TGF beta
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]
Overview

Transforming growth factor beta (TGF-β) is a protein that comes in three isoforms called TGF-β1, TGF-β2 and TGF-β3; it was also the original name for the founding member of this family that is now called TGF-β1. The TGF-β family is part of a superfamily of proteins known as the transforming growth factor beta superfamily, which includes inhibins, activin, anti-müllerian hormone, bone morphogenetic protein, decapentaplegic and Vg-1. TGF beta controls proliferation, differentiation, and other functions in most cell types. It can also act as a negative autocrine growth factor.
TGF-β is a multifunctional peptide that controls proliferation, differentiation, and other functions in many cell types. TGF-β acts synergistically with TGF-α in inducing cellular transformation (MIM 190170). It also acts as a negative autocrine growth factor. Specific receptors for TGF-β activation trigger apoptosis when activated. Many cells synthesize TGF-β and almost all of them have specific receptors for this peptide.
TGF-β1, TGF-β2, and TGF-β3 all function through the same receptor signaling systems.
The Structure of TGF-β
The peptide structures of the three members of the TGF-β family are highly similar. They are all encoded as large protein precursors; TGF-β1 contains 390 amino acids and TGF-β2 and TGF-β3 each contain 412 amino acids. They each have an N-terminal signal peptide of 20-30 amino acids that they require for secretion from a cell, a pro-region (called latency associated peptide or LAP), and a 112-114 amino acid C-terminal region that becomes the mature TGF-β molecule following its release from the pro-region by proteolytic cleavage.[1] The mature TGF-β protein dimerizes to produce a 25 KDa active molecule with many conserved structural motifs.[2] TGF-β has nine cysteine residues that are conserved among its family; eight form disulphide bonds within the molecule to create a cysteine knot structure characteristic of the TGF-β superfamily while the ninth cysteine forms a bond with the ninth cysteine of another TGF-β molecule to produce the dimer.[3] Many other conserved residues in TGF-β are thought to form secondary structure through hydrophobic interactions. The region between the fifth and sixth conserved cysteines houses the most divergent area of TGF-β molecules that is exposed at the surface of the molecule and is implicated in receptor binding and specificity of TGF-β.
Functions
Role in apoptosis
TGF-ß induces apoptosis in numerous cell types. TGF-ß can induce apoptosis in two ways: The SMAD pathway or the DAXX pathway.
SMAD pathway
The SMAD pathway is the classical signaling pathway that TGF-ß family members signal through.
In this pathway TGF-ß dimers binds to a type II receptor which recruits and phosphorylates a type I receptor.
The type I receptor then recruits and phosphorylates a receptor regulated SMAD (R-SMAD). SMAD3, an R-SMAD, is implicated in inducing apoptosis.
The R-SMAD then binds to the common SMAD (coSMAD) SMAD4 and forms a heterodimeric complex. This complex then enters the cell nucleus where it acts as a transcription factor for various genes, including those to activate the Mitogen-activated protein kinase 8 pathway.
This then triggers apoptosis.
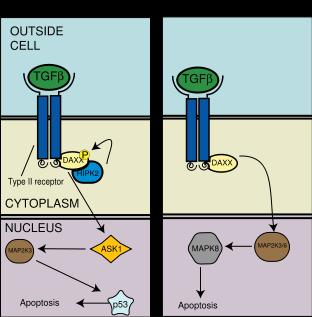
DAXX pathway
TGF-ß may also trigger apoptosis via the death associated protein 6 (DAXX adapter protein).
DAXX has been shown to associate with and bind to the type II TGF-ß receptor kinase.
Role in cell cycle
TGF-ß plays a crucial role in the regulation of the cell cycle.
Role in heart disease
A study at the Saint Louis University School of Medicine has found that cholesterol suppresses the responsiveness of cardiovascular cells to TGF-ß and its protective qualities, thus allowing atherosclerosis to develop. It was also found that statins, drugs that lower cholesterol levels, enhance the responsiveness of cardiovascular cells to the protective actions of TGF-ß, thus helping prevent the development of atherosclerosis and heart disease. [2]
Types
The primary three are:
- TGF beta 1 - TGFB1 (OMIM 190180)
- TGF beta 2 - TGFB2 (OMIM 190220)
- TGF beta 3 - TGFB3 (OMIM 190230)
- TGFβ4 precursor was discovered as a gene upregulated during pre-menstrual phase in the endometrail stroma(Kothapalli et al. 1997) and called EBAF (endometrial bleeding associated factor). Later independently discovered to be involved in vertebrate embryonic left right asymmetry determination, and given the name lefty2 (also called Lefty A).
See also
References
- ↑ Khalil N (1999). "TGF-beta: from latent to active". Microbes Infect. 1 (15): 1255–63. PMID 10611753.
- ↑ Herpin A, Lelong C, Favrel P (2004). "Transforming growth factor-beta-related proteins: an ancestral and widespread superfamily of cytokines in metazoans". Dev Comp Immunol. 28 (5): 461–85. PMID 15062644.
- ↑ Daopin S, Piez K, Ogawa Y, Davies D (1992). "Crystal structure of transforming growth factor-beta 2: an unusual fold for the superfamily". Science. 257 (5068): 369–73. PMID 1631557.
External links
- Description of the TGF beta producing genes at ncbi.nlm.nih.gov
- Diagram of the TGF beta signaling pathway at genome.ad.jp
- TGF-beta at the US National Library of Medicine Medical Subject Headings (MeSH)