Alzheimer's disease pathophysiology
| Title |
| https://https://www.youtube.com/watch?v=v5gdH_Hydes%7C350}} |
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]; Associate Editor(s)-in-Chief: Syed Hassan A. Kazmi BSc, MD [2], Aravind Reddy Kothagadi M.B.B.S[3]
|
Alzheimer's disease Microchapters |
|
Diagnosis |
|---|
|
Treatment |
|
Alzheimer's disease pathophysiology On the Web |
|
American Roentgen Ray Society Images of Alzheimer's disease pathophysiology |
|
Risk calculators and risk factors for Alzheimer's disease pathophysiology |
Overview
Alzheimer disease (AD), is a progressive neurodegenerative disorder. The dysfunction of amyloid precursor protien (APP) metabolism and the resulting build up of of Aβ peptides and their aggregation in the form of senile plaques in the brain parenchyma of individuals have been considered pivotal for neurodegeneration in the disease. Cognitive impairment in patients with AD is closely associated with synaptic loss in the neocortex and limbic system. In familial forms of AD, mutations result in an increased Aβ production or aggregation, in sporadic AD, failure of the clearance mechanisms might play a key role. Loss of mature neurons and alterations in neural progenitor cells (NPCs) in areas such as the dentate gyrus (DG) of the hippocampus have been found to be responsible for manifestations of AD. On gross pathology, temporal atrophy (hippocampus in particular), dilation of lateral ventricles and third ventricle are characteristic findings of Alzheimer's disease. The microscopic histopathological features of alzheimer's disease consist neurofibrillary tangles, senile plaques, neuronal loss, and with or without cerebral amyloid angiopathy.
Pathophysiology
Alzheimer disease (AD), is a progressive neurodegenerative disorder. The dysfunction of amyloid precursor protien (APP) metabolism and the resulting build up of of Aβ peptides and their aggregation in the form of senile plaques in the brain parenchyma of individuals have been considered pivotal for neurodegeneration in the disease. There is also an accumulation of intracellular neurofibrillary tangles that consist of hyperphosphorylated tau protein and a profound loss of basal forebrain cholinergic neurons that innervate the hippocampus, and the neocortex.
Triggers
The following factors lead to the development of Alzheimer's dementia:
- Genetic factors
- Environmental factors
- Chromosomal factors
Pathogenesis
The pathogenesis of Alzheimer's dementia (AD) can be explained by four pathological processes. The processes involved in the development of AD and their molecular basis is as follows:[1][2]
(i) Neuronal loss
- Neurogenesis is a complex process characterized by several progressive steps, including neural progenitor cell (NPC) proliferation, migration, differentiation (cell fate commitment) and maturation, including growth and synapse formation
- Initial synaptic injury is followed by neuronal loss accompanied by astrogliosis and microglial cell proliferation.[3][3]
- Cognitive impairment in patients with AD is closely associated with synaptic loss in the neocortex and limbic system[4][5]
- Increase in neurogenesis in the brains of AD patients may be related to glial and vasculature-associated changes as suggested by an increase in neurogenic markers[6]
- Loss of mature neurons and alterations in neural progenitor cells (NPCs) in areas such as the dentate gyrus (DG) of the hippocampus have been found to be responsible for manifestations of AD
(ii) Aggregation of extra-cellular amyloid β (Aβ)
- Amyloid precursor protein (APP) is physiologically present in normal brains
- It is proteolytically processed by α-, β-, and γ-secretases[7][8]
- In familial forms of AD, mutations result in an increased Aβ production or aggregation, in sporadic AD, failure of the clearance mechanisms might play a key role
- Aβ oligomers are responsible for the synapto-toxic effects of Aβ[9]
Constitutive (nonamyloidogenic) pathway
- In the constitutive pathway, proteolysis of APP by α- and γ-secretases results in nonpathogenic fragments (sAPPα and α-C-terminal fragment)
Amyloidogenic pathway
- In the amyloidogenic pathway, proteolysis of APP by β-secretase and γ-secretase gives rise to a mixture of Aβ peptides with different lengths. There are two major Aβ species: Aβ1–40 (90%) and Aβ1–42 (10%). The Aβ1–42 fragments are more aggregation-prone and are predominantly present in amyloid plaques in brains of AD patients.[10]
- Abnormal accumulation of Aβ is the result of an imbalance between the levels of Aβ production, aggregation and clearance.
