ACE inhibitor
|
WikiDoc Resources for ACE inhibitor |
|
Articles |
|---|
|
Most recent articles on ACE inhibitor Most cited articles on ACE inhibitor |
|
Media |
|
Powerpoint slides on ACE inhibitor |
|
Evidence Based Medicine |
|
Clinical Trials |
|
Ongoing Trials on ACE inhibitor at Clinical Trials.gov Trial results on ACE inhibitor Clinical Trials on ACE inhibitor at Google
|
|
Guidelines / Policies / Govt |
|
US National Guidelines Clearinghouse on ACE inhibitor NICE Guidance on ACE inhibitor
|
|
Books |
|
News |
|
Commentary |
|
Definitions |
|
Patient Resources / Community |
|
Patient resources on ACE inhibitor Discussion groups on ACE inhibitor Patient Handouts on ACE inhibitor Directions to Hospitals Treating ACE inhibitor Risk calculators and risk factors for ACE inhibitor
|
|
Healthcare Provider Resources |
|
Causes & Risk Factors for ACE inhibitor |
|
Continuing Medical Education (CME) |
|
International |
|
|
|
Business |
|
Experimental / Informatics |
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]
Overview
ACE inhibitors, or inhibitors of Angiotensin-Converting Enzyme, are a group of pharmaceuticals that are used primarily in treatment of hypertension and congestive heart failure, in most cases as the drugs of first choice.
Clinical use
Indications for ACE inhibitors include:
- Prevention of cardiovascular disorders
- Congestive heart failure (CHF)
- Hypertension
- Left ventricular dysfunction
- Prevention of nephropathy in diabetes mellitus
In several of these indications, ACE inhibitors are used first-line as several agents in the class have been clinically shown to be superior to other classes of drugs in the reduction of morbidity and mortality.
ACE inhibitors are often combined with diuretics in the control of hypertension (usually a thiazide), when an ACE inhibitor alone proves insufficient; and in chronic heart failure (usually furosemide) for improved symptomatic control. Thus there exists, on the market, combination products combining an ACE inhibitor with a thiazide (usually hydrochlorothiazide) in a single tablet to allow easy administration by patients.
The renin-angiotensin-aldosterone system (RAAS)
This system is activated in response to hypotension, decreased sodium concentration in the distal tubule, decreased blood volume and renal sympathetic nerve stimulation. In such a situation, the kidneys release renin which cleaves the liver derived angiotensinogen into Angiotensin I. Angiotensin I is then converted to angiotensin II via the angiotensin-converting-enzyme (ACE) in the pulmonary circulation. The system in general aims to increase blood pressure.
Effects
ACE inhibitors lower arteriolar resistance and increase venous capacitance; increase cardiac output and cardiac index, stroke work and volume, lower renovascular resistance, and lead to increased natriuresis (excretion of sodium in the urine).
Normally, angiotensin II will have the following effects:
- vasoconstriction (narrowing of blood vessels), which may lead to increased blood pressure and hypertension
- Ventricular remodeling of the heart, which may lead to ventricular hypertrophy and CHF
- stimulate the adrenal cortex to release aldosterone, a hormone that acts on kidney tubules to retain sodium and chloride ions and excrete potassium. Sodium is a "water-holding" molecule, so water is also retained, which leads to increased blood volume, hence an increase in blood pressure.
- stimulate the posterior pituitary into releasing vasopressin (also known as anti-diuretic hormone (ADH)) which also acts on the kidneys to increase water retention.
With ACE inhibitor use, the effects of angiotensin II are prevented, leading to decreased blood pressure.
Epidemiological and clinical studies have shown that ACE inhibitors reduce the progress of diabetic nephropathy independently from their blood pressure-lowering effect. This action of ACE inhibitors is utilised in the prevention of diabetic renal failure.
ACE inhibitors have been shown to be effective for indications other than hypertension even in patients with normal blood pressure. The use of a maximum dose of ACE inhibitors in such patients (including for prevention of diabetic nephropathy, congestive heart failure, prophylaxis of cardiovascular events) is justified because it improves clinical outcomes, independent of the blood pressure lowering effect of ACE inhibitors. Such therapy, of course, requires careful and gradual titration of the dose to prevent the patient suffering from the effects of rapidly decreasing their blood pressure (dizziness, fainting, etc).
Adverse effects
Common adverse drug reactions (≥1% of patients) include: hypotension, cough, hyperkalemia, headache, dizziness, fatigue, nausea, renal impairment.[1]
A persistent dry cough is a relatively common adverse effect believed to be associated with the increases in bradykinin levels produced by ACE inhibitors, although the role of bradykinin in producing these symptoms remains disputed by some authors.[2] Patients who experience this cough are often switched to angiotensin II receptor antagonists.
