Methadone (injection)
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]; Associate Editor(s)-in-Chief: Kiran Singh, M.D. [2]
WikiDoc MAKES NO GUARANTEE OF VALIDITY. WikiDoc is not a professional health care provider, nor is it a suitable replacement for a licensed healthcare provider. WikiDoc is intended to be an educational tool, not a tool for any form of healthcare delivery. The educational content on WikiDoc drug pages is based upon the FDA package insert, National Library of Medicine content and practice guidelines / consensus statements. WikiDoc does not promote the administration of any medication or device that is not consistent with its labeling. Please read our full disclaimer here.
Overview
Methadone (injection) is an analgesic that is FDA approved for the treatment of moderate to severe pain and opioid dependence in patients unable to take oral medication. Common adverse reactions include pruritis, urticaria, abdominal pain, anorexia,constipation, dry mouth, glossitis, asthenia, edema and headache.
Adult Indications and Dosage
FDA-Labeled Indications and Dosage (Adult)
Indications
- For the treatment of moderate to severe pain not responsive to non-narcotic analgesics.
- For use in temporary treatment of opioid dependence in patients unable to take oral medication.
- Outpatient maintenance and outpatient detoxification treatment may be provided only by opioid treatment programs (OTPs) certified by the Federal Substance Abuse and Mental Health Services Administration (SAMHSA) and registered by the Drug Enforcement Administration (DEA). This does not preclude the maintenance treatment of a patient with concurrent opioid addiction who is hospitalized for conditions other than opioid addiction and who requires temporary maintenance during the critical period of hospitalization, or of a patient whose enrollment has been verified in a program which has been certified for maintenance treatment with methadone.
- NOTE: INJECTABLE METHADONE PRODUCTS ARE NOT APPROVED FOR THE OUTPATIENT TREATMENT OF OPIOID DEPENDENCE. IN THIS PATIENT POPULATION, PARENTERAL METHADONE IS TO BE USED ONLY FOR PATIENTS UNABLE TO TAKE ORAL MEDICATION, SUCH AS HOSPITALIZED PATIENTS.
Dosage
- Methadone differs from many other opioid agonists in several important ways. Methadone's pharmacokinetic properties, coupled with high interpatient variability in its absorption, metabolism, and relative analgesic potency, necessitate a cautious and highly individualized approach to prescribing. Particular vigilance is necessary during treatment initiation, during conversion from one opioid to another, and during dose titration.
- While methadone's duration of analgesic action (typically 4 to 8 hours) in the setting of single-dose studies approximates that of morphine, methadone's plasma elimination half-life is substantially longer than that of morphine (typically 8 to 59 hours vs. 1 to 5 hours). Methadone's peak respiratory depressant effects typically occur later, and persist longer than its peak analgesic effects. Also, with repeated dosing, methadone may be retained in the liver and then slowly released, prolonging the duration of action despite low plasma concentrations. For these reasons, steady-state plasma concentrations, and full analgesic effects, are usually not attained until 3 to 5 days of dosing. Additionally, incomplete cross-tolerance between μ-opioid agonists makes determination of dosing during opioid conversion complex.
- All of these characteristics make methadone dosing complex and can contribute to cases of iatrogenic overdose, particularly during treatment initiation and dose titration. A high degree of "opioid tolerance" does not eliminate the possibility of methadone overdose, iatrogenic or otherwise. Deaths have been reported during conversion to methadone from chronic, high-dose treatment with other opioid agonists.
Treatment of Pain
- Optimal methadone initiation and dose titration strategies for the treatment of pain have not been determined. Published equianalgesic conversion ratios between methadone and other opioids are imprecise, providing at best, only population averages that cannot be applied consistently to all patients. It should be noted that many commonly cited equianalgesia tables only present relative analgesic potencies of single opioid doses in non-tolerant patients, thus greatly underestimating methadone's analgesic potency, and its potential for adverse effects in repeated-dose settings. Regardless of the dose determination strategy employed, methadone is most safely initiated and titrated using small initial doses and gradual dose adjustments.
- As with all opioid drugs, it is necessary to adjust the dosing regimen for each patient individually, taking into account the patient's prior analgesic treatment experience. The following dosing recommendations should only be considered as suggested approaches to what is actually a series of clinical decisions over time in the management of the pain of each individual patient. Prescribers should always follow appropriate pain management principles of careful assessment and ongoing monitoring.
- In the selection of an initial dose of Methadone Hydrochloride Injection, attention should be given to the following:
- The total daily dose, potency and specific characteristics of the opioid the patient had been taking previously, if any;
- The relative potency estimate used to calculate an equianalgesic starting methadone dose, in particular, whether it is intended for use in acute or chronic methadone dosing;
- The patient's degree of opioid tolerance;
- The age, general condition and medical status of the patient;
- Concurrent medications, particularly other CNS and respiratory depressants;
- The type, severity and expected duration of the patient's pain;
- The acceptable balance between pain control and adverse side effects.
- Methadone Hydrochloride Injection may be administered intravenously, subcutaneously or intramuscularly. The absorption of subcutaneous and intramuscular methadone has not been well characterized and appears to be unpredictable. Local tissue reactions may occur.
- Parenteral products should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit.
Initiation of Therapy in Opioid Non-Tolerant Patients
- When parenteral methadone is used as the first analgesic in patients who are not already being treated with, and tolerant to, opioids, the usual intravenous methadone starting dose is 2.5 mg to 10 mg every 8 to 12 hours, slowly titrated to effect. More frequent administration may be required during methadone initiation in order to maintain adequate analgesia, and extreme caution is necessary to avoid overdosage, taking into account methadone's long elimination half life.
Conversion from Oral Methadone to Parenteral Methadone
- Conversion from oral methadone to parenteral methadone should initially use a 2:1 dose ratio (e.g., 10 mg oral methadone to 5 mg parenteral methadone).