- Aβ clearance is mediated by proteolytic enzymes such as neprilysin, chaperone molecules such as apoE, lysosomal (e.g. autophagy) and non-lysosomal pathways (e.g. proteasome)[11]
- Nerve damage as described under 'neuronal loss', might result from the conversion of normally non-toxic monomers to toxic oligomers of Aβ peptides[9][12][13]
- Changes in glutamate receptors and increased excitability; mitochondrial dysfunction; lysosomal failure and alterations in signaling pathways related to synaptic plasticity, neuronal cell death and neurogenesis have been proposed as the molecular mechanisms leading to the development of Alzeimer's dementia (AD)[14][15][16]
(iii) CDK5 pathway
- CDK5 is the predominant CDK found in the brain, is expressed heavily in neurons and plays a key part in synaptic integrity and neuronal development
- Increased activation of CDK5/p35/p25 has been linked to the pathogenesis of neurodegenerative diseases such as AD
- CDK5 may mediate changes in neurogenesis in AD via aberrant phosphorylation of CDK5 substrates, which include cytoskeletal (neurofilaments, nestin), synaptic proteins (synapsin)[17]
(iv) Formation of intraneuronal neurofibrillary tangles (tau protein accumulation)
- Aβ is involved in tau deposition in AD pathogenesis and leads to the conversion of tau from a normal to a toxic state, but there is also evidence that toxic tau increases Aβ toxicity via a positive feedback loop
- A protein that functionally links Aβ to tau is fyn. This cytosolic tyrosine kinase positively regulates N-methyl-D-aspartate (NMDA) receptor activity and has been shown to be targeted to postsynaptic sites in dendrites by tau, which binds fyn[18]
- In response to Aβ, tau is relocated from axons and dendrites into the somatodendritic compartment[19]
- Excess fyn accompanies the excess tau in AD dendrites and upregulates NMDA receptor activity in those areas, causing an increased calcium influx. This calcium-driven excitotoxicity can damage postsynaptic sites and cause neurodegeneration
