherapy in patients who are not volume-depleted. Patients requiring greater reductions may be started at the higher dose.
- Dosage range: 80 mg to 320 mg PO qd
- The antihypertensive effect is substantially present within 2 weeks and maximal reduction is generally attained after 4 weeks.
- Maximum: 320 mg
- Addition of a diuretic has a greater effect than dose increases beyond ‘’‘80 mg’‘’.
- No initial dosage adjustment is required for elderly patients, for patients with mild or moderate renal impairment, or for patients with mild or moderate liver insufficiency. Care should be exercised with dosing of valsartan tablets in patients with hepatic or severe renal impairment.
- Valsartan tablets may be administered with other antihypertensive agents.
- Valsartan tablets may be administered with or without food.
Heart Failure
- Dosing information
- Recommended starting dose 40 mg PO bid.
- Uptitration to 80 mg -160 mg PO bid should be done to the highest dose, as tolerated by the patient.
- Consideration should be given to reducing the dose of concomitant diuretics.
- Maximum dosage: 320 mg/day
|offLabelAdultGuideSupport=There is limited information regarding Off-Label Guideline-Supported Use of Valsartan in adult patients.
|offLabelAdultNoGuideSupport=
Prophylaxis of Diabetes mellitus type 2
- Dosing information
- Initial dosage: 80 mg PO qd with an increase to 160 mg PO qd after 2 weeks.[1]
Diabetic nephropathy
- Dosing information
Erectile dysfunction
- Dosing information
- 80- 160 mg PO qd[4]
Impaired cognition
- Dosing information
- 160 PO qd[5]
Left ventricular hypertrophy
- Dosing information
Prophylaxis of Newly detected atrial fibrillation
- Dosing information
- 80 mg/day[8]
Prophylaxis of Restenotic lesion of coronary artery
- Dosing information
- 80 mg/day[9]
|fdaLIADPed=
Hypertension
- Dosing information
- For children who can swallow tablets
- Recommended starting dosage: ’‘’1.3 mg/kg PO qd (up to 40 mg total)‘’‘.
- The dosage should be adjusted according to blood pressure response.
- Doses higher than 2.7 mg/kg (up to 160 mg) once daily have not been studied in pediatric patients 6 to 16 years old.
- For children who can't swallow tablets,or children for whom the calculated dosage (mg/kg) does not correspond to the available tablet strengths of valsartan, the use of a suspension is recommended.
- The dosage should be adjusted according to blood pressure response.
- Doses higher than 2.7 mg/kg (up to 160 mg) once daily have not been studied in pediatric patients 6 to 16 years old.
|offLabelPedGuideSupport=There is limited information regarding Off-Label Guideline-Supported Use of Valsartan in pediatric patients. |offLabelPedNoGuideSupport=There is limited information regarding Off-Label Non–Guideline-Supported Use of Valsartan in pediatric patients. |contraindications=* Do not use in patients with known hypersensitivity to any component.
|warnings====Fetal Toxicity===
Pregnancy Category D
- Use of drugs that act on the renin-angiotensin system during the second and third trimesters of pregnancy reduces fetal renal function and increases fetal and neonatal morbidity and death. Resulting oligohydramnios can be associated with fetal lung hypoplasia and skeletal deformations. Potential neonatal adverse effects include skull hypoplasia, anuria, hypotension, renal failure, and death. When pregnancy is detected, discontinue Diovan as soon as possible.
Hypotension
- Excessive hypotension was rarely seen (0.1%) in patients with uncomplicated hypertension treated with Diovan alone. In patients with an activated renin-angiotensin system, such as volume- and/or salt-depleted patients receiving high doses of diuretics, symptomatic hypotension may occur. This condition should be corrected prior to administration of Diovan, or the treatment should start under close medical supervision.
- Caution should be observed when initiating therapy in patients with heart failure or post-myocardial infarction patients. Patients with heart failure or post-myocardial infarction patients given Diovan commonly have some reduction in blood pressure, but discontinuation of therapy because of continuing symptomatic hypotension usually is not necessary when dosing instructions are followed. In controlled trials in heart failure patients, the incidence of hypotension in valsartan-treated patients was 5.5% compared to 1.8% in placebo-treated patients. In the Valsartan in Acute Myocardial Infarction Trial (VALIANT), hypotension in post-myocardial infarction patients led to permanent discontinuation of therapy in 1.4% of valsartan-treated patients and 0.8% of captopril-treated patients.
- If excessive hypotension occurs, the patient should be placed in the supine position and, if necessary, given an intravenous infusion of normal saline. A transient hypotensive response is not a contraindication to further treatment, which usually can be continued without difficulty once the blood pressure has stabilized.
Impaired Renal Function
- Changes in renal function including acute renal failure can be caused by drugs that inhibit the renin-angiotensin system and by diuretics. Patients whose renal function may depend in part on the activity of the renin-angiotensin system (e.g. patients with renal artery stenosis, chronic kidney disease, severe congestive heart failure, or volume depletion) may be at particular risk of developing acute renal failure on Diovan. Monitor renal function periodically in these patients. Consider withholding or discontinuing therapy in patients who develop a clinically significant decrease in renal function on Diovan.
Hyperkalemia
- Some patients with heart failure have developed increases in potassium. These effects are usually minor and transient, and they are more likely to occur in patients with pre-existing renal impairment. Dosage reduction and/or discontinuation of Diovan may be required.
|clinicalTrials====Clinical Studies Experience===
- Because clinical studies are conducted under widely varying conditions, adverse reaction rates observed in the clinical studies of a drug cannot be directly compared to rates in the clinical studies of another drug and may not reflect the rates observed in practice.
Adult Hypertension
- Diovan (valsartan) has been evaluated for safety in more than 4,000 patients, including over 400 treated for over 6 months, and more than 160 for over 1 year. Adverse reactions have generally been mild and transient in nature and have only infrequently required discontinuation of therapy. The overall incidence of adverse reactions with Diovan was similar to placebo.
- The overall frequency of adverse reactions was neither dose-related nor related to gender, age, race, or regimen. Discontinuation of therapy due to side effects was required in 2.3% of valsartan patients and 2.0% of placebo patients. The most common reasons for discontinuation of therapy with Diovan were headache and dizziness.