Rash and taste disturbances, infrequent with most ACE inhibitors, are more prevalent in captopril and is attributed to its sulfhydryl moiety. This has led to decreased use of captopril in clinical setting, although it is still used in scintigraphy of the kidney.
Renal impairment is a significant adverse effect of all ACE inhibitors, and is associated with their effect on angiotensin II-mediated homeostatic functions such as renal blood flow. Renal blood flow is affected by Angiotensin II because it vasoconstricts the efferent arterioles of the glomeruli of the kidney, thereby increasing glomerular filtration rate (GFR). Hence, by reducing angiotensin II levels, ACE inhibitors reduce GFR, a marker of renal function. Specifically, ACE inhibitors can induce or exacerbate renal impairment in patients with renal artery stenosis. This is especially a problem if the patient is also concomitantly taking an NSAID and a diuretic - the so-called "triple whammy" effect - such patients are at very high risk of developing renal failure.[3]
ACE inhibitors may cause hyperkalemia, because angiotensin II increases aldosterone release. Since aldosterone is responsible for increasing the excretion of potassium, ACE inhibitors ultimately cause retention of potassium.
Some patients develop angioedema due to increased bradykinin levels. There appears to be a genetic predisposition towards this adverse effect in patients who degrade bradykinin slower than average.[4]
Examples
ACE inhibitors can be divided into three groups based on their molecular structure:
Sulfhydryl-containing agents
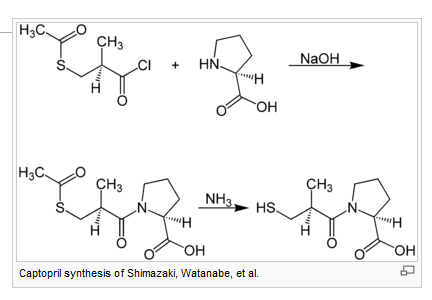
- Captopril (trade name Capoten), the first ACE inhibitor
Dicarboxylate-containing agents
This is the largest group, including:
- Enalapril (Vasotec/Renitec)
- Ramipril (Altace/Tritace/Ramace/Ramiwin)
- Quinapril (Accupril)
- Perindopril (Coversyl/Aceon)
- Lisinopril (Lisodur/Lopril/Novatec/Prinivil/Zestril)
- Benazepril (Lotensin)
Phosphonate-containing agents
- Fosinopril (Monopril), the only member
Naturally occurring
Casokinins and lactokinins are breakdown products of casein and whey that occur naturally after ingestion of milk products, especially cultured milk. Their role in blood pressure control is uncertain.[5]
Comparative information
Comparatively, all ACE inhibitors have similar antihypertensive efficacy when equivalent doses are administered. The main point-of-difference lies with captopril, the first ACE inhibitor, which has a shorter duration of action and increased incidence of certain adverse effects (cf. captopril).
Certain agents in the ACE inhibitor class have been proven, in large clinical studies, to reduce mortality post-myocardial infarction, prevent development of heart failure, etc. While these effects are likely to be class-effects, good evidence-based medicine practice would direct the use of those agents with established clinical efficacy.
Contraindications and precautions
The ACE inhibitors are contraindicated in patients with:
- Previous angioedema associated with ACE inhibitor therapy
- Renal artery stenosis (bilateral, or unilateral with a solitary functioning kidney)
ACE inhibitors should be used with caution in patients with:
- Impaired renal function
- Aortic valve stenosis or cardiac outflow obstruction
- Hypovolemia or dehydration
- Hemodialysis with high flux polyacrylonitrile membranes
ACE inhibitors are ADEC Pregnancy category D, and should be avoided in women who are likely to become pregnant.[1] In the U.S., ACE inhibitors are required to be labelled with a "black box" warning concerning the risk of birth defects when taking during the second and third trimester. It has also been found that use of ACE inhibitors in the first trimester is also associated with a risk of major congenital malformations, particularly affecting the cardiovascular and central nervous systems.[6]
Potassium supplementation should be used with caution and under medical supervision owing to the hyperkalaemic effect of ACE inhibitors.
Angiotensin II receptor antagonists
ACE inhibitors share many common characteristics with another class of cardiovascular drugs called angiotensin II receptor antagonists, which are often used when patients are intolerant of the adverse effects produced by ACE inhibitors. ACE inhibitors do not completely prevent the formation of angiotensin II, as there are other conversion pathways, and so angiotensin II receptor antagonists may be useful because they act to prevent the action of angiotensin II at the AT1 receptor.