Switching Patients to Parenteral Methadone from other Chronic Opioids
- Switching a patient from another chronically administered opioid to methadone requires caution due to the uncertainty of dose conversion ratios and incomplete cross-tolerance. Deaths have occurred in opioid tolerant patients during conversion to methadone.
- Conversion ratios in many commonly used equianalgesic dosing tables do not apply in the setting of repeated methadone dosing. Although with single-dose administration the onset and duration of analgesic action, as well as the analgesic potency of methadone and morphine, are similar methadone's potency increases over time with repeated dosing. Furthermore, the conversion ratio between methadone and other opiates varies dramatically depending on baseline opiate (morphine equivalent) use as shown in the table below.
- The dose conversion scheme below is derived from various consensus guidelines for converting chronic pain patients to methadone from morphine. The guidelines used to construct this table, however, were all designed for converting patients from oral morphine to oral methadone. The third column assumes a 2:1 ratio for converting from oral to intravenous methadone. Clinicians should consult published conversion guidelines to determine the equivalent morphine dose for patients converting from other opioids.
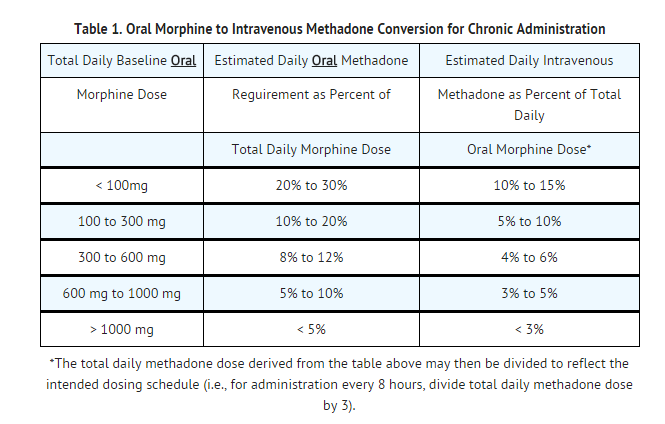
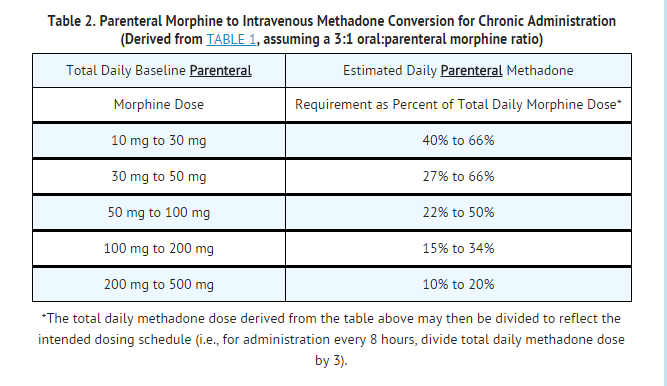
- Note: Equianalgesic methadone dosing varies not only between patients, but also within the same patient, depending on baseline morphine (or other opioid) dose. TABLES 1 and 2 have been included in order to illustrate this concept and to provide a safe starting point for opioid conversion. Methadone dosing should not be based solely on these tables. Methadone conversion and dose titration methods should always be individualized to account for the patient's prior opioid exposure, general medical condition, concomitant medication, and anticipated breakthrough medication use. The endpoint of titration is achievement of adequate pain relief, balanced against tolerability of opioid side effects. If a patient develops intolerable opioid related side effects, the methadone dose, or dosing interval, may need to be decreased.
- Methadone conversion and dose titration methods should always be individualized to account for the patient's prior opioid exposure, general medical condition, concomitant medication, and anticipated breakthrough medication use. The endpoint of titration is achievement of adequate pain relief, balanced against tolerability of opioid side effects. If a patient develops intolerable opioid related side effects, the methadone dose, or dosing interval, may need to be decreased.
Dosage Adjustment During Pregnancy
- Methadone clearance may be increased during pregnancy. Several small studies have demonstrated significantly lower trough methadone plasma concentrations and shorter methadone half-lives in women during their pregnancy compared to after their delivery. During pregnancy a woman's methadone dose may need to be increased, or their dosing interval decreased. Methadone should be used in pregnancy only if the potential benefit justifies the potential risk to the fetus.
Detoxification and Maintenance Treatment of Opiate Dependence
- For detoxification and maintenance of opiate dependence, methadone should be administered in accordance with the treatment standards cited in 42CFR Section 8.12, including limitations on unsupervised administration. Injectable methadone products are not approved for the outpatient treatment of opioid dependence. Parenteral methadone should be used only for patients who are unable to take oral medication, such as during hospitalization. The patient's oral methadone dose should be converted to an equivalent parenteral dose using the considerations above.
Off-Label Use and Dosage (Adult)
Guideline-Supported Use
There is limited information regarding Off-Label Guideline-Supported Use of Methadone (injection) in adult patients.
Non–Guideline-Supported Use
There is limited information regarding Off-Label Non–Guideline-Supported Use of Methadone (injection) in adult patients.
Pediatric Indications and Dosage
FDA-Labeled Indications and Dosage (Pediatric)
There is limited information regarding FDA-Labeled Use of Methadone (injection) in pediatric patients.
Off-Label Use and Dosage (Pediatric)
Guideline-Supported Use
There is limited information regarding Off-Label Guideline-Supported Use of Methadone (injection) in pediatric patients.
Non–Guideline-Supported Use
There is limited information regarding Off-Label Non–Guideline-Supported Use of Methadone (injection) in pediatric patients.
Contraindications
- Methadone Hydrochloride Injection is contraindicated in patients with a known hypersensitivity to methadone hydrochloride or any other ingredient in Methadone Hydrochloride Injection. Methadone Hydrochloride Injection is contraindicated in any situation where opioids are contraindicated such as: patients with respiratory depression (in the absence of resuscitative equipment or in unmonitored settings), and in patients with acute bronchial asthma or hypercarbia.