Genetics
Common genes
Early onset (Alzheimer's dementia-AD 1, 3 and 4)
30-50 percent of early-onset Alzheimer's dementia (AD) is associated with an autosomal dominant inheritance and consists of mutations in the following genes:[21][22]
- Presenilin1 (PS1) gene, also called PSEN1 gene on chromosome 14 (AD3- 20 to 30 percent cases)
- Presenilin 2 (PS2) gene, also called PSEN2 gene on chromosome 1 (AD4- rare)
- Point mutations in amyloid beta A4 protein gene, also called amyloid precursor protein (APP) gene on chromosome 21 are associated in some cases of early onset (< 65 yr) familial AD cases
Late onset (Alzheimer's dementia -AD2)
- Apolipoprotein 4 gene (APOE4) mutation is associated with late onset (>60 years) Alzheimer's dementia (AD)[23]
- p.Arg47His allelic variant in TREM2 gene[24]
Less common genes
Less common genes associated with the development of AD are:
- A2M on chromosome 12
- ABCA7; when suppressed, results in an elevation of amyloid production
- AKAP9, a kinase anchor protein 9 (PRKA) that regulates NMDA channel activity
- There is evidence both for and against ADAM10
- BIN1, a tumor suppressor protein
- CALHM1 on chromosome 10q24; CALHM1 influences calcium homeostchaperon has a single nucleotide polymorphism (SNP) associated with late-onset AD
- CD2AP, an adaptor molecule involved in dynamic actin remodeling and membrane trafficking
- A SNP in CD33
- Clusterin (CLU, APOJ), a molecular chaperon present in senile plaques that has CR1 and PICALM, implicated in two genome-wide association studies (GWAS)
- Dysferlin (encoded by DYSF), associated with several limb-girdle muscular dystrophies; accumulates in Alzheimer patients
- EPHA1 (encoding a protein that belongs to the ephrin receptor subfamily); plays part in synaptic plasticity
- GAB2 on chromosome 11q14 interacting with the APOE e4 allele
- GST01 and GST02 on chromosome 10
- PAX1P1, which encodes for a nuclear protein that may function in DNA repair pathways
- PLD3 on chromosome 19q13.2
- SORL1 on chromosome 11q23, a protein involved with APP protein trafficking