Use in combination
While counterintuitive at first glance, the combination therapy of angiotensin II receptor antagonists with ACE inhibitors may be superior to either agent alone. This combination may increase levels of bradykinin while blocking the generation of angiotensin II and its activity at the AT1 receptor. This 'dual blockade' may be more effective than using an ACE inhibitor alone, because angiotensin II can be generated via non-ACE-dependent pathways. Preliminary studies suggest that this combination of pharmacologic agents may be advantageous in the treatment of essential hypertension, chronic heart failure, and nephropathy.[7][8] However, more studies are needed to confirm these highly preliminary results. While statistically significant results have been obtained for its role in treating hypertension, clinical significance may be lacking.[9]
Patients with heart failure may benefit from the combination in terms of reducing morbidity and ventricular remodeling.[10][11]
The most compelling evidence has been found for the treatment of nephropathy: this combination therapy partially reversed the proteinuria and also exhibited a renoprotective effect in patients afflicted with diabetic nephropathy,[7] and pediatric IgA nephropathy.[12]
History
The first step in the development of (ACE) inhibitors was the discovery of angiotensin converting enzyme (ACE) in plasma by Leonard T. Skeggs and his colleagues in 1956. The conversion of the inactive angiotensin I to the potent angiotensin II was thought to take place in the plasma. However, in 1967, Kevin K. F. Ng and John R. Vane showed that the plasma (ACE) was too slow to account for the conversion of angiotensin I to angiotensin II in vivo. Subsequent investigation showed that rapid conversion occur during its passage through the pulmonary circulation.[13]
Bradykinin is rapidly inactivated in the circulating blood and it disappears completely in a single passage through the pulmonary circulation. Angiotensin I also disappears in the pulmonary circulation due to its conversion to angiotensin II. Furthermore, angiotensin II passes through the lungs without any loss. The inactivation of bradykinin and the conversion of angiotensin I to angiotensin II in the lungs was thought to be caused by the same enzyme.[14] In 1970, Ng and Vane using bradykinin potentiating factor (BPF) provided by Sérgio Henrique Ferreira showed that the conversion of angiotensin I to angiotensin II was inhibited during its passage through the pulmonary circulation.[15]
Bradykinin potentiating factor (BPF) is derived from the venom of the pit viper (Bothrops jararaca). It is a family of peptides and its potentiating action is linked to inhibition of bradykinin by ACE. Molecular analysis of BPF yielded a nonapeptide BPF teprotide (SQ 20,881) which showed the greatest (ACE) inhibition potency and hypotensinve effect in vivo. Teprotide had limited clinical value, due to its peptide nature and lack of activity when given orally. In the early 1970s, knowledge of the structure-activity relationship required for inhibition of ACE was growing. David Cushman, Miguel Ondetti and colleagues used peptide analogues to study the structure of ACE, using carboxypeptidase A as a model. Their discoveries led to the development of captopril, the first orally-active ACE inhibitor in 1975.
Captopril was approved by the United States Food and Drug Administration in 1981. The first non-sulfhydryl-containing (ACE) inhibitor enalapril was marketed two years later. Since then, at least twelve other ACE inhibitors have been marketed.
References
- ↑ 1.0 1.1 Rossi S, editor. Australian Medicines Handbook 2006. Adelaide: Australian Medicines Handbook; 2006. ISBN 0-9757919-2-3.
- ↑ Okumura H, Nishimura E, Kariya S, et al. Angiotensin-converting enzyme (ACE) 阻害薬誘発性の咳嗽発現とACE遺伝子型,血漿中ブラジキニン,サブスタンスP及びACE阻害薬濃度との関連性 [No relation between angiotensin-converting enzyme (ACE) inhibitor-induced cough and ACE gene polymorphism, plasma bradykinin, substance P and ACE inhibitor concentration in Japanese patients]. Yakugaku Zasshi 2001;121(3):253-7. Japanese. PMID 11265121
- ↑ Thomas MC. Diuretics, ACE inhibitors and NSAIDs - the triple whammy. Med J Aust 2000;172(4):184–185. PMID 10772593
- ↑ Molinaro G, Cugno M, Perez M, et al. Angiotensin-converting enzyme inhibitor-associated angioedema is characterized by a slower degradation of des-arginine(9)-bradykinin. J Pharmacol Exp Ther 2002;303:232-7. PMID 12235256.
- ↑ FitzGerald RJ, Murray BA, Walsh DJ. Hypotensive peptides from milk proteins. J Nutr 2004;134:980S-8S. PMID 15051858.