Warnings
Cardiac Conduction Effects

- Methadone should be administered with particular caution to patients already at risk for development of prolonged QT interval (e.g., cardiac hypertrophy, concomitant diuretic use, hypokalemia, hypomagnesemia). Careful monitoring is recommended when using methadone in patients with a history of cardiac conduction abnormalities, those taking medications affecting cardiac conduction, and in other cases where history or physical exam suggest an increased risk of dysrhythmia. QT prolongation has also been reported in patients with no prior cardiac history who have received high doses of methadone. Patients developing QT prolongation while on methadone treatment should be evaluated for the presence of modifiable risk factors, such as concomitant medications with cardiac effects, drugs which might cause electrolyte abnormalities, and drugs which might act as inhibitors of methadone metabolism. For use of methadone to treat pain, the risk of QT prolongation and development of dysrhythmias should be weighed against the benefit of adequate pain management and the availability of alternative therapies.
- Methadone treatment for analgesic therapy in patients with acute or chronic pain should only be initiated if the potential analgesic or palliative care benefit of treatment with methadone has been considered to outweigh the risk of QT prolongation that has been reported with high doses of methadone.
- The use of methadone in patients already known to have a prolonged QT interval has not been systematically studied.
- In using methadone an individualized benefit to risk assessment should be carried out and should include evaluation of patient presentation and complete medical history. For patients judged to be at risk, careful monitoring of cardiovascular status, including QT prolongation and dysrhythmias and those described previously should be performed.
Respiratory Depression
- Respiratory depression is the chief hazard from methadone hydrochloride. Respiratory depression is a particular potential problem in elderly or debilitated patients as well as in those suffering from conditions accompanied by hypoxia or hypercapnia when even moderate therapeutic doses may dangerously decrease pulmonary ventilation.
- Methadone Hydrochloride Injection should be administered with extreme caution to patients with conditions accompanied by hypoxia, hypercapnia, or decreased respiratory reserve such as; asthma, chronic obstructive pulmonary disease or cor pulmonale, severe obesity, sleep apnea syndrome, myxedema, kyphoscoliosis, CNS depression or coma. In these patients even usual therapeutic doses of methadone may decrease respiratory drive while simultaneously increasing airway resistance to the point of apnea. Alternative non-opioid analgesics should be considered, and methadone should be employed only under careful medical supervision at the lowest effective dose.
- Methadone's peak respiratory depressant effects typically occur later, and persist longer than its peak analgesic effects, in the short-term use setting. These characteristics can contribute to cases of iatrogenic overdose, particularly during treatment initiation and dose titration.
Incomplete Cross-Tolerance Between Methadone and Other Opioids
- Patients tolerant to other opioids may be incompletely tolerant to methadone. Incomplete cross-tolerance is a particular concern for patients tolerant to other μ-opioid agonists when converting to methadone, making determination of dosing during opioid conversion complex. Deaths have been reported during conversion from chronic, high dose treatment with other opioid agonists. Therefore, it is critical to understand the pharmacokinetics of methadone when converting patients from other opioids. A high degree of "opioid tolerance" does not eliminate the possibility of methadone toxicity.
Misuse, Abuse, and Diversion of Opioids
- Methadone is a μ-agonist opioid with an abuse liability similar to that of morphine and is a Schedule II controlled substance. Methadone, like morphine and other opioids used for analgesia, has the potential for being abused and is subject to criminal diversion.
- Methadone can be abused in a manner similar to other opioid agonists, legal or illicit. This should be considered when dispensing Methadone Hydrochloride Injection in situations where the clinician is concerned about an increased risk of misuse, abuse, or diversion.
- Concerns about abuse, addiction, diversion should not prevent the proper management of pain.
- Healthcare professionals should contact their State Professional Licensing Board, or State Controlled Substances Authority for information on how to prevent and detect abuse or diversion of this product.
Interactions with other CNS Depressants
- Patients receiving other opioid analgesics, general anesthetics, phenothiazines, other tranquilizers, sedatives, hypnotics, or other CNS depressants (including alcohol) concomitantly with methadone may experience respiratory depression, hypotension, profound sedation, or coma.
Interactions with Alcohol and Drugs of Abuse
- Methadone may be expected to have additive effects when used in conjunction with alcohol, other opioids, or illicit drugs that cause central nervous system depression. Deaths associated with illicit use of methadone have frequently involved concomitant benzodiazepine abuse.
Head Injury and Increased Intracranial Pressure
- The respiratory depressant effects of opioids and their capacity to elevate cerebrospinal-fluid pressure may be markedly exaggerated in the presence of head injury, other intracranial lesions or a pre-existing increase in intracranial pressure. Furthermore, opioids produce effects which may obscure the clinical course of patients with head injuries. In such patients, opioids must be used with caution, and only if it is deemed essential.
Acute Abdominal Conditions
- The administration of opioids may obscure the diagnosis of clinical course of patients with acute abdominal conditions.
Hypotensive Effect
- The administration of methadone may result in severe hypotension in patients whose ability to maintain normal blood pressure is compromised (i.e., severe volume depletion).
Adverse Reactions
Clinical Trials Experience
- Initial Administration: The initial methadone dose should be carefully titrated to the individual. Induction too rapid for the patient's sensitivity is more likely to produce adverse effects.
- The major hazards of methadone are respiratory depression and, to a lesser degree, systemic hypotension. Respiratory arrest, shock, cardiac arrest, and death have occurred.
- The most frequently observed adverse reactions include lightheadedness, dizziness, sedation, nausea, vomiting, and sweating. These effects seem to be more prominent in ambulatory patients and in those who are not suffering severe pain. In such individuals, lower doses of methadone are advisable.
