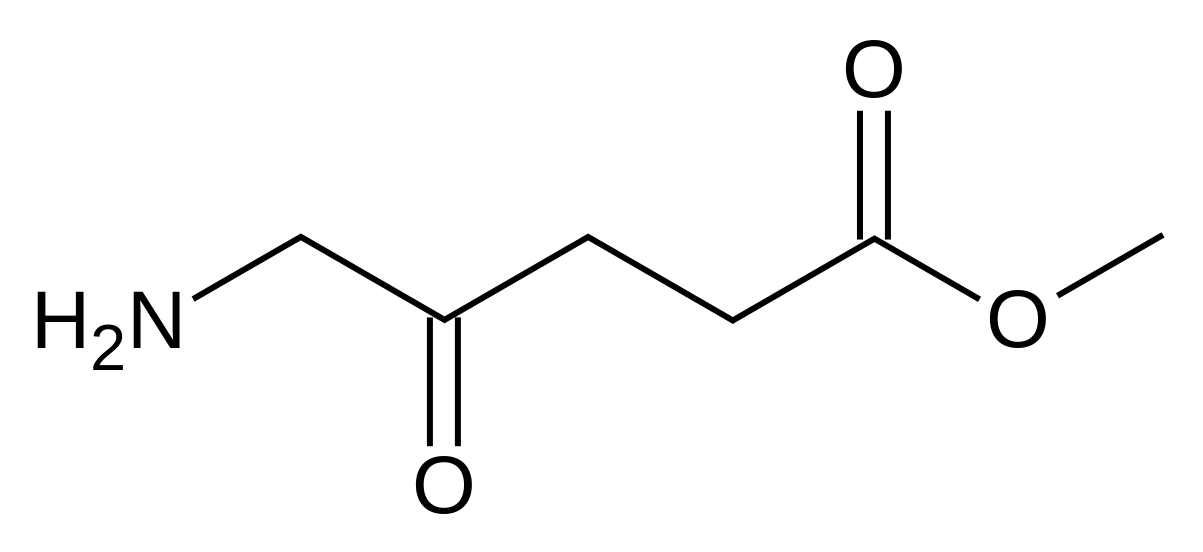
Methyl aminolevulinate
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]; Associate Editor(s)-in-Chief: Ammu Susheela, M.D. [2]
WikiDoc MAKES NO GUARANTEE OF VALIDITY. WikiDoc is not a professional health care provider, nor is it a suitable replacement for a licensed healthcare provider. WikiDoc is intended to be an educational tool, not a tool for any form of healthcare delivery. The educational content on WikiDoc drug pages is based upon the FDA package insert, National Library of Medicine content and practice guidelines / consensus statements. WikiDoc does not promote the administration of any medication or device that is not consistent with its labeling. Please read our full disclaimer here.
Overview
Methyl aminolevulinate is an antioxidant that is FDA approved for the treatment of non-hyperkeratotic actinic keratoses of the face and scalp. Common adverse reactions include serious erythema and facial edema.
Adult Indications and Dosage
FDA-Labeled Indications and Dosage (Adult)
- Metvixia Cream in combination with 570 to 670 nm wavelength red light illumination using the CureLight BroadBand Model CureLight 01 lamp is indicated for treatment of non-hyperkeratotic actinic keratoses of the face and scalp in immunocompetent patients when used in conjunction with lesion preparation (debridement using a sharp dermal curette) in the physician’s office when other therapies are unacceptable or considered medically less appropriate.
- Photodynamic therapy for non-hyperkeratotic actinic keratoses with Metvixia Cream is a multi-stage process as described below: Two treatment sessions 7 days apart should be conducted. Not more than one gram (half a tube) of Metvixia Cream should be applied per treatment session.
- One Metvixia -PDT session consists of:
Lesion debriding
- Before applying Metvixia Cream, the surface of the lesions should be prepared with a small dermal curette to remove scales and crusts and roughen the surface of the lesion. This is to facilitate access of the cream and light to all parts of the lesion.
Off-Label Use and Dosage (Adult)
Guideline-Supported Use
There is limited information regarding Off-Label Guideline-Supported Use of Methyl aminolevulinate in adult patients.
Non–Guideline-Supported Use
There is limited information regarding Off-Label Non–Guideline-Supported Use of Methyl aminolevulinate in adult patients.