- ↑ Cooper WO, Hernandez-Diaz S, Arbogast PG, Dudley JA, Dyer S, Gideon PS, et al. Major congenital malformations after first-trimester exposure to ACE inhibitors. N Engl J Med 2006;354(23):2443-51. PMID 16760444
- ↑ 7.0 7.1 Luno J, Praga M, de Vinuesa SG. The reno-protective effect of the dual blockade of the renin angiotensin system (RAS). Curr Pharm Des 2005;11(10):1291-300. PMID 15853685
- ↑ van de Wal RM, van Veldhuisen DJ, van Gilst WH, Voors AA. Addition of an angiotensin receptor blocker to full-dose ACE-inhibition: controversial or common sense? Eur Heart J 2005;26(22):2361-7. PMID 16105846
- ↑ Finnegan PM, Gleason BL. Combination ACE inhibitors and angiotensin II receptor blockers for hypertension. Ann Pharmacother 2003;37(6):886-9. PMID 12773079
- ↑ Krum H, Carson P, Farsang C, et al. Effect of valsartan added to background ACE inhibitor therapy in patients with heart failure: results from Val-HeFT. Eur J Heart Fail 2004;6(7):937-45. PMID 15556056
- ↑ Solomon SD, Skali H, Anavekar NS, et al. Changes in ventricular size and function in patients treated with valsartan, captopril, or both after myocardial infarction. Circulation 2005;111(25):3411-9. PMID 15967846
- ↑ Yang Y, Ohta K, Shimizu M, et al. Treatment with low-dose angiotensin-converting enzyme inhibitor (ACEI) plus angiotensin II receptor blocker (ARB) in pediatric patients with IgA nephropathy. Clin Nephrol 2005;64(1):35-40. PMID 16047643
- ↑ K.K.F.Ng and J.R.Vane: Conversion of angiotensin I to angiotensin II. Nature 1967, 216, 762-766
- ↑ K.K.F.Ng and J.R.Vane: Fate of angiotensin I in the circulation. Nature 1968, 218, 144-150.
- ↑ K.K.F.Ng and J.R.Vane: Some properties of angiotensin converting enzyme in the lung in vivo. Nature 1970, 225, 1142-1144.
Use of ACE Inhibitors in the Treatment of Hypertension
- In renal artery stenosis, there is decreased perfusion of the kidney and increased release of renin by the juxtaglomerular apparatus. ACE inhibitors inhibit the synthesis of angiotensin II in this hypereninemic state.
- In patients without renal artery stenosis, ACE inhibitors act via 6 mechanisms:
- Inhibition of conversion of circulating angiotensin I to angiotensin II.
- Reduce the secretion of aldosterone to induce natriuresis.
- They induce renal vasodilation and enhance natriuresis.
- The inactivation of vasodilatory bradykinins is reduced.
- Inhibition of local formation of angiotensin II in vascular tissue and myocardium.
- Indirect adrenergic modulation.
- Quality of life. It has been claimed that angiotensin converting enzymes have a favorable side-effect profile and offer an improved quality of life compared with other anti-hypertensives. However, the comparative studies have used medications with central and sexual side-effects, namely methyldopa and Inderal and cough was not included in the analysis.
Angiotensin Converting Enzyme Inhibitors in the Treatment of Congestive Heart failure
- In congestive heart failure, there is a vicious cycle in which a diminished cardiac output decreases baroreceptor stimulation and increases SVR, further increasing the afterload on the failing myocardium.
- Angiotensin converting enzymes work by reducing the SVR and minimizing afterload.
- Patients with CHF are also at risk for VEA and ACE inhibitors may provide a benefit by increasing serum potassium and decreasing circulating norepinephrine levels. Specific studies, however, remain to be done to document this effect.
- Therapy is usually initiated with a test dose of Captopril 6.25 mg because of its shorter half-life and overdiuresis is avoided to prevent a hypereninemic state prior to initiation of the therapy.
- In patients with liver dysfunction Captopril may be preferable to Enalapril because Enalapril requires activation by the liver.
- Diuretics remain the first line agent. ACE inhibitors do not appear to work well in the absence of diuretics and therefore are primarily second line agents.
- Patients with a low sodium concentration ( i.e. below 130 meq/l ) are likely to be hypereninemic and may experience a precipitous fall in blood pressure.
- Avoid hyperkalemia in patients already treated with potassium sparing diuretics or with potassium supplements especially if renal function is already impaired.
Angiotensin Converting Enzymes and Post-Infarction Remodeling
- Both experimental and clinical studies support the use of angiotensin converting enzymes inhibitors to decrease LV dilation post MI.