- Other adverse reactions that have been reported in patients (including opioid addicts taking methadone for detoxification or maintenance) receiving methadone include the following:
- Cardiovascular: Arrhythmias, bigeminal rhythms, bradycardia, extrasystoles, tachycardia, Torsade de Pointes, ventricular fibrillation, ventricular tachycardia. ECG abnormalities, prolonged QT interval T-wave inversion, cardiomyopathy, flushing, heart failure, hypotension, palpitations, phlebitis, syncope.
- Digestive: Abdominal pain, anorexia, biliary tract spasm, constipation, dry mouth, glossitis
- Hematologic and Lymphatic: Reversible thrombocytopenia has been described in opioid addicts with chronic hepatitis.
- Metabolic and Nutritional: Hypokalemia, hypomagnesemia, weight gain
- Respiratory: Pulmonary edema
- Skin and appendages':
- Special senses: Visual disturbances
- Urogenital: Antidiuretic effect, amenorrhea, urinary retention or hesitancy, reduced libido and/or potency
- Maintenance on a Stabilized Dose: During prolonged administration of methadone, there is usually a gradual, yet progressive, disappearance of side effects over a period of several weeks. However, constipation and sweating often persist.
Postmarketing Experience
There is limited information regarding Postmarketing Experience of Methadone (injection) in the drug label.
Drug Interactions
- In vitro results indicate that methadone undergoes hepatic N-demethylation by cytochrome P450 enzymes, principally CYP3A4, and to a lesser extent CYP2D6. Coadministration of methadone with inducers of these enzymes may result in a more rapid metabolism and potential for decreased effects of methadone, whereas administration with inhibitors may reduce metabolism and potentiate methadone's effects. Therefore, drugs administered concomitantly with methadone should be evaluated for interaction potential; clinicians are advised to evaluate individual response to drug therapy.
- Opioid antagonists, mixed agonist/antagonists, and partial agonists: As with other μ-agonists, patients maintained on methadone may experience withdrawal symptoms when given these agents. Examples of such agents are naloxone, naltrexone, pentazocine, nalbuphine, butorphanol, and buprenorphine.
Anti-retroviral agents:
- Nevirapine: Based on the known metabolism of methadone, nevirapine may decrease plasma concentrations of methadone by increasing its hepatic metabolism. Opioid withdrawal syndrome has been reported in patients treated with nevirapine and methadone concomitantly. Methadone-maintained patients beginning nevirapine therapy should be monitored for evidence of withdrawal and methadone dose should be adjusted accordingly.
- Efavirenz: Coadministration of efavirenz in HIV-infected methadone-maintenance patients has resulted in decreased methadone plasma concentrations of methadone associated with signs of opiod withdrawal, and necessitating increases in methadone dose.
- Ritonavir and Ritonavir/Lopinavir: Reduced plasma methadone levels have been observed after administration of ritonavir alone or ritonavir/lopinavir combination. Withdrawal symptoms were however, inconsistently observed. Caution is warranted when administering methadone to patients receiving ritonavir-containing regimens in addition to other drugs known to decrease methadone plasma levels.
- Zidovudine: Experimental evidence suggests that methadone increases the area under the concentration-time curve (AUC) of zidovudine with possible toxic effects.
- Didanosine and Stavudine: Experimental evidence suggests that methadone decreased the AUC and peak levels for didanosine and stavudine, with a more significant decrease for didanosine. Methadone disposition was not substantially altered.
- Cytochrome P450 inducers: The following drug interactions were reported following coadministration of methadone with inducers of cytochrome P450 enzymes.
- Rifampin: In patients well-stabilized on methadone, concomitant administration of rifampin resulted in marked reduction in serum methadone levels and concurrent appearance of withdrawal symptoms.
- Phenytoin: In a pharmacokinetic study with patients on methadone maintenance therapy, phenytoin administration (250 mg b.i.d initially for 1 day followed by 300 mg QD for 3-4 days) resulted in ~50% reduction in methadone exposure and concurrently withdrawal symptoms occurred. Upon discontinuation of phenytoin, the incidence of withdrawal symptoms decreased and the methadone exposure increased and was comparable to pre-phenytoin dose scenario.
- St. John's Wort, phenobarbital, carbamazepine: Administration of methadone along with other CYP3A4 inducers may result in withdrawal symptoms.
- Cytochrome P450 inhibitors: Since the metabolism of methadone is mediated by the CYP3A4 isozyme, coadministration of drugs that inhibit CYP3A4 activity may cause decreased clearance of methadone. The expected clinical results would be increased or prolonged opioid effects. Thus patients coadministered with inhibitors of CYP3A4 such as azole antifungal agents (e.g., ketoconazole), macrolide antibiotics (e.g., erythromycin), while receiving methadone should be carefully monitored and dosage adjustment made if warranted. Some selective serotonin reuptake inhibitors (SSRI's) (i.e., sertraline, fluvoxamine) upon coadministration may increase methadone plasma levels and result in increased opiate effects or toxicity.
Others:
- Monoamine Oxidase (MAO) Inhibitors: Therapeutic doses of meperidine have precipitated severe reactions in patients concurrently receiving monoamine oxidase inhibitors or those who have received such agents within 14 days. Similar reactions thus far have not been reported with methadone; but if the use of methadone is necessary in such patients, a sensitivity test should be performed in which repeated small incremental doses are administered over the course of several hours while the patient's condition and vital signs are under careful observation.
- Desipramine: Blood levels of desipramine have increased with concurrent methadone therapy.
- Potentially Arrhythmogenic Agents: Extreme caution is necessary when any drug known to have the potential to prolong the QT interval is prescribed in conjunction with methadone. Pharmacodynamic interactions may occur with concomitant use of methadone and potentially arrhythmogenic agents such as class I and III antiarrhythmics, some neuroleptics and tricyclic antidepressants, and calcium channel blockers. Caution should also be exercised when prescribing concomitant drugs capable of inducing electrolyte disturbances that may prolong the QT interval (hypomagnesemia, hypokalemia). These include diuretics, laxatives, and in rare cases mineralocorticoid hormones.