- The adverse reactions that occurred in placebo-controlled clinical trials in at least 1% of patients treated with Diovan and at a higher incidence in valsartan (n=2,316) than placebo (n=888) patients included viral infection (3% vs. 2%), fatigue (2% vs. 1%), and abdominal pain (2% vs. 1%).
- Headache, dizziness, upper respiratory infection, cough, diarrhea, rhinitis, sinusitis, nausea, pharyngitis, edema, and arthralgia occurred at a more than 1% rate but at about the same incidence in placebo and valsartan patients.
- In trials in which valsartan was compared to an ACE inhibitor with or without placebo, the incidence of dry cough was significantly greater in the ACE-inhibitor group (7.9%) than in the groups who received valsartan (2.6%) or placebo (1.5%). In a 129-patient trial limited to patients who had had dry cough when they had previously received ACE inhibitors, the incidences of cough in patients who received valsartan, HCTZ, or lisinopril were 20%, 19%, and 69% respectively (p <0.001).
- Dose-related orthostatic effects were seen in less than 1% of patients. An increase in the incidence of dizziness was observed in patients treated with Diovan 320 mg (8%) compared to 10 to 160 mg (2% to 4%).
- Diovan has been used concomitantly with hydrochlorothiazide without evidence of clinically important adverse interactions.
- Other adverse reactions that occurred in controlled clinical trials of patients treated with Diovan (>0.2% of valsartan patients) are listed below. It cannot be determined whether these events were causally related to Diovan.
Body as a Whole: Allergic reaction and asthenia
Cardiovascular: Palpitations
Dermatologic: Pruritus and rash
Digestive: Constipation, dry mouth, dyspepsia, and flatulence
Musculoskeletal: Back pain, muscle cramps, and myalgia
Neurologic and Psychiatric: Anxiety, insomnia, paresthesia, and somnolence
Respiratory: Dyspnea
Special Senses: Vertigo
Urogenital: Impotence
- Other reported events seen less frequently in clinical trials included chest pain, syncope, anorexia, vomiting, and angioedema.
Pediatric Hypertension
- Diovan has been evaluated for safety in over 400 pediatric patients aged 6 to 17 years and more than 160 pediatric patients aged 6 months to 5 years. No relevant differences were identified between the adverse experience profile for pediatric patients aged 6-16 years and that previously reported for adult patients. Headache and hyperkalemia were the most common adverse events suspected to be study drug-related in older children (6 to 17 years old) and younger children (6 months to 5 years old), respectively. Hyperkalemia was mainly observed in children with underlying renal disease. Neurocognitive and developmental assessment of pediatric patients aged 6 to 16 years revealed no overall clinically relevant adverse impact after treatment with Diovan for up to 1 year.
- Diovan is not recommended for pediatric patients under 6 years of age. In a study (n=90) of pediatric patients (1-5 years), two deaths and three cases of on-treatment transaminase elevations were seen in the one-year open-label extension phase. These 5 events occurred in a study population in which patients frequently had significant co-morbidities. A causal relationship to Diovan has not been established. In a second study in which 75 children aged 1 to 6 years were randomized, no deaths and one case of marked liver transaminase elevations occurred during a 1 year open-label extension.
Heart Failure
- The adverse experience profile of Diovan in heart failure patients was consistent with the pharmacology of the drug and the health status of the patients. In the Valsartan Heart Failure Trial, comparing valsartan in total daily doses up to 320 mg (n=2,506) to placebo (n=2,494), 10% of valsartan patients discontinued for adverse reactions vs. 7% of placebo patients.
- The table shows adverse reactions in double-blind short-term heart failure trials, including the first 4 months of the Valsartan Heart Failure Trial, with an incidence of at least 2% that were more frequent in valsartan-treated patients than in placebo-treated patients. All patients received standard drug therapy for heart failure, frequently as multiple medications, which could include diuretics, digitalis, beta-blockers. About 93% of patients received concomitant ACE inhibitors.

- Discontinuations occurred in 0.5% of valsartan-treated patients and 0.1% of placebo patients for each of the following: elevations in creatinine and elevations in potassium.
- Other adverse reactions with an incidence greater than 1% and greater than placebo included headache NOS, nausea, renal impairment NOS, syncope, blurred vision, upper abdominal pain and vertigo. (NOS = not otherwise specified).
- From the long-term data in the Valsartan Heart Failure Trial, there did not appear to be any significant adverse reactions not previously identified.
Post-Myocardial Infarction
- The safety profile of Diovan was consistent with the pharmacology of the drug and the background diseases, cardiovascular risk factors, and clinical course of patients treated in the post-myocardial infarction setting. The table shows the percent of patients discontinued in the valsartan and captopril-treated groups in the Valsartan in Acute Myocardial Infarction Trial (VALIANT) with a rate of at least 0.5% in either of the treatment groups.
- Discontinuations due to renal dysfunction occurred in 1.1% of valsartan-treated patients and 0.8% of captopril-treated patients.

|postmarketing=* The following additional adverse reactions have been reported in post-marketing experience:
- Hypersensitivity: There are rare reports of angioedema. Some of these patients previously experienced angioedemawith other drugs including ACE inhibitors. Diovan should not be re-administered to patients who have had angioedema.
Digestive: Elevated liver enzymes and very rare reports of hepatitis
Renal: Impaired renal function, renal failure
Clinical Laboratory Tests: Hyperkalemia
Dermatologic: Alopecia
Blood and Lymphatic: There are very rare reports of thrombocytopenia
Vascular: Vasculitis
- Rare cases of rhabdomyolysis have been reported in patients receiving angiotensin II receptor blockers.
- Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
|drugInteractions=* No clinically significant pharmacokinetic interactions were observed when Diovan (valsartan) was coadministered with amlodipine, atenolol, cimetidine, digoxin, furosemide, glyburide, hydrochlorothiazide, or indomethacin. The valsartan-atenolol combination was more antihypertensive than either component, but it did not lower the heart rate more than atenolol alone.
- Coadministration of valsartan and warfarin did not change the pharmacokinetics of valsartan or the time-course of the anticoagulant properties of warfarin.
CYP 450 Interactions: In vitro metabolism studies indicate that CYP 450 mediated drug interactions between valsartan and coadministered drugs are unlikely because of the low extent of metabolism.
