Thiazide
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]
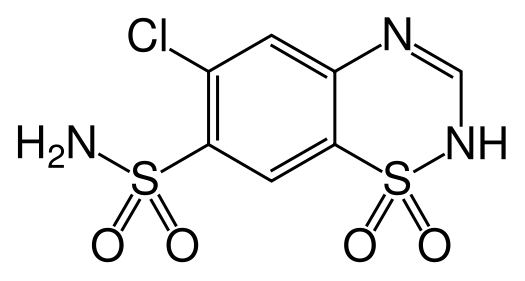
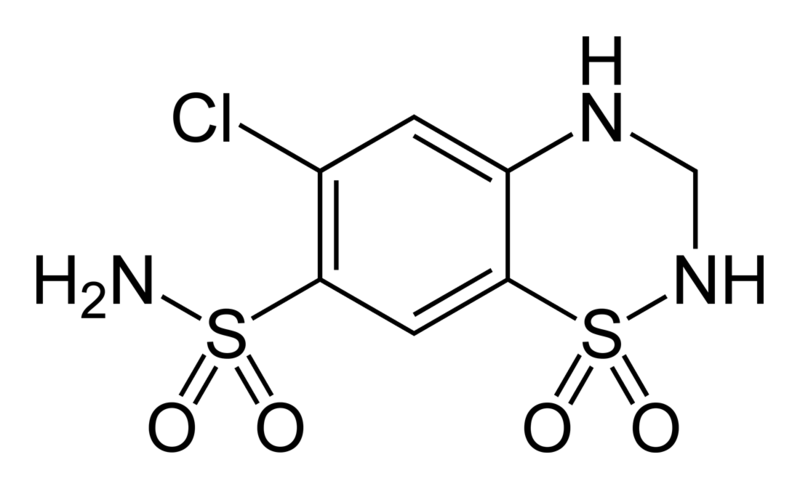
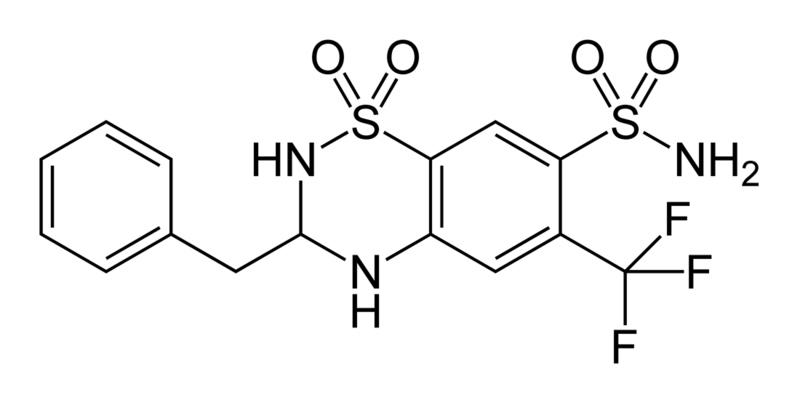
Thiazide is a term used to describe a type of molecule[1] and a class of diuretic.[2]
The members of these diuretics are derived from benzothiadiazine. They inhibit Na+/Cl- reabsorption from the distal convoluted tubules in the kidneys by blocking the thiazide-sensitive Na+-Cl- symporter. Thiazides also cause loss of potassium and an increase in serum uric acid. The chemical structure of the original thiazide diuretics contained a thiazide ring system; the term is also used for drugs with a similar action that are not chemically thiazides, such as chlortalidone and metolazone.
Denomination
That thiazide both refers to the type of molecule and the medication can sometimes lead to confusion, because some molecules can be considered thiazide diuretics, although they are not thiazides from a chemical perspective. In this context, "thiazide" refers to a drug which acts at a "thiazide receptor"[3], which is believed to be a sodium-chloride symporter.
Primary uses
Because of their vasodilator properties, Thiazides are often used to treat hypertension. They are the recommended first-line treatment in the US (JNC VII)[4] guidelines and a recommended treatment in the European (ESC/ESH)[5] guidelines. They have been shown to prevent hypertension-related morbidity and mortality although the mechanism is not fully understood. Thiazides cause vasodilation by desensitizing the vascular smooth muscle cells to calcium release induced by norepinephrine.[6]
Among the thiazides and thiazide-like diurectics, chlorthalidone may be more effective for hypertension control than hydrochlorothiazide.[7]
Side effects
Side effects can include hypokalemia, increased serum cholesterol, and impotence. The side effect of hypokalemia has motivated combining thiazides with potassium sparing diuretics (eg with amiloride in co-amilozide) and with the newer ACE inhibitors, which also lower blood pressure but cause hyperkalemia as a side effect.