- Interactions with other CNS Depressants: Patients receiving other opioid analgesics, general anesthetics, phenothiazines, other tranquilizers, sedatives, hypnotics, or other CNS depressants (including alcohol) concomitantly with methadone may experience respiratory depression, hypotension, profound sedation, or coma.
- Use with Mixed Agonist/Antagonist Opioid Analgesics: Agonist/antagonist analgesics (i.e., pentazocine, nalbuphine, butorphanol, or buprenorphine) should not be administered to patients who have received or are receiving a course of therapy with a pure opioid agonist, such as Methadone Hydrochloride Injection. In this situation, mixed agonist/antagonist analgesics may reduce the analgesic effect of Methadone Hydrochloride Injection and/or may precipitate withdrawal symptoms.
Use in Specific Populations
Pregnancy
Pregnancy Category (FDA): Teratogenic effects: Pregnancy Category C.
- There are no controlled studies of methadone use in pregnant women that can be used to establish safety. However, an expert review of published data on experiences with methadone use during pregnancy by TERIS - the Teratogen Information System -concluded that maternal use of methadone during pregnancy as part of a supervised, therapeutic regimen is unlikely to pose a substantial teratogenic risk (quantity and quality of data assessed as “limited to fair”), however, the data are insufficient to state that there is no risk (TERIS, last reviewed October, 2002). Pregnant women involved in methadone maintenance programs have been reported to have significantly improved prenatal care, improved fetal outcomes, and reduced mortality when compared to pregnant women using illicit drugs. Several factors complicate the interpretation of investigations of the children of women who took methadone during pregnancy. These include: the maternal use of illicit drugs, other maternal factors such as nutrition, infection, and psychosocial circumstances, limited information regarding dose and duration of methadone use during pregnancy, and the fact that most maternal exposure appears to occur after the first trimester of pregnancy. In addition, reported studies generally compare the benefit of methadone to the risk of untreated addiction to illicit drugs; the relevance of these findings to pain patients prescribed methadone during pregnancy is unclear.
- Methadone has been detected in amniotic fluid and cord plasma at concentrations proportional to maternal plasma and in newborn urine at lower concentrations than corresponding maternal urine.
- A retrospective series of 101 pregnant opiate-dependent women who underwent inpatient opiate detoxification with methadone did not demonstrate any increased risk of miscarriage in the 2nd trimester or premature delivery in the 3rd trimester.
- Several studies have suggested that infants born to narcotic-addicted women treated with methadone during all or part of pregnancy have been found to have decreased fetal growth with reduced birth weight, length, and/or head circumference compared to controls. The growth deficit does not appear to persist into later childhood. However, children born to women treated with methadone during pregnancy have been shown to demonstrate mild but persistent deficits in performance on psychometric and behavioral tests.
- Methadone should be used during pregnancy only if the potential benefit justifies the potential risk to the fetus.
- Additional information on the potential risks of methadone may be derived from animal data. Methadone does not appear to be teratogenic in the rat or rabbit models. However, following large doses, methadone produced teratogenic effects in the guinea pig, hamster and mouse. One published study found that in hamster fetuses, subcutaneous methadone doses of 31 mg/kg or greater (estimated exposure was approximately 2 times a human daily oral dose of 120 mg/day on a mg/m2 basis, or equivalent to a human daily intravenous dose of 120 mg/day) on day 8 of gestation produced exencephaly and neurological effects. Some of the reported effects were observed at doses that were maternally toxic. In another study, a single subcutaneous dose of 22-24 mg/kg methadone (estimated exposure was approximately equivalent to a human daily oral dose of 120 mg/day on a mg/m2 basis; or half a human daily intravenous dose of 120 mg/day) on day 9 of gestation in mice also produced exencephaly in 11% of the embryos. However, no effects were reported in rats and rabbits at oral doses up to 40 mg/kg (estimated exposure was approximately 3 and 6 times, respectively, a human daily oral dose of 120 mg/day on a mg/m2 basis; or 1.5 and 3 times a human daily intravenous dose of 120 mg/day) during days 6-15 and 6-18, respectively.
Nonteratogenetic Effects:
- Babies born to mothers who have been taking opioids regularly prior to delivery may be physically dependent. Onset of withdrawal symptoms in infants is usually in the first days after birth but may be delayed for two to four weeks. Withdrawal signs in the newborn include irritability and excessive crying, tremors, hyperactive reflexes, increased respiratory rate, increased stools, sneezing, yawning, vomiting, and fever. The intensity of the syndrome does not always correlate with the duration of maternal opioid dose or maternal dose. There is no consensus on the appropriate management of infant withdrawal.
- There are conflicting reports on whether the risk of sudden infant death syndrome (SIDS) is increased in infants born to women treated with methadone during pregnancy.
- Abnormal fetal nonstress tests (NSTs) have been reported to occur more frequently when the test is performed 1-2 hours after a maintenance dose of methadone in late pregnancy compared to controls. Published animal studies suggest that perinatal exposure to opioids including methadone may alter neuronal development and behavior in the offspring. Perinatal methadone exposure in rats has been linked to alterations in learning ability, motor activity, thermal regulation, nociception responses and sensitivity to other drugs. Additional animal data demonstrates evidence for neurochemical changes in the brains of methadone-treated offspring, including the cholinergic, dopaminergic, noradrenergic and serotonergic systems.
- Clinical Pharmacology for Pregnancy: Pregnant women have significantly lower trough plasma concentrations, increased plasma methadone clearance and shorter half-life than after delivery. Dosage adjustment using higher doses or administering the daily dose in divided doses may be necessary in pregnant women treated with methadone.
