Salmonella
|
Salmonellosis Microchapters |
|
Diagnosis |
|---|
|
Treatment |
|
Case Studies |
|
Salmonella On the Web |
|
American Roentgen Ray Society Images of Salmonella |
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]; Associate Editor(s)-in-Chief: João André Alves Silva, M.D. [2] Jolanta Marszalek, M.D. [3]
Overview
Salmonella is a rod-shaped, facultative intracellular, gram-negative enterobacteria.[1] Salmonella species shows motility, produces hydrogen sulfide, and only 1% is able to ferment lactose.[2] It may be isolated from the stool of infected patients, and grown in culture media, such as MacConkey agar and deoxycholate agar. Salmonella enters the body through contaminated food or water, and invades the intestinal epithelial cells, causing inflammation.
This bacterium may be classified into 2 different species: Salmonella enterica and Salmonella bongori. Salmonella enterica is divided in 6 different subspecies, of which I, contains most pathogenic serotypes for humans. Salmonella may be serogrouped into more than 2500 serovars with polyvalent antisera, according to the capsular antigen, polysaccharide O antigens, and flagellar antigens. The bacteria show tropism for the epithelial cells of the gastrointestinal tract, macrophages, dendritic cells, and neutrophils. Different serotypes of Salmonella may have different natural reservoirs, some have humans as their only natural reservoir (serotypes Sendai and Typhi), while others (serotype Dublin) may infect humans and cattle.
Taxonomy
Cellular organism; Bacteria; Proteobacteria; Gammaproteobacteria; Enterobacteriales; Enterobacteriaceae[3]
Biology
 |
 |
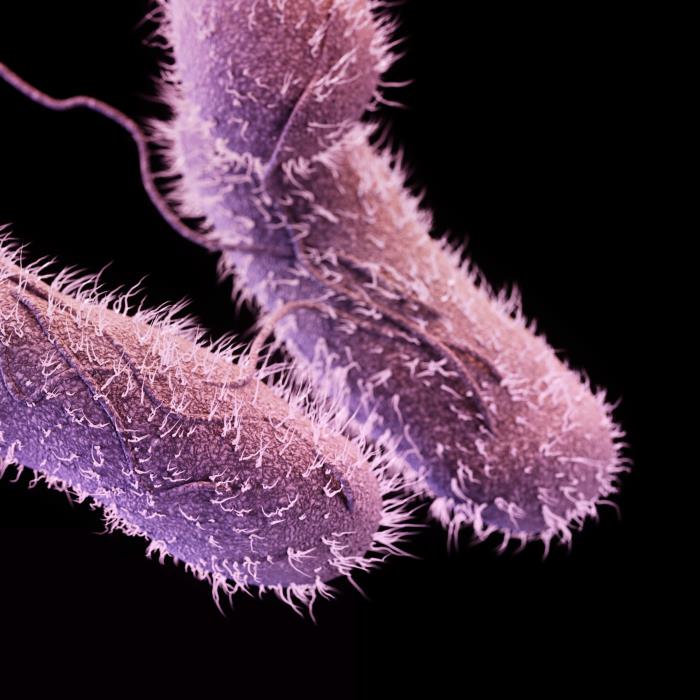
Salmonella is a gram-negative, facultative intracellular, anaerobic, non-spore-forming bacillus. It measures 2 to 3 by 0.4 to 0.6 μm. Salmonella is a non-lactose fermenting bacterium. It reduces nitrates, produces acid on glucose fermentation and does not produce cytochrome oxidase.[5] Due to the presence of flagella, almost all salmonella species are motile. 1% of the bacteria is able to ferment lactose, which may be responsible for its non-detection in some culture media.