Transporters: The results from an in vitro study with human liver tissue indicate that valsartan is a substrate of the hepatic uptake transporter OATP1B1 and the hepatic efflux transporter MRP2. Coadministration of inhibitors of the uptake transporter (rifampin,cyclosporine) or efflux transporter (ritonavir) may increase the systemic exposure to valsartan.
Potassium: Concomitant use of valsartan with other agents that block the renin-angiotensin system, potassium sparing diuretics(e.g. spironolactone, triamterene, amiloride), potassium supplements, or salt substitutes containing potassium may lead to increases in serum potassium and in heart failure patients to increases in serum creatinine. If co-medication is considered necessary, monitoring of serum potassium is advisable.
- Non-Steroidal Anti-Inflammatory Agents including Selective Cyclooxygenase-2 Inhibitors (COX-2 Inhibitors): In patients who are elderly, volume-depleted (including those on diuretic therapy), or with compromised renal function, coadministration of NSAIDs, including selective COX-2 inhibitors, with angiotensin II receptor antagonists, including valsartan, may result in deterioration of renal function, including possible acute renal failure. These effects are usually reversible. Monitor renal function periodically in patients receiving valsartan and NSAID therapy.
- The antihypertensive effect of angiotensin II receptor antagonists, including valsartan may be attenuated by NSAIDs including selective COX-2 inhibitors.
- Dual Blockade of the renin-angiotensin system (RAS): Dual blockade of the RAS with angiotensin receptor blockers, ACE inhibitors, or aliskiren is associated with increased risks of hypotension, hyperkalemia, and changes in renal function (including acute renal failure) compared to monotherapy. Closely monitor blood pressure, renal function and electrolytes in patients on Diovan and other agents that affect the RAS.
- Do not coadminister aliskiren with Diovan in patients with diabetes. Avoid use of aliskiren with Diovan in patients with renal impairment (GFR <60 mL/min).
Clinical Laboratory Test Findings
- In controlled clinical trials, clinically important changes in standard laboratory parameters were rarely associated with administration of Diovan.
Creatinine: Minor elevations in creatinine occurred in 0.8% of patients taking Diovan and 0.6% given placebo in controlled clinical trials of hypertensive patients. In heart failure trials, greater than 50% increases in creatinine were observed in 3.9% of Diovan-treated patients compared to 0.9% of placebo-treated patients. In post-myocardial infarction patients, doubling of serum creatinine was observed in 4.2% of valsartan-treated patients and 3.4% of captopril-treated patients.
Hemoglobin and Hematocrit: Greater than 20% decreases in hemoglobin and hematocrit were observed in 0.4% and 0.8%, respectively, of Diovan patients, compared with 0.1% and 0.1% in placebo-treated patients. One valsartan patient discontinued treatment for microcytic anemia.
Liver Function Tests: Occasional elevations (greater than 150%) of liver chemistries occurred in Diovan-treated patients. Three patients (< 0.1%) treated with valsartan discontinued treatment for elevated liver chemistries.
Neutropenia: Neutropenia was observed in 1.9% of patients treated with Diovan and 0.8% of patients treated with placebo.
Serum Potassium: In hypertensive patients, greater than 20% increases in serum potassium were observed in 4.4% of Diovan-treated patients compared to 2.9% of placebo-treated patients. In heart failure patients, greater than 20% increases in serum potassium were observed in 10.0% of Diovan-treated patients compared to 5.1% of placebo-treated patients.
Blood Urea Nitrogen (BUN): In heart failure trials, greater than 50% increases in BUN were observed in 16.6% of Diovan-treated patients compared to 6.3% of placebo-treated patients. |FDAPregCat=D |useInPregnancyFDA=* Use of drugs that act on the renin-angiotensin system during the second and third trimesters of pregnancy reduces fetal renal function and increases fetal and neonatal morbidity and death. Resulting oligohydramnioscan be associated with fetal lung hypoplasia and skeletal deformations. Potential neonatal adverse effects include skull hypoplasia, anuria, hypotension, renal failure, and death. When pregnancy is detected, discontinue Diovan as soon as possible. These adverse outcomes are usually associated with use of these drugs in the second and third trimesters of pregnancy. Most epidemiologic studies examining fetal abnormalities after exposure to antihypertensive use in the first trimester have not distinguished drugs affecting the renin-angiotensin system from other antihypertensive agents. Appropriate management of maternal hypertension during pregnancy is important to optimize outcomes for both mother and fetus.
- In the unusual case that there is no appropriate alternative to therapy with drugs affecting the renin-angiotensin system for a particular patient, apprise the mother of the potential risk to the fetus. Perform serial ultrasound examinations to assess the intra-amniotic environment. If oligohydramniosis observed, discontinue Diovan, unless it is considered lifesaving for the mother. Fetal testing may be appropriate, based on the week of pregnancy. Patients and physicians should be aware, however, that oligohydramniosmay not appear until after the fetus has sustained irreversible injury. Closely observe infants with histories of in utero exposure to Diovan for hypotension, oliguria, and hyperkalemia.
|useInNursing=* It is not known whether Diovan is excreted in human milk. Diovan was excreted in the milk of lactating rats; however, animal breast milk drug levels may not accurately reflect human breast milk levels. Because many drugs are excreted into human milk and because of the potential for adverse reactions in nursing infants from Diovan, a decision should be made whether to discontinue nursing or discontinue the drug, taking into account the importance of the drug to the mother. |useInPed=* The antihypertensive effects of Diovan have been evaluated in two randomized, double-blind clinical studies in pediatric patients from 1-5 and 6-16 years of age. The pharmacokinetics of Diovan have been evaluated in pediatric patients 1 to 16 years of age. Diovan was generally well tolerated in children 6-16 years and the adverse experience profile was similar to that described for adults.
- In children and adolescents with hypertension where underlying renal abnormalities may be more common, renal function and serum potassium should be closely monitored as clinically indicated.
- Diovan is not recommended for pediatric patients under 6 years of age due to safety findings for which a relationship to treatment could not be excluded.
- No data are available in pediatric patients either undergoing dialysis or with a glomerular filtration rate <30 mL/min/1.73 m2.
- There is limited clinical experience with Diovan in pediatric patients with mild to moderate hepatic impairment.