Pregnancy Category (AUS):
There is no Australian Drug Evaluation Committee (ADEC) guidance on usage of Methadone (injection) in women who are pregnant.
Labor and Delivery
- As with all opioids, administration of methadone to the mother shortly before delivery may result in some degree of respiratory depression in the newborn, especially if higher doses are used. Methadone is not recommended for obstetric analgesia because its long duration of action increases the probability of respiratory depression in the newborn. Narcotics with mixed agonist-antagonist properties should not be used for pain control during labor in patients chronically treated with methadone because they may precipitate acute withdrawal.
Nursing Mothers
- Methadone is secreted into human milk. There is no information on use of parenteral methadone in breast feeding, or on the safety of the high doses of methadone typically used in chronic pain treatment. The safety of breastfeeding while taking oral methadone is also controversial. At maternal oral doses of 10-80 mg/day, methadone concentrations from 50 to 570 ug/L in milk have been reported, which, in the majority of samples, were lower than maternal serum drug concentrations at steady state. Peak methadone levels in milk occur approximately 4-5 hours after an oral dose. Based on an average milk consumption of 150 mL/kg/day, an infant would consume approximately 17.4 ug/kg/day, which is approximately 2-3% of the oral maternal dose. Methadone has been detected in very low plasma concentrations in some infants whose mothers were taking methadone. Women on high dose methadone maintenance, who are already breast feeding, should be counseled to wean breast-feeding gradually in order to prevent neonatal abstinence syndrome. Methadone-treated mothers considering nursing an opioid-naïve infant should be counseled of the presence of methadone in breast milk.
- Because of the potential for serious adverse reactions in nursing infants from methadone, a decision should be made whether to discontinue nursing or to discontinue the drug, taking into account the importance of the drug to the mother.
Pediatric Use
- Safety and effectiveness in pediatric patients below the age of 18 years have not been established.
Geriatic Use
- Clinical studies of Methadone Hydrochloride Injection did not include sufficient numbers of subjects aged 65 and over to determine whether they respond differently from younger subjects. Other reported clinical experience has not identified differences in responses between the elderly and younger patients. In general, dose selection for elderly patients should be cautious, usually starting at the low end of the dosing range, reflecting the greater frequency of decreased hepatic, renal, or cardiac function and of concomitant disease or other drug therapy.
Gender
- The use of methadone has not been evaluated for gender specificity.
Race
There is no FDA guidance on the use of Methadone (injection) with respect to specific racial populations.
Renal Impairment
- The use of methadone has not been extensively evaluated in patients with renal insufficiency.
Hepatic Impairment
- The use of methadone has not been extensively evaluated in patients with hepatic insufficiency. Methadone is metabolized in the liver and patients with liver impairment may be at risk of accumulating methadone after multiple dosing.
Females of Reproductive Potential and Males
There is no FDA guidance on the use of Methadone (injection) in women of reproductive potentials and males.
Immunocompromised Patients
There is no FDA guidance one the use of Methadone (injection) in patients who are immunocompromised.
Administration and Monitoring
Administration
- Intravenous
Monitoring
There is limited information regarding Monitoring of Methadone (injection) in the drug label.
IV Compatibility
There is limited information regarding IV Compatibility of Methadone (injection) in the drug label.
Overdosage
Signs and Symptoms
Serious overdosage of methadone is characterized by respiratory depression (a decrease in respiratory rate and/or tidal volume, Cheyne-Stokes respiration, cyanosis), extreme somnolence progressing to stupor or coma, maximally constricted pupils, skeletal-muscle flaccidity, cold and clammy skin, and sometimes, bradycardia and hypotension. In severe overdosage, particularly by the intravenous route, apnea, circulatory collapse, cardiac arrest, and death may occur.
Treatment
- Primary attention should be given to the reestablishment of adequate respiratory exchange through provision of a patent airway and institution of assisted or controlled ventilation. If a non-tolerant person takes a large dose of methadone, effective opioid antagonists are available to counteract the potentially lethal respiratory depression. The physician must remember, however, that methadone is a long-acting depressant (36 to 48 hours), whereas the antagonists act for much shorter periods (one to three hours). The patient must, therefore, be monitored continuously for recurrence of respiratory depression and may need to be treated repeatedly with the narcotic antagonist. If the diagnosis is correct and respiratory depression is due only to overdosage of methadone, the use of other respiratory stimulants is not indicated.
- Opioid antagonists should not be administered in the absence of clinically significant respiratory or cardiovascular depression. In an individual physically dependent on opioids, the administration of the usual dose of an opioid antagonist may precipitate an acute withdrawal syndrome. The severity of this syndrome will depend on the degree of physical dependence and the dose of the antagonist administered. If antagonists must be used to treat serious respiratory depression in the physically dependent patient, the antagonist should be administered with extreme care and by titration with smaller than usual doses of the antagonist.
- Intravenously administered naloxone or nalmefene may be used to reverse signs of intoxication. Because of the relatively short half-life of naloxone as compared with methadone, repeated injections may be required until the status of the patient remains satisfactory. Naloxone may also be administered by continuous intravenous infusion.
- Oxygen, intravenous fluids, vasopressors, and other supportive measures should be employed as indicated.
Pharmacology
Mechanism of Action
- Methadone hydrochloride is a μ agonist; a synthetic opioid analgesic with multiple actions qualitatively similar to those of morphine, the most prominent of which involve the central nervous system and organs composed of smooth muscle. The principal therapeutic uses for methadone are for analgesia and for detoxification or maintenance in opioid addiction. The methadone abstinence syndrome, although qualitatively similar to that of morphine, differs in that the onset is slower, the course is more prolonged, and the symptoms are less severe. Some data also indicate that methadone acts as an antagonist at the N-methyl-D-aspartate (NMDA) receptor. The contribution of NMDA receptor antagonism to methadone's efficacy is unknown. Other NMDA receptor antagonists have been shown to produce neurotoxic effects in animals.
