Methotrexate (injection)
 | |
| Clinical data | |
|---|---|
| Pregnancy category | |
| Routes of administration | oral, IV, IM, SC, intrathecal |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | 17–90% |
| Metabolism | hepatic |
| Elimination half-life | 3–15 hours (dose dependent) |
| Excretion | renal 48–100% |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| DrugBank | |
| E number | {{#property:P628}} |
| ECHA InfoCard | {{#property:P2566}}Lua error in Module:EditAtWikidata at line 36: attempt to index field 'wikibase' (a nil value). |
| Chemical and physical data | |
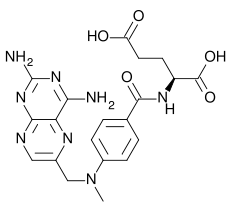
| Formula | C20H22N8O5 |
| Molar mass | 454.44 g/mol |
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]
Overview
Methotrexate (rINN) (IPA: Template:IPA), abbreviated MTX and formerly known as amethopterin, is an antimetabolite and antifolate drug used in treatment of cancer and autoimmune diseases. It acts by inhibiting the metabolism of folic acid. Methotrexate replaced the more powerful and toxic antifolate aminopterin, and the two should not be confused with each other.
History
Methotrexate originated in the 1940s when Sidney Farber at Children's Hospital Boston was testing the effects of folic acid on cancer. This inspired chemists at the drug company Lederle to start looking for antimetabolites resembling folate. The result was methotrexate, which was developed in 1948 by Yellapragada Subbarao from Lederle. Methotrexate gained Food and Drug Administration (FDA) approval as an oncology drug in 1953.
Uses
In chemotherapy
Methotrexate was originally used, as part of combination chemotherapy regimens, to treat many kinds of cancers. It is still the mainstay for the treatment of many neoplastic disorders including acute lymphoblastic leukemia.
Other uses
More recently it has come into use as a treatment for some autoimmune diseases, including ankylosing spondylitis, Crohn's disease, psoriasis, psoriatic arthritis, and rheumatoid arthritis (see disease-modifying antirheumatic drugs). A parallel use with TNFα blockers as infliximab or etanercept has been shown to markedly improve symptoms.[1]
Although not indicated for this use, methotrexate is also sometimes used (generally in combination with misoprostol) to terminate early pregnancies, particularly ectopic pregnancies.
Pharmacokinetics
Methotrexate is a weak dicarboxylic acid with pKa 4.8 and 5.5, and thus it is mostly ionized at physiologic pH. Oral absorption is saturatable and thus dose-dependent, with doses less than 40mg/M2 having 42% bioavailability and doses greater than 40mg/M2 only 18%. Mean oral bioavailability is 33% (13-76% range), and there is no clear benefit to subdividing an oral dose. Mean intramuscular bioavailability is 76%.
Methotrexate is metabolized by intestinal bacteria to the inactive metabolite 4-amino-4-deoxy-N-methylpteroic acid (DAMPA) and accounts for less than 5% loss of the oral dose.
Factors that decrease absorption include food, oral non-absorbable antibiotics (e.g. vancomycin, neomycin, and bacitracin), and more rapid transit through the Gastrointestinal (GI) tract such as diarrhea, while slower transit time in the GI tract from constipation will increase absorption.
Administration
It can be taken orally or administered by injection (subcutaneous, intramuscular, intravenous or intrathecal). Although daily preparations are occasionally used, most patients take weekly doses, which decreases the risk of certain side-effects.
Adverse effects
Possible side effects can include anemia, neutropenia, increased risk of bruising, nausea and vomiting, dermatitis and diarrhea. A small percentage of patients develop hepatitis, and there is an increased risk of pulmonary fibrosis.
The higher doses of methotrexate often used in cancer chemotherapy can cause toxic effects to the rapidly-dividing cells of bone marrow and gastrointestinal mucosa. The resulting myelosuppression and mucositis are often prevented (termed methotrexate "rescue") by using folinic acid supplements (not to be confused with folic acid).
Methotrexate is a highly teratogenic drug and categorized in Pregnancy Category X by the FDA. Women must not take the drug during pregnancy, if there is a risk of becoming pregnant, or if they are breastfeeding. Men who are trying to get their partner pregnant must also not take the drug. To engage in any of these activities (after discontinuing the drug), women must wait until the end of a full ovulation cycle and men must wait three months.
There is a risk of a severe adverse reaction if penicillin is prescribed alongside methotrexate.
Interestingly, there have also been some reports of central nervous system reactions to methotrexate especially when given via the intrathecal route which include myelopathies and leucoencephalopathies.
Mode of action
Methotrexate competitively and reversibly inhibits dihydrofolate reductase (DHFR), an enzyme that is part of the folate synthesis metabolic pathway. The affinity of methotrexate for DHFR is about one thousand-fold that of folate for DHFR. Dihydrofolate reductase catalyses the conversion of dihydrofolate to the active tetrahydrofolate. Folic acid is needed for the de novo synthesis of the nucleoside thymidine, required for DNA synthesis. Also, folate is needed for purine base synthesis, so all purine synthesis will be inhibited. Methotrexate, therefore, inhibits the synthesis of DNA, RNA, thymidylates, and proteins.
Methotrexate acts specifically during DNA and RNA synthesis, and thus it is cytotoxic during the S-phase of the cell cycle. Logically, it therefore has a greater toxic effect on rapidly dividing cells (such as malignant and myeloid cells, and GI & oral mucosa), which replicate their DNA more frequently, and thus inhibits the growth and proliferation of these non-cancerous cells as well causing side effects listed above.
Lower doses of methotrexate have been shown to be very effective for the management of rheumatoid arthritis and psoriasis. In these cases inhibition of dihydrofolate reductase (DHFR) is not thought to be the main mechanism, rather the inhibition of enzymes involved in purine metabolism, leading to accumulation of adenosine, or the inhibition of T cell activation and suppression of intercellular adhesion molecule expression by T cells.[2]
References
- ↑ Klareskog L, van der Heijde D, de Jager JP, Gough A, Kalden J, Malaise M, Martin Mola E, Pavelka K, Sany J, Settas L, Wajdula J, Pedersen R, Fatenejad S, Sanda M (2004). "Therapeutic effect of the combination of etanercept and methotrexate compared with each treatment alone in patients with rheumatoid arthritis: double-blind randomised controlled trial". Lancet. 363 (9410): 675–81. PMID 15001324.
- ↑ Johnston A, Gudjonsson JE, Sigmundsdottir H, Ludviksson BR, Valdimarsson H; (2005). "The anti-inflammatory action of methotrexate is not mediated by lymphocyte apoptosis, but by the suppression of activation and adhesion molecules". Clin Immunol. 114 (Feb.): 154–163. PMID 15639649.
- Pages with script errors
- CS1 maint: Multiple names: authors list
- E number from Wikidata
- ECHA InfoCard ID from Wikidata
- Chemical articles with unknown parameter in Infobox drug
- Articles without EBI source
- Chemical pages without ChemSpiderID
- Articles without KEGG source
- Articles without InChI source
- Articles without UNII source
- Articles containing unverified chemical infoboxes
- Drugs
- Chemotherapeutic agents
- Immunosuppressive agents
- Dihydrofolate reductase inhibitors