- Daily oral dosing of neonatal/juvenile rats with valsartan at doses as low as 1 mg/kg/day (about 10% of the maximum recommended pediatric dose on a mg/m2 basis) from postnatal day 7 to postnatal day 70 produced persistent, irreversible kidney damage. These kidney effects in neonatal rats represent expected exaggerated pharmacological effects that are observed if rats are treated during the first 13 days of life. Since this period coincides with up to 44 weeks after conception in humans, it is not considered to point toward an increased safety concern in 6 to 16 year old children.
Neonates with a history of in utero exposure to Diovan:
- If oliguria or hypotension occurs, direct attention toward support of blood pressure and renal perfusion. Exchange transfusions or dialysis may be required as a means of reversing hypotension and/or substituting for disordered renal function.
|useInGeri=* In the controlled clinical trials of valsartan, 1,214 (36.2%) hypertensive patients treated with valsartan were ≥65 years and 265 (7.9%) were ≥75 years. No overall difference in the efficacy or safety of valsartan was observed in this patient population, but greater sensitivity of some older individuals cannot be ruled out.
- Of the 2,511 patients with heart failure randomized to valsartan in the Valsartan Heart Failure Trial, 45% (1,141) were 65 years of age or older. In the Valsartan in Acute Myocardial Infarction Trial (VALIANT), 53% (2,596) of the 4,909 patients treated with valsartan and 51% (2,515) of the 4,885 patients treated with valsartan + captopril were 65 years of age or older. There were no notable differences in efficacy or safety between older and younger patients in either trial.
|useInRenalImpair=* Safety and effectiveness of Diovan in patients with severe renal impairment (CrCl ≤ 30 mL/min) have not been established. No dose adjustment is required in patients with mild (CrCl 60-90 mL/min) or moderate (CrCl 30-60) renal impairment. |useInHepaticImpair=* No dose adjustment is necessary for patients with mild-to-moderate liver disease. No dosing recommendations can be provided for patients with severe liver disease. |administration=* Oral |IVCompat=Oral |overdose=FDA Package Insert for Valsartan contains no information regarding drug overdose. |drugBox={{Drugbox2 | verifiedrevid = 470628341 | IUPAC_name = (S)-3-methyl-2-(N-{[2'-(2H-1,2,3,4-tetrazol-5-yl)biphenyl-4-yl]methyl}pentanamido)butanoic acid | image = Valsartan.png | image2 = Valsartan ball-and-stick.png
| tradename = Diovan | Drugs.com = Monograph | MedlinePlus = a697015 | licence_US = Valsartan | pregnancy_US = D | pregnancy_category = | legal_US = Rx-only | legal_status = | routes_of_administration = oral
| bioavailability = 25% | protein_bound = 95% | metabolism = | elimination_half-life = 6 hours | excretion = Renal 30%, biliary 70%
| CASNo_Ref =
| CAS_number_Ref =
| CAS_number = 137862-53-4
| ATC_prefix = C09
| ATC_suffix = CA03
| ATC_supplemental =
| PubChem = 60846
| IUPHAR_ligand =3937
| DrugBank_Ref =
| DrugBank = DB00177
| ChemSpiderID_Ref =
| ChemSpiderID = 54833
| UNII_Ref =
| UNII = 80M03YXJ7I
| KEGG_Ref =
| KEGG = D00400
| ChEBI_Ref =
| ChEBI = 9927
| ChEMBL_Ref =
| ChEMBL = 1069
| C=24 | H=29 | N=5 | O=3
| molecular_weight = 435.519 g/mol
| smiles = O=C(O)[C@@H](N(C(=O)CCCC)Cc3ccc(c1ccccc1c2nnnn2)cc3)C(C)C
| InChI = 1/C24H29N5O3/c1-4-5-10-21(30)29(22(16(2)3)24(31)32)15-17-11-13-18(14-12-17)19-8-6-7-9-20(19)23-25-27-28-26-23/h6-9,11-14,16,22H,4-5,10,15H2,1-3H3,(H,31,32)(H,25,26,27,28)/t22-/m0/s1
| InChIKey = ACWBQPMHZXGDFX-QFIPXVFZBU
| StdInChI_Ref =
| StdInChI = 1S/C24H29N5O3/c1-4-5-10-21(30)29(22(16(2)3)24(31)32)15-17-11-13-18(14-12-17)19-8-6-7-9-20(19)23-25-27-28-26-23/h6-9,11-14,16,22H,4-5,10,15H2,1-3H3,(H,31,32)(H,25,26,27,28)/t22-/m0/s1
| StdInChIKey_Ref =
| StdInChIKey = ACWBQPMHZXGDFX-QFIPXVFZSA-N
}}
|mechAction=* Angiotensin II is formed from angiotensin I in a reaction catalyzed by angiotensin-converting enzyme (ACE, kininase II). angiotensinII is the principal pressor agent of the renin-angiotensin system, with effects that include vasoconstriction, stimulation of synthesis and release of aldosterone, cardiac stimulation, and renal reabsorption of sodium. Diovan (valsartan) blocks the vasoconstrictor and aldosterone-secreting effects of angiotensinII by selectively blocking the binding of angiotensin II to the AT1 receptor in many tissues, such as vascular smooth muscle and the adrenal gland. Its action is therefore independent of the pathways for angiotensin II synthesis.
- There is also an AT2 receptor found in many tissues, but AT2 is not known to be associated with cardiovascular homeostasis. Valsartan has much greater affinity (about 20,000-fold) for the AT1 receptor than for the AT2 receptor. The increased plasma levels of angiotensin II following AT1 receptor blockade with valsartan may stimulate the unblocked AT2 receptor. The primary metabolite of valsartan is essentially inactive with an affinity for the AT1 receptor about one-200th that of valsartan itself.
- Blockade of the renin-angiotensin system with ACE inhibitors, which inhibit the biosynthesis of angiotensin II from angiotensin I, is widely used in the treatment of hypertension. ACE inhibitors also inhibit the degradation of bradykinin, a reaction also catalyzed by ACE. Because valsartan does not inhibit ACE (kininase II), it does not affect the response to bradykinin. Whether this difference has clinical relevance is not yet known. Valsartan does not bind to or block other hormone receptors or ion channels known to be important in cardiovascular regulation.
- Blockade of the angiotensin II receptor inhibits the negative regulatory feedback of angiotensinII on renin secretion, but the resulting increased plasma renin activity and angiotensin II circulating levels do not overcome the effect of valsartan on blood pressure.
|structure=* iovan (valsartan) is a nonpeptide, orally active, and specific angiotensin II receptor blocker acting on the AT1 receptor subtype.