Angiotensin Converting Enzyme Inhibitors in Patients with Renal Impairment
- Exert a renal vasodilatory effect by dilating efferent arterioles and relieving intraglomerular pressure.
- In diabetic nephropathy, Captopril has been shown to decrease proteinuria without changing creatinine which has been thought to be due to reduced intraglomerular hypertension i.e. reducing the filtered load.
- The role of Captopril in other forms of renal failure is less clear.
Class Side Effects
- Cough, dry, irritating, non-productive. Most common side effect. Patients with CHF frequently cough anyway. Cough was ignored in the "quality of life" study. Incidence may be as high as 10-15%. Thought to be secondary to increased formation of bradykinin and prostaglandins. NSAIDs may help, but at the expense of renal vasodilatory effects of ACE inhibitors.
- Hypotension: When initial blood pressure is very low, further afterload reduction can cause hypotension and renal hypoperfusion.
- Renal side effects: Again, hypotension can lead to renal failure which is usually temporary. 3 situations in which this is a possibility include
- severe CHF
- unilateral renal artery stenosis
- bilateral renal artery stenosis
- Occasionally irreversible renal failure has been precipitated in patients with bilateral renal artery stenosis and this is a contraindication.
- To minimize these possibilities a low dose of short acting Captopril 6.25 mg is given to patients with CHF.
- Angioedema rare, .1% life-threatening. No known method of prediction.
Coadministration with Other Drugs
- Avoid hyperkalemia with K+ sparing agents
- Monitor plasma lithium levels
- Combination with thiazide diuretics increases the hypotensive effects of ACE inhibitors.
- Captopril increases digoxin levels.
- ACE inhibitors and β blockers both decrease renin levels, but their is some evidence to suggest that the effect is additive.
- ACE inhibitors and calcium channel blockers are a good combination because both are free of CNS effects.
Captopril
- The first ACE inhibitor
- t1/2 = 2 to 3 hours
- Dosing:
- HTN: 25 to 50 mg bid
- CHF: 75 to 150 mg daily in 2 to 3 divided doses. Test dose of 6.25 mg, particularly if recently diuresed
- Improves proteinuria in diabetic nephropathy
- Contraindications:
- Bilateral renal artery stenosis
- Unilateral renal artery stenosis in a single kidney
- Immune-based renal disease (4/100 incidence of neutropenia in those with collagen vascular disease and renal impairment).
- Cr > 1.6 mg/dl
- Pre-existing neutropenia
- Hypotension
- Immune-based side-effects peculiar to captopril include taste disturbances (2-7%), skin rashes (4-10%) and neutropenia (1/8600). In patients with renal impairment at the time of initiation of therapy, then WBC counts need to be checked q 2 wks for 3 mos.
- Proteinuria occurs in 1% of patients taking captopril and is due to immune mechanisms and decreased renal perfusion.
Enalapril (Vasotec)
- Longer half-life (4 to 5 hours in HTN and 7 to 8 hours in CHF)
- Requires hydrolysis of the pro-drug to the active drug, enalaprilat in the liver, thus in liver disease requires higher dosing.
- Does not have the sulfhydryl group and therefore less of a risk of immune-based side effects.
- 95% renal excretion
- Peak antihypertensive effect in 4 to 6 hours after the initial dose.
- Dosing:
- HTN: 2.5 to 20 mg daily as 1 or 2 daily doses
- CHF: Initial dosing 2.5 mg to avoid the risk of hypotension and renal failure
- If GFR < 30 ml/min, then reduce the dose
- Side-effects: neutropenia less frequent. Cough still occurs. Can be safe when captopril causes a rash. Regular neutrophil monitoring is not required.
Lisinopril (Zestril, Prinivil)
- Is not a pro-drug, excreted unchanged by the kidney.
- t1/2 = 12 - 14 hours.
- No sulfhydryl group, therefore few immune-based side effects.
- Dosing similar to enalapril.
Benazepril
- Studied in 95 american studies.
- The prodrug is benazepril, the esterified form.
- Benazepril is hydrolyzed in the liver to the active unesterified form, benazeprilat.
- Initial dose: 10 mg PO qd. Chronic dosing: 20 mg or 40 mg PO qd. Occasionally a patient will require 40 mg PO bid.
- Five most common side effects in 3,500 patients were headache, dizziness, fatigue, cough (3.4% in treatment group compared with 1.3% in placebo arm), and nausea.
- In patients with a creatinine clearance less than 30 ml/min a reduction in the initial dosage is decreased.
- No need to reduce dose in cirrhosis.
- No dose reduction in the elderly.