Long-term usage of thiazides is also linked to increased levels of homocysteine, a toxic amino acid byproduct, that has been associated with atherosclerosis. It is recommended that patients receiving long-term thiazide treatments also receive folic acid supplements to combat the risk. They have been known to cause a paradoxical effect in Diabetes insipidus, where they reduce the volume of urine. Thiazide diuretics are capable of inhibiting urate secretions.
About 3% of the population will have an allergic reaction to a thiazide. Signs and symptoms of an allergic reaction include a rash(either eczematous reaction with red itchy scaly skin and crusting or erythema anular centrifugum), hives, itching, nasal congestion, shortness of breath, wheezing, tachycardia, cough, nausea or diarrhea. Anaphylactic reactions with hypotension, angioedema and a leukocytoclastic vasculitis are rare.
Thiazide-induced hyponatremia
"Fractional uric acid clearance was elevated and serum uric acid levels were low. These results are consistent with excess ADH activity as having caused hyponatraemia induced by thiazides in [most] cases"[8].
Epidemiology
Over 5 years of treatment, one in 15 patients treated with thiazides may have a low sodium value (sodium≤130 mmol/L) and a mean of 1.75 years after treatment started[9].
However, a more recent report by Kaiser, who also published that a hypertension clinical practice guideline that is based on a thiazide-angiotensin converting enzyme inhibitor combination, has great improved hypertension control over a large population[10] and that their decline and cardiovascular mortality exceeds the national decline[11], found 1.4% per year rate of hyponatremia among patients treated with the combination pill[12].
Predictors include baseline hyponatremia (sodium < 140 mEq/L)[13]
Hypokalemia at baseline may be a better predictor than hyonatremia at baseline[14].
Practice gaps
In 18% of patients on thiazides the sodium was not monitored[9] over the course of study and in another study, 42% were not montiored in the first 90 days[13]. Other studies have found gaps[15].
In half of cases where a sodium less than 130 is found, the diuretic is not stopped[16].
Diagnosis
Fractional uric acid clearance is elevated and serum uric acid levels are low.[8].
If the FEurate is normal (4 to 11%), this suggests psychogenic polydipsia or reset osmostat[17].
Treatment
Various studies of have compared loop diurectics, including furosemide, with thiazide diuretics as they have opposite handling of sodium in the kidney[18][19]. Trial have been summarized in a systematic review by the Cochrane Collaboration[20].
Potassium replacement may help[21].
Other uses
Thiazides also lower urinary calcium excretion, making them useful in preventing calcium-containing kidney stones. This effect is associated with positive calcium balance and is associated with an increase in bone mineral density and reductions in fracture rates attributable to osteoporosis. Because of those properties, they are also used in the treatment of Dent's Disease or idiopathic hypercalciuria.
Thiazide may be combined with ACE inhibitors to increase diuresis without changing plasma potassium concentrations. While ACE inhibitors cause diuresis with potassium retention, thiazide increases potassium excretion. Their combined effects on potassium cancel each other out.
Breast milk
It should be noted that thiazides pass through breast milk, and in some cases, decrease the flow of breast milk. There is no specific information regarding the use of thiazides in children, but it is still advised that mothers avoid using thiazides during the first month of breast feeding.
Mechanisms of hypokalemia
There are several mechanisms by which thiazide diuretics cause hypokalaemia (decreased plasma potassium concentration):
- Increased delivery of sodium to the collecting ducts causes the Na/K exchanger to be activated resulting in K (and H+) loss.
- Activation of renin-angiotensin-aldosterone system by the diuretic hypovolaemia: body responds to hypovolaemia by opposing diuresis, one effect of which is to produce aldosterone which stimulates the Na/K exchanger, resulting in further loss of potassium. For this reason, ACE inhibitors, which inhibit angiotensin II production and therefore aldosterone activation, are frequently used in combination with thiazides to combat hypokalaemia.
- Flow rate in nephron is increased under diuresis, reducing potassium concentration in the lumen, thus increasing the potassium gradient. Potassium loss through the many potassium channels, such as ROMK. These are not exchangers; they allow facilitated diffusion, so the increased gradient is directly responsible for increased diffusion.