- TOMM40, located on chromosome 19q very close to the APOE locus,TOMM40 has been implicated in late-onset AD both by linkage analysis and by the presence of a variable length poly-T repeat within the gene
- UNC5C is enriched in neurons of the hippocampal pyramidal layer
- In a large GWAS meta-analysis, the following genes have been identified as rare causes of Alzheimer's disease:
- Several other potential loci under investigation on the following chromosomes:
- Chromosome 12
- Chromosome 10
- Chromosome 2q
- Chromosome 9p
- Chromosome 15q
- Chromosome 19p13
- Chromosome 7q36
- Chromosome 9q22 (UBQLN1)
- Chromosome 1q22
- Chromosome 3q23
- Chromosome 10q22
- Chromosome 11q25
Associated Conditions
- Cerebral amyloid angiopathy
- Down's Syndrome
- Reccurent respiratory infections (pneumonia)
Gross Pathology
- On gross pathology, temporal atrophy (hippocampus in particular), dilation of lateral ventricles and third ventricle are characteristic findings of Alzheimer's disease.

Microscopic Pathology
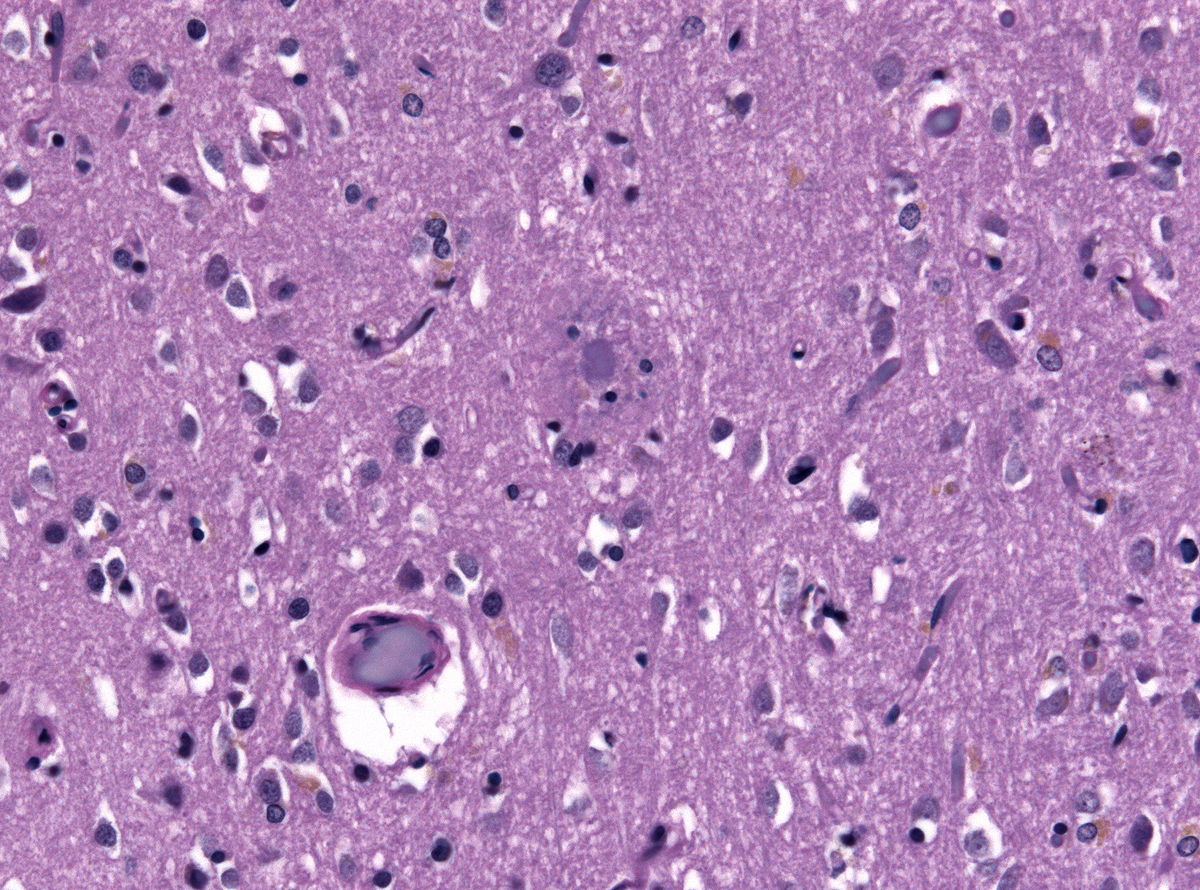
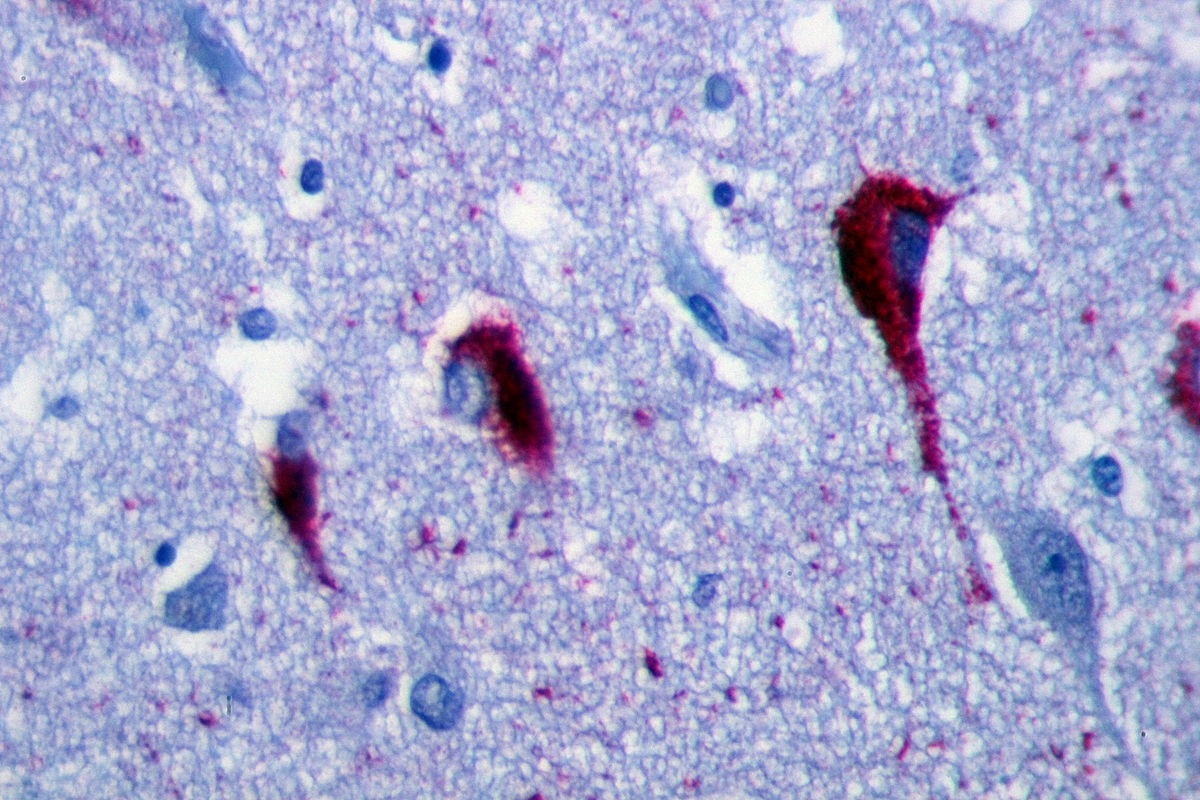
- The microscopic histopathological features of alzheimer's disease represent neurofibrillary tangles, senile plaques, neuronal loss, and with or without cerebral amyloid angiopathy:
- Neurofibrillary tangles: Consists of tau, location in the hippocampus, cerebral cortex, hypothalamus. Dementia severity correlates better with neurofibrillary tangles number rather than senile plaque number
- Senile plaques or the neuritic plaques consists of two components which are A-beta amyloid which radiate from the center and the neurites with swollen axons. Senile plaques are considered to be more specific for alzheimer's than neurofibrillary tangles
- Loss of neurons
- With or without cerebral amyloid angiopathy

References
- ↑ Crews L, Masliah E (2010). "Molecular mechanisms of neurodegeneration in Alzheimer's disease". Hum. Mol. Genet. 19 (R1): R12–20. doi:10.1093/hmg/ddq160. PMC 2875049. PMID 20413653.