For the isolation of salmonella in culture, freshly passed stool are preferred. Common media for the growth of salmonella include: MacConkey agar, deoxycholate agar, and xylose-lysine-deoxycholate agar.[6]
When the sample has a low number of bacteria, special enrichment broths, such as the selenite-based enrichment broth, may be used to raise the number of bacteria.[6]
Infectious Cycle
Salmonella enterica enters the body through the mouth, by ingestion of contaminated food and water. For the bacteria to cause disease, an inoculum of about 50 000 pathogens is often required. Once in the intestine, the bacteria will first infect the apical epithelium. Salmonella will then initiate bacterial mechanisms that allow invasion of the host cells, inducing inflammatory changes, such as:[7][8][9][10]
- Diffuse and focal infiltration of PMN
- Crypt abscesses
- Necrosis of the epithelium
- Fluid secretion
- Edema
Different serovars will have different preferable intestinal locations. An example is the enterocolitis at the terminal ileum, cecum, and proximal colon caused by serovar Typhimurium. Intestinal disease is marked by neutrophil migration to the intestinal epithelium. This recruitment is done by the secretion of interleukin-8, induced by Salmonella.[11]
Classification
Before 1983 Salmonella was classified in several species. However, its genome study has shown high levels of DNA similarities among different types of salmonella, which leads to the actual classification of salmonella in 2 different species:
Salmonella enterica
- Contains six subspecies - I, II, IIIa, IIIb, IV, and VI:[12][13]
- I - enterica
- II - salamae
- III - arizonae
- IIIb - diarizonae
- IV - houtenae
- VI - indica
- Subspecies I contains most pathogenic serotypes for humans
- Subspecies IIIa and IIIb, formerly belonging to the genus Arizonae, are responsible for rare human infections
Salmonella bongori
- Formerly subspecies V[14]
Serovars
Salmonella subspecies may be serogrouped into more than 2500 serovars with polyvalent antisera. For this division the following bacterial structures are considered:[15]
- Capsular antigen
- Polysaccharide O antigens
- Flagellar antigens
Different salmonella serotypes may also be distinguished in culture, according to their different metabolism of sugars.[16] Different serovars may also have different disease manifestations. For example, Salmonella enterica, serovar Typhimurium is the causative agent of typhoid fever, and is not associated with classical salmonellosis.
Tropism
In vitro, Salmonella is able to interact with different types of cells. However, in vivo, the bacteria was found to enter only certain human cells, namely:[17]
- Epithelial cells of the gastrointestinal tract - first cells to be infected by Salmonella
- Macrophages - where Salmonella survives and replicates
- Dendritic cells - relevant for dissemination of bacteria in the initial stages of infection, yet, they are not considered adequate reservoirs for Salmonella at latter stages
- Neutrophils - interaction is based on the immune response against the bacteria, hence this might not be considered true tropism
Differential diagnosis
Salmonella gastroenteritis must be differentiated from other causes of viral, bacterial, and parasitic gastroentritis.
| Organism | Age predilection | Travel History | Incubation Size (cell) | Incubation Time | History and Symptoms | Diarrhea type8 | Food source | Specific consideration | |||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Fever | N/V | Cramping Abd Pain | Small Bowel | Large Bowel | Inflammatory | Non-inflammatory | |||||||||
| Viral | Rotavirus | <2 y | - | <102 | <48 h | + | + | - | + | + | - | Mostly in day cares, most common in winter. | |||
| Norovirus | Any age | - | 10 -103 | 24-48 h | + | + | + | + | + | - | Most common cause of gastroenteritis, abdominal tenderness, | ||||
| Adenovirus | <2 y | - | 105 -106 | 8-10 d | + | + | + | + | + | - | No seasonality | ||||
| Astrovirus | <5 y | - | 72-96 h | + | + | + | + | + | Seafood | Mostly during winter | |||||
| Bacterial | Escherichia coli | ETEC | Any age | + | 108 -1010 | 24 h | - | + | + | + | + | - | Causes travelers diarrhea, contains heat-labile toxins (LT) and heat-stable toxins (ST) | ||
| EPEC | <1 y | - | 10† | 6-12 h | - | + | + | + | + | Raw beef and chicken | - | ||||
| EIEC | Any ages | - | 10† | 24 h | + | + | + | + | + | Hamburger meat and unpasteurized milk | Similar to shigellosis, can cause bloody diarrhea | ||||
| EHEC | Any ages | - | 10 | 3-4 d | - | + | + | + | + | Undercooked or raw hamburger (ground beef) | Known as E. coli O157:H7, can cause HUS/TTP. | ||||
| EAEC | Any ages | + | 1010 | 8-18 h | - | - | + | + | + | - | May cause prolonged or persistent diarrhea in children | ||||
| Salmonella sp. | Any ages | + | 1 | 6 to 72 h | + | + | + | + | + | Meats, poultry, eggs, milk and dairy products, fish, shrimp, spices, yeast, coconut, sauces, freshly prepared salad. | Can cause salmonellosis or typhoid fever. | ||||
| Shigella sp. | Any ages | - | 10 - 200 | 8-48 h | + | + | + | + | + | Raw foods, for example, lettuce, salads (potato, tuna, shrimp, macaroni, and chicken) | Some strains produce enterotoxin and Shiga toxin similar to those produced by E. coli O157:H7 | ||||
| Campylobacter sp. | <5 y, 15-29 y | - | 104 | 2-5 d | + | + | + | + | + | Undercooked poultry products, unpasteurized milk and cheeses made from unpasteurized milk, vegetables, seafood and contaminated water. | May cause bacteremia, Guillain-Barré syndrome (GBS), hemolytic uremic syndrome (HUS) and recurrent colitis | ||||
| Yersinia enterocolitica | <10 y | - | 104 -106 | 1-11 d | + | + | + | + | + | Meats (pork, beef, lamb, etc.), oysters, fish, crabs, and raw milk. | May cause reactive arthritis; glomerulonephritis; endocarditis; erythema nodosum.