Thiazides overuse must be differentiated from other causes of hypercalcaemia
| Differential diagnosis of hyperparathyroidism on the basis of hypercalcemia | ||||||||
|---|---|---|---|---|---|---|---|---|
| Disorder | Mechanism of hypercalcemia | Clinical features | Laboratory findings | Imaging & diagnostic modalities | ||||
| PTH | Calcium | Phosphate | Other findings | |||||
| Hyperparathyroidism | Primary hyperparathyroidism | Increase in secretion of parathyroid hormone (PTH) from a primary process in parathyroid gland. Parathyroid hormone causes increase in serum calcium. |
|
↑ | ↑ | ↓/Normal | Normal/↑ calcitriol | Findings of bone resorption:
Preoperative localization of hyperfunctioning parathyroid gland:
Predicting post-operative success:
|
| Secondary hyperparathyroidism | Increase in secretion of parathyroid hormone (PTH) from a secondary process. Parathyroid hormone causes increase in serum calcium after long periods. |
|
↑ | ↓/Normal | ↑ | -- | ||
| Tertiary hyperparathyroidism | Continuous elevation of parathyroid hormone (PTH) even after successful treatment of the secondary cause of elevated parathyroid hormone. Parathyroid hormone causes increase in serum calcium. |
|
↑ | ↑ | ↑ | -- | ||
| Familial hypocalciuric hypercalcemia | This is a genetic disorder caused my mutation in calcium-sensing receptor gene. |
|
Normal/↑ | Normal/↑ | -- | -- |
| |
| Malignancy[22] | Humoral hypercalcemia of malignancy[23][24][25] | Tumor cells secretes parathyroid hormone-related protein (PTHrP) which has similar action as parathyroid hormone. |
|
-- | ↑ | ↓/Normal | ↑ PTHrP
Normal/↑ calcitriol |
|
| Osteolytic tumors | Multiple myeloma produces osteolysis of bones causing hypercalcemia. Osteolytic metasteses can cause bone resorption causing hypercalcemia. |
|
↓ | ↑ | -- | -- | ||
| Production of calcitirol | Some tumors has ectopic activity of 1-alpha-hydroxylase leading to increased production of calcitriol. Calcitriol is active form of vitamin D and causes hypercalcemia. |
|
-- | ↑ | -- | ↑ Calcitriol | ||
| Ectopic parathyroid hormone[26] | Some tumors leads to ectopic production of parathyroid hormone. |
|
↑ | ↑ | ↓/Normal | Normal/↑ calcitriol | ||
| Medication induced | Lithium[27] | Lithium lowers urinary calcium and causes hypercalcemia. Lithium has been reported to cause an increase in parathyroid hormone and enlargement if parathyroid gland after weeks to months of therapy. |
|
↑ | ↑ | -- | -- |
|
| Thiazide diuretics | Thiazide diuretics lowers urinary calcium excretion and causes hypercalcemia. |
|
-- | ↑ | -- | -- | -- | |
| Nutritional | Milk-alkali syndrome | Hypercalcemia is be caused by high intake of calcium carbonate. |
|
-- | ↑ | -- | -- | |
| Vitamin D toxicity | Excess vitamin D causes increased absorption of calcium from intestine causing hypercalcemia. |
|
-- | ↑ | -- | ↑ Vitamin D (calcidiol and/or calcitriol) | -- | |
| Granulomatous disease | Sarcoidosis[30] | Hypercalcemia is causes by endogeous production of calcitriol by disease-activated macrophages. |
|
-- | ↑ | -- | ↑ Calcitriol
↑ ACE levels |
|
References
Moreno, E (2006). Affinity-defining Domains in the Na-Cl Cotransporter. J. Biol. Chem. 281, 17266-17275.
- ↑ Thiazides at the US National Library of Medicine Medical Subject Headings (MeSH)
- ↑ Thiazide+Diuretics at the US National Library of Medicine Medical Subject Headings (MeSH)
- ↑ thiazide+receptor at the US National Library of Medicine Medical Subject Headings (MeSH)
- ↑ "The Seventh Report of the Joint National Committee on Prevention, Detection, Evaluation, and Treatment of High Blood Pressure (JNC 7)".
- ↑ "escardio.org".