- Valsartan is chemically described as N-(1-oxopentyl)-N-[ [2′-(1H-tetrazol-5-yl) [1,1′-biphenyl]-4-yl]methyl]-L-valine. Its empirical formula is C24H29N5O3, its molecular weight is 435.5, and its structural formula is


- Valsartan is a white to practically white fine powder. It is soluble in ethanol and methanol and slightly soluble in water.
|PD=Valsartan inhibits the pressor effect of angiotensin II infusions. An oral dose of 80 mg inhibits the pressor effect by about 80% at peak with approximately 30% inhibition persisting for 24 hours. No information on the effect of larger doses is available.
- Removal of the negative feedback of angiotensin II causes a 2- to 3-fold rise in plasma renin and consequent rise in angiotensin II plasma concentration in hypertensive patients. Minimal decreases in plasma aldosterone were observed after administration of valsartan; very little effect on serum potassium was observed.
- In multiple-dose studies in hypertensive patients with stable renal insufficiency and patients with renovascular hypertension, valsartan had no clinically significant effects on glomerular filtration rate, filtration fraction, creatinine clearance, or renal plasma flow.
- In multiple-dose studies in hypertensive patients, valsartan had no notable effects on total cholesterol, fasting triglycerides, fasting serum glucose, or uric acid.
|PK=* Valsartan peak plasma concentration is reached 2 to 4 hours after dosing. Valsartan shows bi-exponential decay kinetics following intravenous administration, with an average elimination half-life of about 6 hours. Absolute bioavailability for Diovan is about 25% (range 10%-35%). The bioavailability of the suspension is 1.6 times greater than with the tablet. With the tablet, food decreases the exposure (as measured by AUC) to valsartan by about 40% and peak plasma concentration (Cmax) by about 50%. AUC and Cmax values of valsartan increase approximately linearly with increasing dose over the clinical dosing range. Valsartan does not accumulate appreciably in plasma following repeated administration.
Metabolism and Elimination: Valsartan, when administered as an oral solution, is primarily recovered in feces (about 83% of dose) and urine (about 13% of dose). The recovery is mainly as unchanged drug, with only about 20% of dose recovered as metabolites. The primary metabolite, accounting for about 9% of dose, is valeryl 4-hydroxy valsartan. In vitro metabolism studies involving recombinant CYP 450 enzymes indicated that the CYP 2C9 isoenzyme is responsible for the formation of valeryl-4-hydroxy valsartan. Valsartan does not inhibit CYP 450 isozymes at clinically relevant concentrations. CYP 450 mediated drug interaction between valsartan and coadministered drugs are unlikely because of the low extent of metabolism.
- Following intravenous administration, plasma clearance of valsartan is about 2 L/h and its renal clearance is 0.62 L/h (about 30% of total clearance).
Distribution: The steady state volume of distribution of valsartan after intravenous administration is small (17 L), indicating that valsartan does not distribute into tissues extensively. Valsartan is highly bound to serum proteins (95%), mainly serum albumin.
Special Populations
Pediatric: In a study of pediatric hypertensive patients (n=26, 1-16 years of age) given single doses of a suspension of Diovan (mean: 0.9 to 2 mg/kg), the clearance (L/h/kg) of valsartan for children was similar to that of adults receiving the same formulation.
Geriatric: Exposure (measured by AUC) to valsartan is higher by 70% and the half-life is longer by 35% in the elderly than in the young. No dosage adjustment is necessary.
Gender: Pharmacokinetics of valsartan does not differ significantly between males and females.
Heart Failure: The average time to peak concentration and elimination half-life of valsartan in heart failure patients are similar to those observed in healthy volunteers. AUC and Cmax values of valsartan increase linearly and are almost proportional with increasing dose over the clinical dosing range (40 to 160 mg twice a day). The average accumulation factor is about 1.7. The apparent clearance of valsartan following oral administration is approximately 4.5 L/h. Age does not affect the apparent clearance in heart failure patients.
Renal Insufficiency: There is no apparent correlation between renal function (measured by creatinine clearance) and exposure (measured by AUC) to valsartan in patients with different degrees of renal impairment. Consequently, dose adjustment is not required in patients with mild-to-moderate renal dysfunction. No studies have been performed in patients with severe impairment of renal function (creatinine clearance <10 mL/min). Valsartan is not removed from the plasma by hemodialysis. In the case of severe renal disease, exercise care with dosing of valsartan.
Hepatic Insufficiency: On average, patients with mild-to-moderate chronic liver disease have twice the exposure (measured by AUC values) to valsartan of healthy volunteers (matched by age, sex and weight). In general, no dosage adjustment is needed in patients with mild-to-moderate liver disease. Care should be exercised in patients with liver disease |nonClinToxic====Carcinogenesis, Mutagenesis, Impairment of Fertility===
- There was no evidence of carcinogenicity when valsartan was administered in the diet to mice and rats for up to 2 years at doses up to 160 and 200 mg/kg/day, respectively. These doses in mice and rats are about 2.6 and 6 times, respectively, the maximum recommended human dose on a mg/m2 basis. (Calculations assume an oral dose of 320 mg/day and a 60-kg patient.)
- Mutagenicity assays did not reveal any valsartan-related effects at either the gene or chromosome level. These assays included bacterial mutagenicity tests with Salmonella (Ames) and E coli; a gene mutation test with Chinese hamster V79 cells; a cytogenetic test with Chinese hamster ovary cells; and a rat micronucleus test.
- Valsartan had no adverse effects on the reproductive performance of male or female rats at oral doses up to 200 mg/kg/day. This dose is 6 times the maximum recommended human dose on a mg/m2 basis. (Calculations assume an oral dose of 320 mg/day and a 60-kg patient.)