can mimic appendicitis and mesenteric lymphadenitis. | ||||
| Clostridium perfringens | Any ages | > 106 | 16 h | - | - | + | + | + | Meats (especially beef and poultry), meat-containing products (e.g., gravies and stews), and Mexican foods. | Can survive high heat, | |||||
| Vibrio cholerae | Any ages | - | 106-1010 | 24-48 h | - | + | + | + | + | Seafoods, including molluscan shellfish (oysters, mussels, and clams), crab, lobster, shrimp, squid, and finfish. | Hypotension, tachycardia, decreased skin turgor. Rice-water stools | ||||
| Parasites | Protozoa | Giardia lamblia | 2-5 y | + | 1 cyst | 1-2 we | - | - | + | + | + | Contaminated water | May cause malabsorption syndrome and severe weight loss | ||
| Entamoeba histolytica | 4-11 y | + | <10 cysts | 2-4 we | - | + | + | + | + | Contaminated water and raw foods | May cause intestinal amebiasis and amebic liver abscess | ||||
| Cryptosporidium parvum | Any ages | - | 10-100 oocysts | 7-10 d | + | + | + | + | + | Juices and milk | May cause copious diarrhea and dehydration in patients with AIDS especially with 180 > CD4 | ||||
| Cyclospora cayetanensis | Any ages | + | 10-100 oocysts | 7-10 d | - | + | + | + | + | Fresh produce, such as raspberries, basil, and several varieties of lettuce. | More common in rainy areas | ||||
| Helminths | Trichinella spp | Any ages | - | Two viable larvae (male and female) | 1-4 we | - | + | + | + | + | Undercooked meats | More common in hunters or people who eat traditionally uncooked meats | |||
| Taenia spp | Any ages | - | 1 larva or egg | 2-4 m | - | + | + | + | + | Undercooked beef and pork | Neurocysticercosis: Cysts located in the brain may be asymptomatic or seizures, increased intracranial pressure, headache. | ||||
| Diphyllobothrium latum | Any ages | - | 1 larva | 15 d | - | - | - | + | + | Raw or undercooked fish. | May cause vitamin B12 deficiency | ||||
8Small bowel diarrhea: watery, voluminous with less than 5 WBC/high power field
Large bowel diarrhea: Mucousy and/or bloody with less volume and more than 10 WBC/high power field
† It could be as high as 1000 based on patient's immunity system.