Pediatric Indications and Dosage
FDA-Labeled Indications and Dosage (Pediatric)
There is limited information regarding FDA-Labeled Use of Methyl aminolevulinate in pediatric patients.
Off-Label Use and Dosage (Pediatric)
Guideline-Supported Use
There is limited information regarding Off-Label Guideline-Supported Use of Methyl aminolevulinate in pediatric patients.
Non–Guideline-Supported Use
There is limited information regarding Off-Label Non–Guideline-Supported Use of Methyl aminolevulinate in pediatric patients.
Contraindications
- Metvixia Cream is contraindicated in patients with cutaneous photosensitivity, or known allergies to porphyrins, and in patients with known sensitivities to any of the components of Metvixia Cream, which includes peanut and almond oil.
- This product contains refined peanut oil
Warnings
- Metvixia Cream is intended for topical use in the physician’s office by trained physicians only. Do not apply to the eyes or to mucous membranes.
- Metvixia Cream has demonstrated a high rate of contact sensitization (allergenicity). Care should be taken by the physician applying Metvixia Cream to avoid inadvertent skin contact. Nitrile gloves should be worn when applying and removing the cream. Vinyl and latex gloves do not provide adequate protection when using this product.
- Metvixia Cream when used with CureLight BroadBand Model CureLight 01 lamp must be used with appropriate protective sleeves obtained from the product manufacturer to decrease the risk of blood-borne transmitted diseases (hepatitis, HIV, etc.). Change the disposable covers for the device (probe and horseshoe positioning device) between patients. Universal Precautions should be used with this treatment.
Adverse Reactions
Clinical Trials Experience
Dermal Safety Studies
- Provocative studies to evaluate irritancy and sensitization have demonstrated that Metvixia Cream is an irritant and sensitizer. A provocative cumulative irritancy and sensitization (allergenicity) study of Metvixia Cream with a cross-sensitization challenge with ALA was performed in 156 subjects. Only 98 of the 156 subjects tested entered the challenge phase. Fifty-two percent of the subjects (30/58), who agreed to challenge with Metvixia Cream, were positive (sensitized). Forty subjects refused challenge with Metvixia Cream and 60 withdrew. At least 58 of the 60 subjects who withdrew from the study discontinued due to irritation/sensitization.
- Ninety-eight subjects agreed to challenge with ALA. Two percent of the ALA challenged subjects (2/98) were scored as equivocal reactions and 2% in the paraffin vehicle group were scored as positive.
Adverse Events
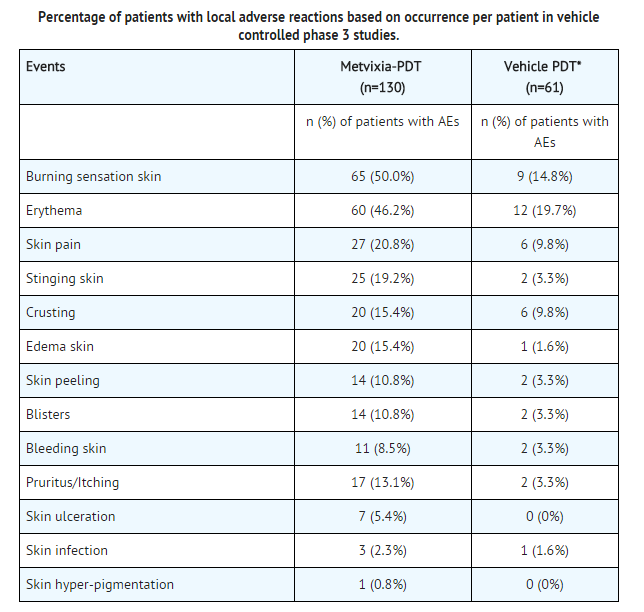
- In vehicle-controlled phase 3 studies of actinic keratosis, 88% of patients treated with Metvixia Cream reported one or more adverse events.
- Burning was the most frequent complaint, reported by 50% of patients (ranging from mild, to severe) and 9% of those patients reported severe burning sensation. Pain in the skin was reported by 21% of patients and 7% had severe pain. Local erythema lasting up to two weeks and edema up to one week after treatment were reported by 31% and 6% of patients.
- Symptoms and signs of local phototoxicity were observed in 88% of patients treated with Metvixia Cream in all clinical studies of Metvixia -PDT for actinic keratoses.