Structure
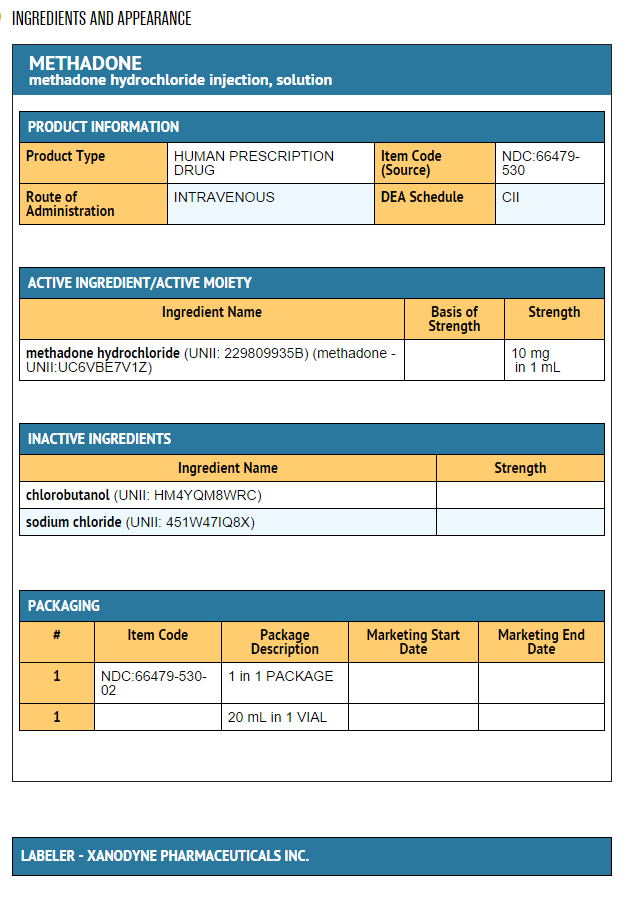
- Methadone Hydrochloride Injection, USP, 10 mg/mL is an opioid analgesic.
- Each milliliter of Methadone Hydrochloride Injection contains 10 mg (0.029 mmol) of methadone hydrochloride, equivalent to 8.95 mg of methadone free base.
- Methadone hydrochloride is a white, crystalline material that is water-soluble.
- Methadone hydrochloride is chemically described as 6-(dimethylamino)-4,4-diphenyl-3-hepatanone hydrochloride. Its molecular formula is C21H27NO•HCl and it has a molecular weight of 345.91. Methadone hydrochloride has a melting point of 235° C, and a pKa of 8.25 in water at 20°C. Its octanol/water partition coefficient at pH 7.4 is 117. A solution (1:100) in water has a pH between 4.5 and 6.5.
- It has the following structural formula:

- Methadone Hydrochloride Injection is a sterile injectable solution containing the following inactive ingredients: chlorobutanol, 0.5%, as a preservative, and sodium chloride. The pH of the sterile injectable solution may have been adjusted during manufacturing with sodium hydroxide and/or hydrochloric acid.
Pharmacodynamics
There is limited information regarding Pharmacodynamics of Methadone (injection) in the drug label.
Pharmacokinetics
Absorption:
- Methadone Hydrochloride Injection is intended for parenteral (intravenous, subcutaneous and intramuscular) administration. Methadone pharmacokinetics following subcutaneous and intramuscular administration have not been systematically studied and differences among the various parenteral routes have not been well characterized. As with many drugs, absorption into the systemic circulation may vary with subcutaneous and intramuscular administration.
Distribution:
- Methadone is a lipophilic drug and the steady state volume of distribution ranges between 2 -6 L/kg. In plasma, methadone is predominantly bound to α1-acid glycoprotein (85% - 90%). Methadone is secreted in saliva, breast milk, amniotic fluid and umbilical cord plasma.
Metabolism:
- Methadone is primarily metabolized by N-demethylation to an inactive metabolite, 2-ethylidene-1,5-dimethyl-3,3-diphenylpyrrolidene (EDDP). Cytochrome P450 enzymes, primarily CYP3A4 and to a lesser extent CYP2D6, are responsible for conversion of methadone to EDDP and other inactive metabolites, which are excreted mainly in urine.
Excretion:
- Elimination of methadone is mediated by extensive biotransformation, followed by renal and fecal excretion. After single intravenous dose administration the plasma clearance of methadone ranged between 3-10 L/h and the terminal half-life (t½) ranged between 8 - 59 hours. Methadone has been known to persist in the liver and other tissues. Slow release from the liver and other tissues may prolong the duration of methadone action despite low plasma concentrations.
Pharmacokinetics in Special Populations:
Pregnancy
- There are no pharmacokinetic studies of parenteral methadone in pregnancy. The disposition of oral methadone has been studied in approximately 30 pregnant patients in 2nd and 3rd trimesters. Elimination of methadone was significantly changed in pregnancy. Total body clearance of methadone was increased in pregnant patients compared to the same patients postpartum or to non-pregnant opioid-dependent women. The terminal half-life of methadone is decreased during second and third trimesters. The decrease in plasma half-life and increased clearance of methadone resulting in lower methadone trough levels during pregnancy can lead to withdrawal symptoms in some pregnant patients. The dosage may need to be increased or the dosing interval decreased in pregnant patients receiving methadone (See DOSAGE AND ADMINISTRATION).
Renal Impairment
- Methadone pharmacokinetics have not been extensively evaluated in patients with renal insufficiency. Unchanged methadone and its metabolites are excreted in urine to a variable degree. Methadone is a basic (pKa=9.2) compound and the luminal pH of the urinary tract can affect its extraction from plasma. Urine acidification has been shown to increase renal elimination of methadone. Forced diuresis, peritoneal dialysis, hemodialysis, or charcoal hemoperfusion have not been established as beneficial for increasing methadone or metabolite elimination.