- ↑ Weller J, Budson A (2018). "Current understanding of Alzheimer's disease diagnosis and treatment". F1000Res. 7. doi:10.12688/f1000research.14506.1. PMC 6073093. PMID 30135715.
- ↑ 3.0 3.1 Beach TG, Walker R, McGeer EG (1989). "Patterns of gliosis in Alzheimer's disease and aging cerebrum". Glia. 2 (6): 420–36. doi:10.1002/glia.440020605. PMID 2531723.
- ↑ DeKosky ST, Scheff SW (1990). "Synapse loss in frontal cortex biopsies in Alzheimer's disease: correlation with cognitive severity". Ann. Neurol. 27 (5): 457–64. doi:10.1002/ana.410270502. PMID 2360787.
- ↑ Terry RD, Masliah E, Salmon DP, Butters N, DeTeresa R, Hill R, Hansen LA, Katzman R (1991). "Physical basis of cognitive alterations in Alzheimer's disease: synapse loss is the major correlate of cognitive impairment". Ann. Neurol. 30 (4): 572–80. doi:10.1002/ana.410300410. PMID 1789684.
- ↑ Boekhoorn K, Joels M, Lucassen PJ (2006). "Increased proliferation reflects glial and vascular-associated changes, but not neurogenesis in the presenile Alzheimer hippocampus". Neurobiol. Dis. 24 (1): 1–14. doi:10.1016/j.nbd.2006.04.017. PMID 16814555.
- ↑ Selkoe DJ (1989). "Amyloid beta protein precursor and the pathogenesis of Alzheimer's disease". Cell. 58 (4): 611–2. PMID 2504495.
- ↑ Tanzi RE, Gusella JF, Watkins PC, Bruns GA, St George-Hyslop P, Van Keuren ML, Patterson D, Pagan S, Kurnit DM, Neve RL (1987). "Amyloid beta protein gene: cDNA, mRNA distribution, and genetic linkage near the Alzheimer locus". Science. 235 (4791): 880–4. PMID 2949367.
- ↑ 9.0 9.1 Walsh DM, Selkoe DJ (2004). "Oligomers on the brain: the emerging role of soluble protein aggregates in neurodegeneration". Protein Pept. Lett. 11 (3): 213–28. PMID 15182223.
- ↑ Van Cauwenberghe C, Van Broeckhoven C, Sleegers K (2016). "The genetic landscape of Alzheimer disease: clinical implications and perspectives". Genet. Med. 18 (5): 421–30. doi:10.1038/gim.2015.117. PMC 4857183. PMID 26312828.
- ↑ Bendiske J, Bahr BA (2003). "Lysosomal activation is a compensatory response against protein accumulation and associated synaptopathogenesis--an approach for slowing Alzheimer disease?". J. Neuropathol. Exp. Neurol. 62 (5): 451–63. PMID 12769185.
- ↑ Volles MJ, Lansbury PT (2002). "Vesicle permeabilization by protofibrillar alpha-synuclein is sensitive to Parkinson's disease-linked mutations and occurs by a pore-like mechanism". Biochemistry. 41 (14): 4595–602. PMID 11926821.
- ↑ Selkoe DJ (1999). "Translating cell biology into therapeutic advances in Alzheimer's disease". Nature. 399 (6738 Suppl): A23–31. PMID 10392577.