- The majority of patients in all the clinical trials had local pain or discomfort upon illumination. There were 4 (1.0%) withdrawals/discontinuations among 383 patients treated with Metvixia Cream in all the clinical trials of actinic keratosis, all of which were due to the adverse event of local pain on illumination.
Postmarketing Experience
- There have been reported instances of patients treated with Metvixia Cream (2 out of 130) who have developed squamous cell and basal cell carcinoma at the site of treatment. The relationship to treatment with Metvixia Cream is unknown.
- Serious erythema and facial edema have been described in European post-marketing reports.
Drug Interactions
There is limited information regarding Methyl aminolevulinate Drug Interactions in the drug label.
Use in Specific Populations
Pregnancy
- Animal reproduction studies have not been conducted with Metvixia Cream. It is also not known whether Metvixia Cream can cause fetal harm when administered to a pregnant woman or can affect reproduction capacity.
Metvixia Cream should be given to a pregnant woman only if clearly needed.
Pregnancy Category (AUS):
- Australian Drug Evaluation Committee (ADEC) Pregnancy Category
There is no Australian Drug Evaluation Committee (ADEC) guidance on usage of Methyl aminolevulinate in women who are pregnant.
Labor and Delivery
There is no FDA guidance on use of Methyl aminolevulinate during labor and delivery.
Nursing Mothers
- The amount of methyl aminolevulinate secreted into human breast milk following topical administration of Metvixia Cream is not known. Because many drugs are secreted in human milk, caution should be exercised when Metvixia Cream is administered to a nursing mother. If Metvixia Cream is used in a nursing mother, a decision should be made whether or not to stop nursing.
Pediatric Use
- It is not recommended that Metvixia Cream be used in pediatric patients. Actinic keratosis is rarely found in pediatric patients.
Geriatic Use
- Seventy percent (269 among 383) of the patients treated with Metvixia Cream in all clinical studies of actinic keratosis were 65 years of age or older. No overall differences in safety and efficacy were observed between patients aged 65 years and older and those who were younger.
Gender
There is no FDA guidance on the use of Methyl aminolevulinate with respect to specific gender populations.
Race
There is no FDA guidance on the use of Methyl aminolevulinate with respect to specific racial populations.
Renal Impairment
There is no FDA guidance on the use of Methyl aminolevulinate in patients with renal impairment.
Hepatic Impairment
There is no FDA guidance on the use of Methyl aminolevulinate in patients with hepatic impairment.
Females of Reproductive Potential and Males
There is no FDA guidance on the use of Methyl aminolevulinate in women of reproductive potentials and males.
Immunocompromised Patients
There is no FDA guidance one the use of Methyl aminolevulinate in patients who are immunocompromised.
Administration and Monitoring
Administration
Monitoring
There is limited information regarding Monitoring of Methyl aminolevulinate in the drug label.
IV Compatibility
There is limited information regarding IV Compatibility of Methyl aminolevulinate in the drug label.
Overdosage
Metvixia Cream Overdose
- Metvixia Cream overdose has not been reported. If the patient for any reason cannot have the red light treatment during the prescribed period after application (the 3 hour timespan), the cream should be rinsed off, and the patient should protect the exposed area from sunlight, prolonged or intense light for two days.
Red Light Overdose
- There is no information on overdose of red light following Metvixia Cream application.
- In case of red light overexposure and skin burn occurs, the patient should be treated according to standard of practice guidelines for treatment of cutaneous burns.
Pharmacology
| |
Methyl aminolevulinate
| |
| Systematic (IUPAC) name | |
| methyl 5-amino-4-oxo-pentanoate | |
| Identifiers | |
| CAS number | |
| ATC code | L01 |
| PubChem | |
| DrugBank | |
| Chemical data | |
| Formula | Template:OrganicBox atomTemplate:OrganicBox atomTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBox atomTemplate:OrganicBoxTemplate:OrganicBox atomTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBox |
| Mol. mass | 145.156 g/mol |
| SMILES | & |
| Pharmacokinetic data | |
| Bioavailability | ? |
| Metabolism | ? |
| Half life | ? |
| Excretion | ? |
| Therapeutic considerations | |
| Pregnancy cat. |
? |
| Legal status | |
| Routes | ? |
Mechanism of Action
- Photosensitization following application of Metvixia Cream occurs through the metabolic conversion of methyl aminolevulinate (prodrug) to photoactive porphyrins (PAP), which accumulates in the skin lesions to which Metvixia Cream has been applied. When exposed to light of appropriate wavelength and energy, the accumulated photoactive porphyrins produce a photodynamic reaction, resulting in a cytotoxic process dependent upon the simultaneous presence of oxygen. The absorption of light results in an excited state of porphyrin molecules, and subsequent spin transfer from photoactive porphyrins to molecular oxygen generates singlet oxygen, which can further react to form superoxide and hydroxyl radicals. Photosensitization of actinic (solar) keratosis lesions using Metvixia Cream, plus illumination with a CureLight BroadBand Model CureLight 01 (a red light of 570 to 670 nm wavelength) at 75 J/cm2, is the basis for Metvixia photodynamic therapy (PDT).