The table below summarizes the findings that differentiate inflammatory causes of chronic diarrhea[18][19][20][21][21]
| Cause | History | Laboratory findings | Diagnosis | Treatment |
|---|---|---|---|---|
| Diverticulitis |
|
|
Abdominal CT scan with oral and intravenous (IV) contrast | bowel rest, IV fluid resuscitation, and broad-spectrum antimicrobial therapy which covers anaerobic bacteria and gram-negative rods |
| Ulcerative colitis |
|
|
Endoscopy | Induction of remission with mesalamine and corticosteroids followed by the administration of sulfasalazine and 6-Mercaptopurine depending on the severity of the disease. |
| Entamoeba histolytica |
|
cysts shed with the stool | detects ameba DNA in feces | Amebic dysentery
Luminal amebicides for E. histolytica in the colon:
For amebic liver abscess:
|
Natural Reservoir
Different salmonella serovars may have different natural reservoirs. Common types of serovars of salmonella enterica that infect the human gastrointestinal tract include serovars Sendai, Typhi, and Paratyphi. Humans are their only natural reservoir. Other serotypes, such as serotype Dublin, have cattle as their natural reservoir, but has also the capacity to cause infection in humans.[22][23]
Drug Side Effect
Related Chapters
- 1984 Rajneeshee bioterror attack
- List of foodborne illness outbreaks
- Food Testing Strips
Gallery
-
Salmonella bacteria in tetrathionate enrichment broth stained using direct FA staining technique. From Public Health Image Library (PHIL). [4]
-
Salmonella bacteria in tetrathionate enrichment broth stained using direct FA staining technique. From Public Health Image Library (PHIL). [4]
-
Triple sugar iron agar (TSI) tested for Salmonella (H2S+) and (H2S-); Citrobacter sp. and S. arizonae. From Public Health Image Library (PHIL). [4]
-
Gram-negative bacilli, or rod-shaped Salmonella sp. bacteria. From Public Health Image Library (PHIL). [4]
-
SEM depicts a number of red-colored Salmonella sp. bacteria invading a mustard-colored ruffled immune cell. From Public Health Image Library (PHIL). [4]
References
- ↑ Ryan KJ, Ray CG (editors) (2004). Sherris Medical Microbiology (4th ed. ed.). McGraw Hill. ISBN 0-8385-8529-9.
- ↑ Giannella RA (1996). "Salmonella". In Baron S et al (eds.). Baron's Medical Microbiology (4th ed. ed.). Univ of Texas Medical Branch. ISBN 0-9631172-1-1.
- ↑ "Salmonella (Taxonomy)".
- ↑ 4.0 4.1 4.2 4.3 4.4 4.5 4.6 "Public Health Image Library (PHIL), Centers for Disease Control and Prevention".
- ↑ Mandell, Gerald (2010). Mandell, Douglas, and Bennett's principles and practice of infectious diseases. Philadelphia, PA: Churchill Livingstone/Elsevier. ISBN 0443068399.
- ↑ 6.0 6.1 Perez, J. M.; Cavalli, P.; Roure, C.; Renac, R.; Gille, Y.; Freydiere, A. M. (2003). "Comparison of Four Chromogenic Media and Hektoen Agar for Detection and Presumptive Identification of Salmonella Strains in Human Stools". Journal of Clinical Microbiology. 41 (3): 1130–1134. doi:10.1128/JCM.41.3.1130-1134.2003. ISSN 0095-1137.
- ↑ McGovern VJ, Slavutin LJ (1979). "Pathology of salmonella colitis". Am J Surg Pathol. 3 (6): 483–90. PMID 534385.
- ↑ Giannella RA, Formal SB, Dammin GJ, Collins H (1973). "Pathogenesis of salmonellosis. Studies of fluid secretion, mucosal invasion, and morphologic reaction in the rabbit ileum". J Clin Invest. 52 (2): 441–53. doi:10.1172/JCI107201. PMC 302274. PMID 4630603.
- ↑ Clarke RC, Gyles CL (1987). "Virulence of wild and mutant strains of Salmonella typhimurium in ligated intestinal segments of calves, pigs, and rabbits". Am J Vet Res. 48 (3): 504–10. PMID 3551701.
- ↑ Finlay BB, Heffron F, Falkow S (1989). "Epithelial cell surfaces induce Salmonella proteins required for bacterial adherence and invasion". Science. 243 (4893): 940–3. PMID 2919285.
- ↑ McCormick BA, Colgan SP, Delp-Archer C, Miller SI, Madara JL (1993). "Salmonella typhimurium attachment to human intestinal epithelial monolayers: transcellular signalling to subepithelial neutrophils". J Cell Biol. 123 (4): 895–907. PMC 2200157. PMID 8227148.