- ↑ Zhu Z, Zhu S, Liu D, Cao T, Wang L, Tepel M (2005). "Thiazide-like diuretics attenuate agonist-induced vasoconstriction by calcium desensitization linked to Rho kinase". Hypertension. 45 (2): 233–9. doi:10.1161/01.HYP.0000152701.97426.5f. PMID 15611360.
- ↑ Bakris GL, Sica D, White WB, Cushman WC, Weber MA, Handley A; et al. (2012). "Antihypertensive Efficacy of Hydrochlorothiazide vs Chlorthalidone Combined with Azilsartan Medoxomil". Am J Med. 125 (12): 1229.e1–1229.e10. doi:10.1016/j.amjmed.2012.05.023. PMID 22939358.
- ↑ 8.0 8.1 Sonnenblick M, Rosin AJ (1986). "Significance of the measurement of uric acid fractional clearance in diuretic induced hyponatraemia". Postgrad Med J. 62 (728): 449–52. doi:10.1136/pgmj.62.728.449. PMC 2418766. PMID 3774674.
- ↑ 9.0 9.1 Leung AA, Wright A, Pazo V, Karson A, Bates DW (2011). "Risk of thiazide-induced hyponatremia in patients with hypertension". Am J Med. 124 (11): 1064–72. doi:10.1016/j.amjmed.2011.06.031. PMID 22017784. Review in: Ann Intern Med. 2012 Apr 17;156(8):JC4-11
- ↑ Jaffe MG, Young JD (2016). "The Kaiser Permanente Northern California Story: Improving Hypertension Control From 44% to 90% in 13 Years (2000 to 2013)". J Clin Hypertens (Greenwich). 18 (4): 260–1. doi:10.1111/jch.12803. PMID 26939059.
- ↑ Sidney S, Sorel ME, Quesenberry CP, Jaffe MG, Solomon MD, Nguyen-Huynh MN; et al. (2018). "Comparative Trends in Heart Disease, Stroke, and All-Cause Mortality in the United States and a Large Integrated Healthcare Delivery System". Am J Med. 131 (7): 829–836.e1. doi:10.1016/j.amjmed.2018.02.014. PMC 6005733. PMID 29625083.
- ↑ An, Jaejin; Mefford, Matthew; Ni, Liang; Wei, Rong; Zhou, Hui; Harrison, Teresa N; Brettler, Jeff; Muntner, Paul; Reynolds, Kristi (September 2021). "Abstract P108: Side Effects Of Initial Combination Versus Monotherapy For Patients With Hypertension". Hypertension. 78 (Suppl 1). doi:10.1161/hyp.78.suppl_1.P108. eISSN 1524-4563. ISSN 0194-911X.
- ↑ 13.0 13.1 Makam AN, Boscardin WJ, Miao Y, Steinman MA (2014). "Risk of thiazide-induced metabolic adverse events in older adults". J Am Geriatr Soc. 62 (6): 1039–45. doi:10.1111/jgs.12839. PMC 4128471. PMID 24823661. Review in: Ann Intern Med. 2014 Oct 21;161(8):JC11
- ↑ Chow KM, Szeto CC, Kwan BC, Ma TK, Leung CB, Li PK (2012). "Fracture risk after thiazide-associated hyponatraemia". Intern Med J. 42 (7): 760–4. doi:10.1111/j.1445-5994.2011.02642.x. PMID 22150918.
- ↑ McDowell SE, Ferner RE (2011). "Biochemical monitoring of patients treated with antihypertensive therapy for adverse drug reactions: a systematic review". Drug Saf. 34 (11): 1049–59. doi:10.2165/11593980-000000000-00000. PMID 21981433.
- ↑ Burst V, Grundmann F, Kubacki T, Greenberg A, Becker I, Rudolf D; et al. (2017). "Thiazide-Associated Hyponatremia, Report of the Hyponatremia Registry: An Observational Multicenter International Study". Am J Nephrol. 45 (5): 420–430. doi:10.1159/000471493. PMC 5452281. PMID 28419981.
- ↑ Maesaka JK, Imbriano LJ, Miyawaki N (2018). "Determining Fractional Urate Excretion Rates in Hyponatremic Conditions and Improved Methods to Distinguish Cerebral/Renal Salt Wasting From the Syndrome of Inappropriate Secretion of Antidiuretic Hormone". Front Med (Lausanne). 5: 319. doi:10.3389/fmed.2018.00319. PMC 6284366. PMID 30560127.