Structure
Pharmacodynamics
There is limited information regarding Pharmacodynamics of Methyl aminolevulinate in the drug label.
Pharmacokinetics
- The time-course of PAPs after application of Metvixia Cream has been monitored by means of fluorescence. After application of Metvixia Cream to actinic keratosis lesions in 8 patients, fluorescence was measured at several time points over 28 hours. Three hours after the application of Metvixia Cream the fluorescence in the treated lesions was significantly greater than that seen in both treated and untreated normal skin, and after application of vehicle cream (not containing methyl aminolevulinate) to normal skin. After application of Metvixia Cream for 28 hours and subsequent illumination with red light of 570 to 670 nm wavelength at a total light dose of 75 J/cm2, complete photobleaching (photodegradation) of Protoporphyrin IX occurred with levels of Protoporphyrin IX returning to pre-treatment values within 1 hour after illumination. However, the fate of other photoactive porphyrins are unknown.
- The clinical dose of methyl aminolevulinate cream and duration of application were derived from a study in which three different strengths of the cream (16, 80 and 160 mg/g methyl aminolevulinate, as hydrochloride), each applied for 3 hours or 18 hours, were tested in 16 patients.
Nonclinical Toxicology
There is limited information regarding Nonclinical Toxicology of Methyl aminolevulinate in the drug label.
Clinical Studies
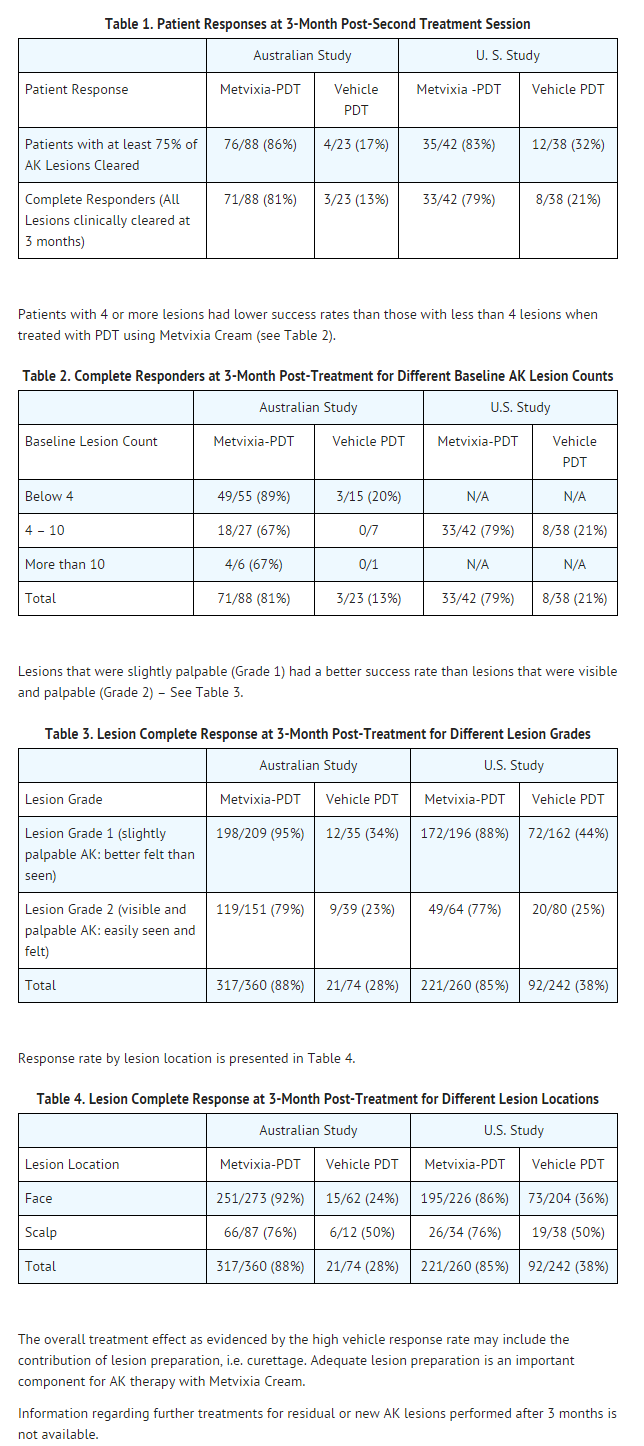
- Metvixia Cream plus illumination with the CureLight BroadBand Model CureLight 01 (a red light of 570 to 670 nm wavelength) at 75 J/cm2 has been studied in 130 patients with non-hyperkeratotic actinic keratoses in two clinical trials. These trials were not identical; however, both were randomized, multicenter, and double-blinded with patients randomized to Metvixia-PDT and Vehicle-PDT study arms that required two treatment sessions (7 days apart). One study was conducted in the U.S. and patients were randomized 1:1 Metvixia to Vehicle and one study was conducted in Australia with patients randomized 4:1 Metvixia to Vehicle. In both studies treatment consisted of a multi-step process that was repeated after 7 days consisting of 1) Lesion preparation (debridement with sharp curette) to roughen the surface of the lesion. 2) Metvixia or Vehicle Cream application to lesions with occlusion with an adhesive, non-absorbent dressing, 3) Waiting at least 2.5 hours, but no more than 4 hours to allow for conversion of the methyl aminolevulinate, 4) Removal of cream with gauze and saline, 5) Red light Dosimetry and Illumination with the CureLight BroadBand Model CureLight 01 (a red light of 570 to 670 nm wavelength).