- ↑ Lin H, Bhatia R, Lal R (2001). "Amyloid beta protein forms ion channels: implications for Alzheimer's disease pathophysiology". FASEB J. 15 (13): 2433–44. doi:10.1096/fj.01-0377com. PMID 11689468.
- ↑ Nakamura T, Lipton SA (2010). "Redox regulation of mitochondrial fission, protein misfolding, synaptic damage, and neuronal cell death: potential implications for Alzheimer's and Parkinson's diseases". Apoptosis. 15 (11): 1354–63. doi:10.1007/s10495-010-0476-x. PMC 2978885. PMID 20177970.
- ↑ Nixon RA, Cataldo AM (2006). "Lysosomal system pathways: genes to neurodegeneration in Alzheimer's disease". J. Alzheimers Dis. 9 (3 Suppl): 277–89. PMID 16914867.
- ↑ Matsubara M, Kusubata M, Ishiguro K, Uchida T, Titani K, Taniguchi H (1996). "Site-specific phosphorylation of synapsin I by mitogen-activated protein kinase and Cdk5 and its effects on physiological functions". J. Biol. Chem. 271 (35): 21108–13. PMID 8702879.
- ↑ Ittner LM, Ke YD, Delerue F, Bi M, Gladbach A, van Eersel J, Wölfing H, Chieng BC, Christie MJ, Napier IA, Eckert A, Staufenbiel M, Hardeman E, Götz J (2010). "Dendritic function of tau mediates amyloid-beta toxicity in Alzheimer's disease mouse models". Cell. 142 (3): 387–97. doi:10.1016/j.cell.2010.06.036. PMID 20655099.
- ↑ Delacourte A, Flament S, Dibe EM, Hublau P, Sablonnière B, Hémon B, Shérrer V, Défossez A (1990). "Pathological proteins Tau 64 and 69 are specifically expressed in the somatodendritic domain of the degenerating cortical neurons during Alzheimer's disease. Demonstration with a panel of antibodies against Tau proteins". Acta Neuropathol. 80 (2): 111–7. PMID 2117840.
- ↑ "Alzheimer Disease Overview - GeneReviews® - NCBI Bookshelf".
- ↑ Campion D, Dumanchin C, Hannequin D, Dubois B, Belliard S, Puel M, Thomas-Anterion C, Michon A, Martin C, Charbonnier F, Raux G, Camuzat A, Penet C, Mesnage V, Martinez M, Clerget-Darpoux F, Brice A, Frebourg T (1999). "Early-onset autosomal dominant Alzheimer disease: prevalence, genetic heterogeneity, and mutation spectrum". Am. J. Hum. Genet. 65 (3): 664–70. doi:10.1086/302553. PMC 1377972. PMID 10441572.
- ↑ Tsuang D, Larson EB, Bowen J, McCormick W, Teri L, Nochlin D, Leverenz JB, Peskind ER, Lim A, Raskind MA, Thompson ML, Mirra SS, Gearing M, Schellenberg GD, Kukull W (1999). "The utility of apolipoprotein E genotyping in the diagnosis of Alzheimer disease in a community-based case series". Arch. Neurol. 56 (12): 1489–95. PMID 10593304.
- ↑ Khachaturian AS, Corcoran CD, Mayer LS, Zandi PP, Breitner JC (2004). "Apolipoprotein E epsilon4 count affects age at onset of Alzheimer disease, but not lifetime susceptibility: The Cache County Study". Arch. Gen. Psychiatry. 61 (5): 518–24. doi:10.1001/archpsyc.61.5.518. PMID 15123497.
- ↑ Jonsson T, Stefansson H, Steinberg S, Jonsdottir I, Jonsson PV, Snaedal J, Bjornsson S, Huttenlocher J, Levey AI, Lah JJ, Rujescu D, Hampel H, Giegling I, Andreassen OA, Engedal K, Ulstein I, Djurovic S, Ibrahim-Verbaas C, Hofman A, Ikram MA, van Duijn CM, Thorsteinsdottir U, Kong A, Stefansson K (2013). "Variant of TREM2 associated with the risk of Alzheimer's disease". N. Engl. J. Med. 368 (2): 107–16. doi:10.1056/NEJMoa1211103. PMC 3677583. PMID 23150908.