Animal Toxicology and/or Pharmacology
Reproductive Toxicology Studies
- No teratogenic effects were observed when valsartan was administered to pregnant mice and rats at oral doses up to 600 mg/kg/day and to pregnant rabbits at oral doses up to 10 mg/kg/day. However, significant decreases in fetal weight, pup birth weight, pup survival rate, and slight delays in developmental milestones were observed in studies in which parental rats were treated with valsartan at oral, maternally toxic (reduction in body weight gain and food consumption) doses of 600 mg/kg/day during organogenesis or late gestation and lactation. In rabbits, fetotoxicity (i.e., resorptions, litter loss, abortions, and low body weight) associated with maternal toxicity (mortality) was observed at doses of 5 and 10 mg/kg/day. The no observed adverse effect doses of 600, 200 and 2 mg/kg/day in mice, rats and rabbits represent 9, 6, and 0.1 times, respectively, the maximum recommended human dose on a mg/m2 basis. Calculations assume an oral dose of 320 mg/day and a 60-kg patient.
|clinicalStudies====Hypertension===
Adult Hypertension
- The antihypertensive effects of Diovan (valsartan) were demonstrated principally in 7 placebo-controlled, 4- to 12-week trials (one in patients over 65) of dosages from 10 to 320 mg/day in patients with baseline diastolic blood pressures of 95-115. The studies allowed comparison of once-daily and twice-daily regimens of 160 mg/day; comparison of peak and trough effects; comparison (in pooled data) of response by gender, age, and race; and evaluation of incremental effects of hydrochlorothiazide.
- Administration of valsartan to patients with essential hypertension results in a significant reduction of sitting, supine, and standing systolic and diastolic blood pressure, usually with little or no orthostatic change.
- In most patients, after administration of a single oral dose, onset of antihypertensive activity occurs at approximately 2 hours, and maximum reduction of blood pressure is achieved within 6 hours. The antihypertensive effect persists for 24 hours after dosing, but there is a decrease from peak effect at lower doses (40 mg) presumably reflecting loss of inhibition of angiotensin II. At higher doses, however (160 mg), there is little difference in peak and trough effect. During repeated dosing, the reduction in blood pressure with any dose is substantially present within 2 weeks, and maximal reduction is generally attained after 4 weeks. In long-term follow-up studies (without placebo control), the effect of valsartan appeared to be maintained for up to 2 years. The antihypertensive effect is independent of age, gender or race. The latter finding regarding race is based on pooled data and should be viewed with caution, because antihypertensive drugs that affect the renin-angiotensin system (that is, ACE inhibitors and angiotensin-II blockers) have generally been found to be less effective in low-renin hypertensives (frequently blacks) than in high-renin hypertensives (frequently whites). In pooled, randomized, controlled trials of Diovan that included a total of 140 blacks and 830 whites, valsartan and an ACE-inhibitor control were generally at least as effective in blacks as whites. The explanation for this difference from previous findings is unclear.
- Abrupt withdrawal of valsartan has not been associated with a rapid increase in blood pressure.
- The blood pressure lowering effect of valsartan and thiazide-type diuretics are approximately additive.
- The 7 studies of valsartan monotherapy included over 2,000 patients randomized to various doses of valsartan and about 800 patients randomized to placebo. Doses below 80 mg were not consistently distinguished from those of placebo at trough, but doses of 80, 160 and 320 mg produced dose-related decreases in systolic and diastolic blood pressure, with the difference from placebo of approximately 6-9/3-5 mmHg at 80-160 mg and 9/6 mmHg at 320 mg. In a controlled trial the addition of HCTZ to valsartan 80 mg resulted in additional lowering of systolic and diastolic blood pressure by approximately 6/3 and 12/5 mmHg for 12.5 and 25 mg of HCTZ, respectively, compared to valsartan 80 mg alone.
- Patients with an inadequate response to 80 mg once daily were titrated to either 160 mg once daily or 80 mg twice daily, which resulted in a comparable response in both groups.
- In controlled trials, the antihypertensive effect of once-daily valsartan 80 mg was similar to that of once-daily enalapril 20 mg or once-daily lisinopril 10 mg.
- There are no trials of Diovan demonstrating reductions in cardiovascular risk in patients with hypertension, but at least one pharmacologically similar drug has demonstrated such benefits.
- There was essentially no change in heart rate in valsartan-treated patients in controlled trials.
Pediatric Hypertension
- The antihypertensive effects of Diovan were evaluated in two randomized, double-blind clinical studies.
- In a clinical study involving 261 hypertensive pediatric patients 6 to 16 years of age, patients who weighed < 35 kg received 10, 40 or 80 mg of valsartan daily (low, medium and high doses), and patients who weighed ≥ 35 kg received 20, 80, and 160 mg of valsartan daily (low, medium and high doses). Renal and urinary disorders, and essential hypertension with or without obesity were the most common underlying causes of hypertension in children enrolled in this study. At the end of 2 weeks, valsartan reduced both systolic and diastolic blood pressure in a dose-dependent manner. Overall, the three dose levels of valsartan (low, medium and high) significantly reduced systolic blood pressure by -8, -10, -12 mm Hg from the baseline, respectively. Patients were re-randomized to either continue receiving the same dose of valsartan or were switched to placebo. In patients who continued to receive the medium and high doses of valsartan, systolic blood pressure at trough was -4 and -7 mm Hg lower than patients who received the placebo treatment. In patients receiving the low dose of valsartan, systolic blood pressure at trough was similar to that of patients who received the placebo treatment. Overall, the dose-dependent antihypertensive effect of valsartan was consistent across all the demographic subgroups.
- In a clinical study involving 90 hypertensive pediatric patients 1 to 5 years of age with a similar study design, there was some evidence of effectiveness, but safety findings for which a relationship to treatment could not be excluded mitigate against recommending use in this age group.
Heart Failure
- The Valsartan Heart Failure Trial (Val-HeFT) was a multinational, double-blind study in which 5,010 patients with NYHA class II (62%) to IV (2%) heart failure and LVEF <40%, on baseline therapy chosen by their physicians, were randomized to placebo or valsartan (titrated from 40 mg twice daily to the highest tolerated dose or 160 mg twice daily) and followed for a mean of about 2 years. Although Val-HeFT’s primary goal was to examine the effect of valsartan when added to an ACE inhibitor, about 7% were not receiving an ACE inhibitor. Other background therapy included diuretics (86%), digoxin (67%), and beta-blockers (36%). The population studied was 80% male, 46% 65 years or older and 89% Caucasian. At the end of the trial, patients in the valsartan group had a blood pressure that was 4 mmHg systolic and 2 mmHg diastolic lower than the placebo group. There were two primary end points, both assessed as time to first event: all-cause mortality and heart failure morbidity, the latter defined as all-cause mortality, sudden death with resuscitation, hospitalization for heart failure, and the need for intravenous inotropic or vasodilatory drugs for at least 4 hours. These results are summarized in the table below.