Hepatic Impairment
- Methadone pharmacokinetics have not been extensively evaluated in patients with hepatic insufficiency. Methadone is metabolized in the liver and patients with liver impairment may be at risk of accumulating methadone after multiple dosing.
Gender
- The pharmacokinetics of methadone have not been evaluated for gender specificity.
Race
- The pharmacokinetics of methadone have not been evaluated for race specificity.
Geriatric
- The pharmacokinetics of methadone have not been evaluated in geriatric population.
Pediatric
- The pharmacokinetics of methadone have not been evaluated in pediatric population.
Drug Interactions
- Methadone undergoes hepatic N-demethylation by cytochrome P-450 isoforms, principally CYP3A4, and to a lesser extent CYP2D6. Coadministration of methadone with inducers of these enzymes may result in more rapid methadone metabolism and potentially, decreased effects of methadone. Conversely, administration with CYP3A4 or CYP2D6 inhibitors may reduce metabolism and potentiate methadone's effects. Therefore, drugs administered concomitantly with methadone should be evaluated for interaction potential.
Nonclinical Toxicology
Carcinogenesis, Mutagenesis, Impairment of Fertility
- Data from published reports of carcinogenicity studies indicate that there was a significant increase in pituitary adenomas in female B6C2F1 mice consuming 15 mg/kg/day methadone for two years. This dose was approximately 0.6 times a human daily oral dose of 120 mg/day, on a body surface area basis. However, this finding was not seen in mice consuming 60 mg/kg/day (approximately 2.5 times a human daily oral dose of 120 mg/day). Furthermore, in a two-year study of dietary administration of methadone to Fischer 344 rats, there was no clear evidence for treatment related increase in the incidence of neoplasms, at doses as high as 28 mg/kg/day in males and 88 mg/kg/day in females (approximately 2.3 times and 7.1 times, respectively, a human daily oral dose of 120 mg/day) based on body surface area comparison.
- In published reports, methadone tested negative in tests for chromosome breakage and disjunction and sex-linked recessive lethal gene mutations in germ cells of Drosophila using feeding and injection procedures. Methadone treatment of male mice increased sex chromosome and autosome univalent chromosomes and translocations in multivalent chromosomes. Methadone tested positive in the E.coli DNA repair system and Neurospora crassa and mouse lymphoma forward mutation assays.
Clinical Studies
There is limited information regarding Clinical Studies of Methadone (injection) in the drug label.
How Supplied
- Multiple-Dose Vials: 10 mg/mL, 20 mL, NDC 66479-530-02
- One vial per package
- Protect from light. Store in carton until contents have been used.
Storage
- Store at 25°C (77°F); excursions are permitted to 15°-30°C (59°-86°F)
Images
Drug Images
{{#ask: Page Name::Methadone (injection) |?Pill Name |?Drug Name |?Pill Ingred |?Pill Imprint |?Pill Dosage |?Pill Color |?Pill Shape |?Pill Size (mm) |?Pill Scoring |?NDC |?Drug Author |format=template |template=DrugPageImages |mainlabel=- |sort=Pill Name }}
Package and Label Display Panel
{{#ask: Label Page::Methadone (injection) |?Label Name |format=template |template=DrugLabelImages |mainlabel=- |sort=Label Page }}
Patient Counseling Information
Information for Patients
- Methadone, like all opioids, may impair the mental and/or physical abilities required for the performance of potentially hazardous tasks such as driving or operating machinery. The patient should be cautioned accordingly.
- Methadone, like other opioids, may produce orthostatic hypotension in ambulatory patients.
- Alcohol and other CNS depressants may produce an additive CNS depression, when taken with methadone, and should be avoided.
- If a patient taking methadone experiences symptoms suggestive of an arrhythmia (such as palpitations, dizziness, lightheadedness, or syncope), that patient should seek immediate medical attention.
Precautions with Alcohol
- Alcohol-Methadone (injection) interaction has not been established. Talk to your doctor about the effects of taking alcohol with this medication.
Brand Names
- METHADONE®[3]
Look-Alike Drug Names
- A® — B®[4]
Price
References
The contents of this FDA label are provided by the National Library of Medicine.
- ↑ 1.0 1.1 Fredheim, OM; Moksnes, K; Borchgrevink, PC; Kaasa, S; Dale, O (August 2008). "Clinical pharmacology of methadone for pain". Acta Anaesthesiologica Scandinavica. 52 (7): 879–89. doi:10.1111/j.1399-6576.2008.01597.x. PMID 18331375.
- ↑ 2.0 2.1 2.2 Brown, R; Kraus, C; Fleming, M; Reddy, S (November 2004). "Methadone: applied pharmacology and use as adjunctive treatment in chronic pain" (PDF). Postgraduate Medical Journal. 80 (949): 654–9. doi:10.1136/pgmj.2004.022988. PMC 1743125. PMID 15537850.
- ↑ "methadone hydrochloride injection, solution".
- ↑ "http://www.ismp.org". External link in
|title=(help)
{{#subobject:
|Page Name=Methadone (injection)
|Pill Name=No image.jpg
|Drug Name=
|Pill Ingred=|+sep=;
|Pill Imprint=
|Pill Dosage={{{dosageValue}}} {{{dosageUnit}}}
|Pill Color=|+sep=;
|Pill Shape=
|Pill Size (mm)=
|Pill Scoring=
|Pill Image=
|Drug Author=
|NDC=
}}
{{#subobject:
|Label Page=Methadone (injection) |Label Name=Methadone (injection)11.png
}}
{{#subobject:
|Label Page=Methadone (injection) |Label Name=Methadone (injection)11.png
}}