- ↑ "The type species of the genus Salmonella Lignieres 1900 is Salmonella enterica (ex Kauffmann and Edwards 1952) Le Minor and Popoff 1987, with the type strain LT2T, and conservation of the epithet enterica in Salmonella enterica over all earlier epithets that may be applied to this species. Opinion 80". Int J Syst Evol Microbiol. 55 (Pt 1): 519–20. 2005. PMID 15653929.
- ↑ Tindall BJ; Grimont PAD, Garrity GM; Euzéby JP (2005). "Nomenclature and taxonomy of the genus Salmonella". Int J Syst Evol Microbiol. 55: 521&ndash, 524. PMID 15653930.
- ↑ Popoff MY, Bockemühl J, Gheesling LL (2004). "Supplement 2002 (no. 46) to the Kauffmann-White scheme". Res Microbiol. 155 (7): 568–70. doi:10.1016/j.resmic.2004.04.005. PMID 15313257.
- ↑ Murray, Patrick (2013). Medical microbiology. Philadelphia: Elsevier/Saunders. ISBN 0323086926.
- ↑ Mandell, Gerald (2010). Mandell, Douglas, and Bennett's principles and practice of infectious diseases. Philadelphia, PA: Churchill Livingstone/Elsevier. ISBN 0443068399.
- ↑ Santos RL, Bäumler AJ (2004). "Cell tropism of Salmonella enterica". Int J Med Microbiol. 294 (4): 225–33. doi:10.1016/j.ijmm.2004.06.029. PMID 15532980.
- ↑ Konvolinka CW (1994). "Acute diverticulitis under age forty". Am J Surg. 167 (6): 562–5. PMID 8209928.
- ↑ Silverberg MS, Satsangi J, Ahmad T, Arnott ID, Bernstein CN, Brant SR; et al. (2005). "Toward an integrated clinical, molecular and serological classification of inflammatory bowel disease: report of a Working Party of the 2005 Montreal World Congress of Gastroenterology". Can J Gastroenterol. 19 Suppl A: 5A–36A. PMID 16151544.
- ↑ Satsangi J, Silverberg MS, Vermeire S, Colombel JF (2006). "The Montreal classification of inflammatory bowel disease: controversies, consensus, and implications". Gut. 55 (6): 749–53. doi:10.1136/gut.2005.082909. PMC 1856208. PMID 16698746.
- ↑ 21.0 21.1 Haque R, Huston CD, Hughes M, Houpt E, Petri WA (2003). "Amebiasis". N Engl J Med. 348 (16): 1565–73. doi:10.1056/NEJMra022710. PMID 12700377.
- ↑ Mandell, Gerald (2010). Mandell, Douglas, and Bennett's principles and practice of infectious diseases. Philadelphia, PA: Churchill Livingstone/Elsevier. ISBN 0443068399.
- ↑ "Salmonella enterica Serotypes and Food Commodities, United States, 1998–2008".
- Pages with reference errors
- CS1 maint: Extra text: authors list
- CS1 maint: Extra text
- CS1 maint: Extra text: editors list
- CS1 maint: Multiple names: authors list
- CS1 maint: PMC format
- CS1 maint: Explicit use of et al.
- Bacterial diseases
- Gram negative bacteria
- Enterobacteria
- Foodborne illnesses
- Zoonoses
- Disease
- Emergency medicine
- Intensive care medicine
- Rat carried diseases
![Salmonella bacteria in tetrathionate enrichment broth stained using direct FA staining technique. From Public Health Image Library (PHIL). [4]](/images/0/02/Enterobacteria64.jpeg)
![Salmonella bacteria in tetrathionate enrichment broth stained using direct FA staining technique. From Public Health Image Library (PHIL). [4]](/images/d/d0/Enterobacteria63.jpeg)
![Triple sugar iron agar (TSI) tested for Salmonella (H2S+) and (H2S-); Citrobacter sp. and S. arizonae. From Public Health Image Library (PHIL). [4]](/images/d/dc/Enterobacteria55.jpeg)
![Gram-negative bacilli, or rod-shaped Salmonella sp. bacteria. From Public Health Image Library (PHIL). [4]](/images/2/28/Enterobacteria38.jpeg)
![SEM depicts a number of red-colored Salmonella sp. bacteria invading a mustard-colored ruffled immune cell. From Public Health Image Library (PHIL). [4]](/images/0/07/Enterobacteria01.jpeg)