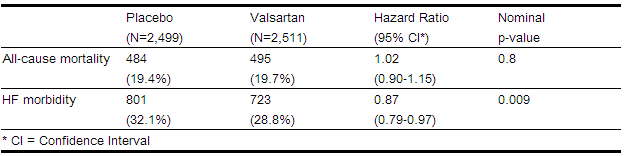
- Although the overall morbidity result favored valsartan, this result was largely driven by the 7% of patients not receiving an ACE inhibitor, as shown in the following table.

- The modest favorable trend in the group receiving an ACE inhibitor was largely driven by the patients receiving less than the recommended dose of ACE inhibitor. Thus, there is little evidence of further clinical benefit when valsartan is added to an adequate dose of ACE inhibitor.
- Secondary end points in the subgroup not receiving ACE inhibitors were as follows.

- In patients not receiving an ACE inhibitor, valsartan-treated patients had an increase in ejection fraction and reduction in left ventricular internal diastolic diameter (LVIDD).
- Effects were generally consistent across subgroups defined by age and gender for the population of patients not receiving an ACE inhibitor. The number of black patients was small and does not permit a meaningful assessment in this subset of patients.
Post-Myocardial Infarction
- The VALsartan In Acute myocardial infarction trial (VALIANT) was a randomized, controlled, multinational, double-blind study in 14,703 patients with acute myocardial infarction and either heart failure (signs, symptoms or radiological evidence) or left ventricular systolic dysfunction (ejection fraction ≤40% by radionuclide ventriculography or ≤35% by echocardiography or ventricular contrast angiography). Patients were randomized within 12 hours to 10 days after the onset of myocardial infarction symptoms to one of three treatment groups: valsartan (titrated from 20 or 40 mg twice daily to the highest tolerated dose up to a maximum of 160 mg twice daily), the ACE inhibitor, captopril (titrated from 6.25 mg three times daily to the highest tolerated dose up to a maximum of 50 mg three times daily), or the combination of valsartan plus captopril. In the combination group, the dose of valsartan was titrated from 20 mg twice daily to the highest tolerated dose up to a maximum of 80 mg twice daily; the dose of captopril was the same as for monotherapy. The population studied was 69% male, 94% Caucasian, and 53% were 65 years of age or older. Baseline therapy included aspirin (91%), beta-blockers (70%), ACE inhibitors (40%), thrombolytics (35%) and statins (34%). The mean treatment duration was 2 years. The mean daily dose of Diovan in the monotherapy group was 217 mg.

- The primary endpoint was time to all-cause mortality. Secondary endpoints included (1) time to cardiovascular (CV) mortality, and (2) time to the first event of cardiovascular mortality, reinfarction, or hospitalization for heart failure. The results are summarized in the table below:
- There was no difference in overall mortality among the three treatment groups. There was thus no evidence that combining the ACE inhibitor captopril and the angiotensin II blocker valsartan was of value.
- The data were assessed to see whether the effectiveness of valsartan could be demonstrated by showing in a non-inferiority analysis that it preserved a fraction of the effect of captopril, a drug with a demonstrated survival effect in this setting. A conservative estimate of the effect of captopril(based on a pooled analysis of 3 post-infarction studies of captopril and 2 other ACE inhibitors) was a 14-16% reduction in mortality compared to placebo. Valsartan would be considered effective if it preserved a meaningful fraction of that effect and unequivocally preserved some of that effect. As shown in the table, the upper bound of the CI for the hazard ratio (valsartan/captopril) for overall or CV mortality is 1.09-1.11, a difference of about 9-11%, thus making it unlikely that valsartan has less than about half of the estimated effect of captopril and clearly demonstrating an effect of valsartan. The other secondary endpoints were consistent with this conclusion.

|howSupplied=* Valsartan is available as tablets containing valsartan, USP 40 mg, 80 mg, 160 mg, or 320 mg. All strengths are packaged in bottles as described below. 40 mg tablets are yellow colored, film-coated, oval-shaped tablets debossed with ' RX121' on one side and break line on the other side NDC 51660-140-03 Bottles of 10 NDC 51660-140-30 Bottles of 30 NDC 51660-140-05 Bottles of 500 80 mg tablets are yellowish brown colored, film-coated, oval-shaped tablets debossed with ' RX124' on one side and plain on the other side NDC 51660-141-03 Bottles of 10 NDC 51660-141-90 Bottles of 90 NDC 51660-141-05 Bottles of 500 160 mg tablets are pink colored, film-coated, oval-shaped tablets debossed with ' RX125' on one side and plain on the other side NDC 51660-142-03 Bottles of 10 NDC 51660-142-90 Bottles of 90 NDC 51660-142-05 Bottles of 500 320 mg tablets are brown colored, film-coated, oval-shaped tablets debossed with ' RX126' on one side and plain on the other side NDC 51660-143-03 Bottles of 10 NDC 51660-143-90 Bottles of 90 NDC 51660-143-05 Bottles of 500 |storage=* Store at 20 o - 25o C (68o - 77o F) [See USP Controlled Room Temperature]. Protect from moisture. Dispense in tight container (USP). |fdaPatientInfo=Information for Patients Pregnancy: Female patients of childbearing age should be told about the consequences of exposure to valsartan during pregnancy. Discuss treatment options with women planning to become pregnant. Patients should be asked to report pregnancies to their physicians as soon as possible. Distributed by: Ohm Laboratories Inc. North Brunswick, NJ 08902 USA March 2014 FDA-02 |alcohol=Alcohol-Valsartan interaction has not been established. Talk to your doctor about the effects of taking alcohol with this medication. |brandNames=* Diovan |lookAlike=There is limited information about the Look-Alike Drug Names. }} {{#subobject:
|Page Name=Valsartan |Pill Name=No_image.jpg |Drug Name=valsartan tablet, film coated |Pill Ingred=SILICON DIOXIDE
CROSPOVIDONE HYPROMELLOSES MAGNESIUM STEARATE CELLULOSE, MICROCRYSTALLINE STARCH, CORN TALC TITANIUM DIOXIDE POLYETHYLENE GLYCOLS FERRIC OXIDE YELLOW|+sep=;
|Pill Imprint=RX121 |Pill Dosage=40 mg |Pill Color=Yellow|+sep=; |Pill Shape=Oval |Pill Size (mm)=9.00 |Pill Scoring=2 |Pill Image= |Drug Author=Ohm Laboratories Inc. |NDC=51660-140
}}
{{#subobject:
|Page Name=Valsartan |Pill Name=No_image.jpg |Drug Name=valsartan tablet, film coated |Pill Ingred=SILICON DIOXIDE
CROSPOVIDONE HYPROMELLOSES MAGNESIUM STEARATE CELLULOSE, MICROCRYSTALLINE STARCH, CORN TALC TITANIUM DIOXIDE POLYETHYLENE GLYCOLS FERRIC OXIDE YELLOW|+sep=;
|Pill Imprint=RX124 |Pill Dosage=80 mg |Pill Color=Yellow|+sep=; |Pill Shape=Oval |Pill Size (mm)=12.00 |Pill Scoring= |Pill Image= |Drug Author=Ohm Laboratories Inc. |NDC=51660-141
}}
{{#subobject:
|Page Name=Valsartan |Pill Name=No_image.jpg |Drug Name=valsartan tablet, film coated |Pill Ingred=SILICON DIOXIDE
CROSPOVIDONE HYPROMELLOSES MAGNESIUM STEARATE CELLULOSE, MICROCRYSTALLINE STARCH, CORN TALC TITANIUM DIOXIDE POLYETHYLENE GLYCOLS FERRIC OXIDE YELLOW|+sep=;
|Pill Imprint=RX125 |Pill Dosage=160 mg |Pill Color=Pink|+sep=; |Pill Shape=Oval |Pill Size (mm)=15.00 |Pill Scoring= |Pill Image= |Drug Author=Ohm Laboratories Inc. |NDC=51660-142
}}
{{#subobject:
|Page Name=Valsartan |Pill Name=No_image.jpg |Drug Name=valsartan tablet, film coated |Pill Ingred=SILICON DIOXIDE
CROSPOVIDONE HYPROMELLOSES MAGNESIUM STEARATE CELLULOSE, MICROCRYSTALLINE STARCH, CORN TALC TITANIUM DIOXIDE POLYETHYLENE GLYCOLS FERRIC OXIDE YELLOW|+sep=;
|Pill Imprint=RX126 |Pill Dosage=320 mg |Pill Color=Brown|+sep=; |Pill Shape=Oval |Pill Size (mm)=19.00 |Pill Scoring= |Pill Image= |Drug Author=Ohm Laboratories Inc. |NDC=51660-143
}}
{{#subobject:
|Label Page=Valsartan |Label Name=Valsartan_label_01.jpg
}}
{{#subobject:
|Label Page=Valsartan |Label Name=Valsartan_label_02.jpg
}}
{{#subobject:
|Label Page=Valsartan |Label Name=Valsartan_label_03.jpg
}}
{{#subobject:
|Label Page=Valsartan |Label Name=Valsartan_label_04.jpg
}}
{{#subobject:
|Label Page=Valsartan |Label Name=Valsartan_panel_01.png
}}
{{#subobject:
|Label Page=Valsartan |Label Name=Valsartan_label_02.png
}}
{{#subobject:
|Label Page=Valsartan |Label Name=Valsartan_label_03.png
}}
{{#subobject:
|Label Page=Valsartan |Label Name=Valsartan_panel_04.png
}}
- ↑ NAVIGATOR Study Group. McMurray JJ, Holman RR, Haffner SM, Bethel MA, Holzhauer B; et al. (2010). "Effect of valsartan on the incidence of diabetes and cardiovascular events". N Engl J Med. 362 (16): 1477–90. doi:10.1056/NEJMoa1001121. PMID 20228403. Review in: Ann Intern Med. 2010 Jun 15;152(12):JC6-9 Review in: Evid Based Med. 2011 Aug;16(4):122-3
- ↑ Suzuki K, Souda S, Ikarashi T, Kaneko S, Nakagawa O, Aizawa Y (2002). "Renoprotective effects of low-dose valsartan in type 2 diabetic patients with diabetic nephropathy". Diabetes Res Clin Pract. 57 (3): 179–83. PMID 12126767.
- ↑ Jacobsen P, Andersen S, Jensen BR, Parving HH (2003). "Additive effect of ACE inhibition and angiotensin II receptor blockade in type I diabetic patients with diabetic nephropathy". J Am Soc Nephrol. 14 (4): 992–9. PMID 12660333.
- ↑ Düsing R (2003). "Effect of the angiotensin II antagonist valsartan on sexual function in hypertensive men". Blood Press Suppl. 2: 29–34. PMID 14761074.
- ↑ Fogari R, Mugellini A, Zoppi A, Marasi G, Pasotti C, Poletti L; et al. (2004). "Effects of valsartan compared with enalapril on blood pressure and cognitive function in elderly patients with essential hypertension". Eur J Clin Pharmacol. 59 (12): 863–8. doi:10.1007/s00228-003-0717-9. PMID 14747881.
- ↑ Suzuki K, Kato K, Soda S, Kamimura T, Aizawa Y (2004). "The effect of valsartan on regression of left ventricular hypertrophy in type 2 diabetic patients". Diabetes Obes Metab. 6 (3): 195–9. doi:10.1111/j.1462-8902.2004.00331.x. PMID 15056127.
- ↑ Mutlu H, Ozhan H, Okçün B, Okuyan E, Yigit Z, Erbaş C; et al. (2002). "The efficacy of valsartan in essential hypertension and its effects on left ventricular hypertrophy". Blood Press. 11 (1): 53–5. PMID 11926352.
- ↑ Schmieder RE, Kjeldsen SE, Julius S, McInnes GT, Zanchetti A, Hua TA; et al. (2008). "Reduced incidence of new-onset atrial fibrillation with angiotensin II receptor blockade: the VALUE trial". J Hypertens. 26 (3): 403–11. doi:10.1097/HJH.0b013e3282f35c67. PMID 18300848.
- ↑ Peters S, Götting B, Trümmel M, Rust H, Brattström A (2001). "Valsartan for prevention of restenosis after stenting of type B2/C lesions: the VAL-PREST trial". J Invasive Cardiol. 13 (2): 93–7. PMID 11176015.