- Study patients had previously untreated facial and scalp actinic keratoses (AKs) that were slightly palpable (better felt than seen). Hyperkeratotic actinic keratoses were excluded. In the U.S. study 100% of patients had 4 to 10 lesions at baseline. However, in the Australian study, 63% (70/111) of patients had less than 4 lesions at baseline, 31% (34/111) of the enrolled patients had 4 to 10 lesions, and 6% (7/111) had more than 10 lesions at baseline (a maximum of 6 treatment fields were allowed in this study).
- A “Cleared” AK lesion was defined as being not visible and not palpable as assessed 3 months after the second treatment session. Patients with all treated lesions cleared at 3 months were defined as Complete Responders.
- The percentage of patients in whom 75% or more of the treated lesions were clear and the percent of patients in whom 100% of the treated lesions were clear 3 months after the second Metvixia-PDT treatment session are shown in Table 1 for each of the two studies.
How Supplied
- Metvixia Cream, 16.8%, is available as the following:
- NDC 63069-401-01, 2 gram aluminum tube, box of 1
- Product Package
- Keep out of reach of children
Storage
- Store refrigerated, 2-8°C (36-46°F).
- Use contents within one week after opening.
- Should not be used after 24 hours out of refrigerator.
Images
Drug Images
{{#ask: Page Name::Methyl aminolevulinate |?Pill Name |?Drug Name |?Pill Ingred |?Pill Imprint |?Pill Dosage |?Pill Color |?Pill Shape |?Pill Size (mm) |?Pill Scoring |?NDC |?Drug Author |format=template |template=DrugPageImages |mainlabel=- |sort=Pill Name }}
Package and Label Display Panel
{{#ask: Label Page::Methyl aminolevulinate |?Label Name |format=template |template=DrugLabelImages |mainlabel=- |sort=Label Page }}
Patient Counseling Information
PATIENT INFORMATION
- Metvixia™ Cream 16.8% (phonetic)
- Generic name: methyl aminolevulinate hydrochloride
- Read this Patient Information before you get treated with Metvixia Cream and each time you get a treatment. There may be new information. This leaflet does not take the place of talking with your doctor about your condition or treatment. Ask your healthcare provider about anything you do not understand about Metvixia Cream.
- What is the most important thing I need to know about Metvixia Cream?
- Metvixia Cream with light treatment (Photodynamic therapy or PDT) is only done in medical offices by trained doctors.
- Metvixia Cream is not applied by patients. Metvixia Cream should not be applied by doctors who have not been trained in its use.
- What is Metvixia Cream?