- ↑ Valmin K, Hansen T (1975). "Treatment of benign essential hypertension: comparison of furosemide and hydrochlorothiazide". Eur J Clin Pharmacol. 8 (6): 393–401. doi:10.1007/BF00562312. PMID 786684.
- ↑ Araoye MA, Chang MY, Khatri IM, Freis ED (1978). "Furosemide compared with hydrochlorothiazide. Long-term treatment of hypertension". JAMA. 240 (17): 1863–6. PMID 357764.
- ↑ Musini VM, Rezapour P, Wright JM, Bassett K, Jauca CD (2015). "Blood pressure-lowering efficacy of loop diuretics for primary hypertension". Cochrane Database Syst Rev (5): CD003825. doi:10.1002/14651858.CD003825.pub4. PMC 7156893 Check
|pmc=value (help). PMID 26000442. - ↑ Fichman MP, Vorherr H, Kleeman CR, Telfer N (1971). "Diuretic-induced hyponatremia". Ann Intern Med. 75 (6): 853–63. doi:10.7326/0003-4819-75-6-853. PMID 4944156.
- ↑ Mirrakhimov AE (2015). "Hypercalcemia of Malignancy: An Update on Pathogenesis and Management". N Am J Med Sci. 7 (11): 483–93. doi:10.4103/1947-2714.170600. PMC 4683803. PMID 26713296.
- ↑ Ratcliffe WA, Hutchesson AC, Bundred NJ, Ratcliffe JG (1992). "Role of assays for parathyroid-hormone-related protein in investigation of hypercalcaemia". Lancet. 339 (8786): 164–7. doi:10.1016/0140-6736(92)90220-W. PMID 1346019.
- ↑ Ikeda K, Ohno H, Hane M, Yokoi H, Okada M, Honma T, Yamada A, Tatsumi Y, Tanaka T, Saitoh T (1994). "Development of a sensitive two-site immunoradiometric assay for parathyroid hormone-related peptide: evidence for elevated levels in plasma from patients with adult T-cell leukemia/lymphoma and B-cell lymphoma". J. Clin. Endocrinol. Metab. 79 (5): 1322–7. doi:10.1210/jcem.79.5.7962324. PMID 7962324.
- ↑ Horwitz MJ, Tedesco MB, Sereika SM, Hollis BW, Garcia-Ocaña A, Stewart AF (2003). "Direct comparison of sustained infusion of human parathyroid hormone-related protein-(1-36) [hPTHrP-(1-36)] versus hPTH-(1-34) on serum calcium, plasma 1,25-dihydroxyvitamin D concentrations, and fractional calcium excretion in healthy human volunteers". J. Clin. Endocrinol. Metab. 88 (4): 1603–9. doi:10.1210/jc.2002-020773. PMID 12679445.
- ↑ VanHouten JN, Yu N, Rimm D, Dotto J, Arnold A, Wysolmerski JJ, Udelsman R (2006). "Hypercalcemia of malignancy due to ectopic transactivation of the parathyroid hormone gene". J. Clin. Endocrinol. Metab. 91 (2): 580–3. doi:10.1210/jc.2005-2095. PMID 16263810.
- ↑ Mallette LE, Khouri K, Zengotita H, Hollis BW, Malini S (1989). "Lithium treatment increases intact and midregion parathyroid hormone and parathyroid volume". J. Clin. Endocrinol. Metab. 68 (3): 654–60. doi:10.1210/jcem-68-3-654. PMID 2918061.
- ↑ Jacobus CH, Holick MF, Shao Q, Chen TC, Holm IA, Kolodny JM, Fuleihan GE, Seely EW (1992). "Hypervitaminosis D associated with drinking milk". N. Engl. J. Med. 326 (18): 1173–7. doi:10.1056/NEJM199204303261801. PMID 1313547.
- ↑ Hoeck HC, Laurberg G, Laurberg P (1994). "Hypercalcaemic crisis after excessive topical use of a vitamin D derivative". J. Intern. Med. 235 (3): 281–2. PMID 8120527.
- ↑ Dusso AS, Kamimura S, Gallieni M, Zhong M, Negrea L, Shapiro S, Slatopolsky E (1997). "gamma-Interferon-induced resistance to 1,25-(OH)2 D3 in human monocytes and macrophages: a mechanism for the hypercalcemia of various granulomatoses". J. Clin. Endocrinol. Metab. 82 (7): 2222–32. doi:10.1210/jcem.82.7.4074. PMID 9215298.
External links