- Metvixia Cream is a prescription cream used with PDT (light treatment) to treat skin growths on the face and scalp called actinic keratosis (AK).
- Metvixia Cream is only used for AK skin growths that are thin and not dark colored. AK skin growths are not cancer. AK skin growths are caused partly by too much sun exposure. Metvixia Cream and PDT work together to treat AK skin growths.
- Metvixia Cream has not been studied in children for any condition and should not be used in children.
- Who should not use Metvixia Cream?
- Do not use Metvixia Cream if:
- your skin over reacts to sun or light (photosensitivity)
- you are allergic to porphyrins or to any of the ingredients in Metvixia Cream. The active ingredient is methyl aminolevulinate hydrochloride. Metvixia Cream also contains peanut and almond oil. See the end of this leaflet for a complete list of INGREDIENTS in Metvixia Cream.
- Before treatment with Metvixia Cream, tell your doctor:
- about your medical conditions, including if you
- are pregnant or planning to become pregnant. It is not known if Metvixia Cream can harm your unborn baby.
- Are breastfeeding. It is not known if Metvixia Cream passes into your milk and if it can harm your baby. You should decide whether or not to stop breastfeeding while getting treatment with Metvixia Cream. Talk to your doctor for help with this choice.
- Are allergic to nuts or peanuts
- Have or had skin cancer or other skin growths on your body
- Have bleeding problems.
- About all the medicines you take, including prescription and non-prescription medicines, vitamins and herbal supplements. It is not known if Metvixia Cream and other medicines can affect each other.
- How should I use Metvixia Cream?
- Metvixia Cream and PDT treatment is only done by trained doctors.
- You will receive 2 treatments with Metvixia Cream and PDT 7 days (1 week) apart. Your doctor will check you three months after treatment to see if the treatment worked for you.
- Metvixia Cream is for skin use only. Do not get Metvixia Cream in your eyes, mouth, or nose. Tell your doctor right away if this happens.
- What should I avoid while using Metvixia Cream?
- During the 3 hours that Metvixia Cream is on your skin:
- Avoid exposure to sunlight or bright indoor light during the 3 hours that Metvixia Cream is on your skin. Wear a protective hat and clothing if you need to be outside in the sun.
- Avoid exposure to cold temperatures during the 3 hours that Metvixia Cream is on your skin. Wear warm clothing and keep your treated skin site covered * * If you are in cold temperatures.
- If for some unavoidable reason you are not treated with the lamp you should.
- Carefully rinse off the Cream.
- Avoid exposure to sunlight, prolonged or intense light for two days after treatment.
- What are the possible side effects of Metvixia Cream with PDT treatment?
- Common side effects of Metvixia Cream with PDT treatment include the following skin reactions at the treated site:
- Tell your doctor if you get any of these side effects. Your healthcare provider should be able to treat these reactions according to standard treatments for such skin reactions. These reactions usually go away within 10 days of treatment. Redness may last for up to 1 month. If any of your skin reactions get worse and last longer than 3 weeks, call your doctor.
- These are not all the side effects of Metvixia Cream with PDT. Ask your doctor or pharmacist for more information.
General information about Metvixia Cream
- Medicines are sometimes prescribed for conditions that are not mentioned in patient information leaflets.
- This leaflet summarizes the most important information about Metvixia Cream. If you would like more information, talk with your doctor. You can ask your doctor for information about Metvixia Cream that is written for health professionals.
- Toll-free number and/or website will be provided when available for the US market.
- What are the ingredients in Metvixia Cream?
- Active Ingredient: methyl aminolevulinate hydrochloride
- Other Ingredients: Glyceryl monostearate, cetostearyl alcohol, poloxyl stearate, cholesterol, oleyl alcohol glycerin, white petrolatum, isopropyl myristate, refined peanut oil, refined almond oil, edetate disodium, methylparaben and propylparaben. The color of the product is cream to pale yellow.
Precautions with Alcohol
- Alcohol-Methyl aminolevulinate interaction has not been established. Talk to your doctor about the effects of taking alcohol with this medication.
Brand Names
- METVIXIA®[1]
Look-Alike Drug Names
There is limited information regarding Methyl aminolevulinate Look-Alike Drug Names in the drug label.
Price
References
The contents of this FDA label are provided by the National Library of Medicine.
{{#subobject:
|Label Page=Methyl aminolevulinate |Label Name=Methyl 05.png
}}