Cephalosporin
|
WikiDoc Resources for Cephalosporin |
|
Articles |
|---|
|
Most recent articles on Cephalosporin Most cited articles on Cephalosporin |
|
Media |
|
Powerpoint slides on Cephalosporin |
|
Evidence Based Medicine |
|
Clinical Trials |
|
Ongoing Trials on Cephalosporin at Clinical Trials.gov Trial results on Cephalosporin Clinical Trials on Cephalosporin at Google
|
|
Guidelines / Policies / Govt |
|
US National Guidelines Clearinghouse on Cephalosporin NICE Guidance on Cephalosporin
|
|
Books |
|
News |
|
Commentary |
|
Definitions |
|
Patient Resources / Community |
|
Patient resources on Cephalosporin Discussion groups on Cephalosporin Patient Handouts on Cephalosporin Directions to Hospitals Treating Cephalosporin Risk calculators and risk factors for Cephalosporin
|
|
Healthcare Provider Resources |
|
Causes & Risk Factors for Cephalosporin |
|
Continuing Medical Education (CME) |
|
International |
|
|
|
Business |
|
Experimental / Informatics |
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]
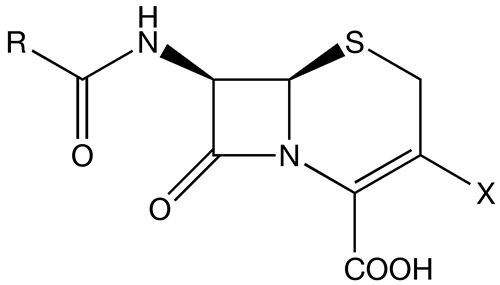
The cephalosporins (IPA: Template:IPA) are a class of β-lactam antibiotics. Together with cephamycins they belong to a sub-group called cephems.
History
Cephalosporin compounds were first isolated from cultures of Cephalosporium acremonium from a sewer in Sardinia in 1948 by Italian scientist Giuseppe Brotzu. He noticed that these cultures produced substances that were effective against Salmonella typhi, the cause of typhoid fever. Researchers at the Sir William Dunn School of Pathology at the University of Oxford isolated cephalosporin C, which had stability to β-lactamases but was not sufficiently potent for clinical use. The cephalosporin nucleus, 7-aminocephalosporanic acid (7-ACA), was derived from cephalosporin C and proved to be analogous to the penicillin nucleus 6-aminopenicillanic acid. Modification of the 7-ACA side-chains resulted in the development of useful antibiotic agents, and the first agent cephalothin (cefalotin) was launched by Eli Lilly in 1964.
Mode of action
Cephalosporins are bactericidal and have the same mode of action as other beta-lactam antibiotics (such as penicillins). Cephalosporins disrupt the synthesis of the peptidoglycan layer of bacterial cell walls. The peptidoglycan layer is important for cell wall structural integrity, especially in Gram-positive organisms. The final transpeptidation step in the synthesis of the peptidoglycan is facilitated by transpeptidases known as penicillin binding proteins (PBPs).
Clinical use
Indications
Cephalosporins are indicated for the prophylaxis and treatment of bacterial infections caused by susceptible organisms. First-generation cephalosporins are predominantly active against Gram-positive bacteria, and successive generations have increased activity against Gram-negative bacteria (albeit often with reduced activity against Gram-positive organisms).
Adverse effects
Common adverse drug reactions (ADRs) (≥1% of patients) associated with cephalosporin therapy include: diarrhea, nausea, rash, electrolyte disturbances, and/or pain and inflammation at injection site. Infrequent ADRs (0.1–1% of patients) include: vomiting, headache, dizziness, oral and vaginal candidiasis, pseudomembranous colitis, superinfection, eosinophilia, and/or fever.
Approximately 5–10% of patients with allergic hypersensitivity to penicillins and/or carbapenems will also have cross-reactivity with cephalosporins. Thus, they are contraindicated in patients with a history of severe, immediate allergic reactions (urticaria, anaphylaxis, interstitial nephritis, etc) to penicillins, carbapenems or cephalosporins.[1] This however should be viewed in the light of recent epidemiological work suggesting that for many 2nd generation (or later) cephalosporins that the cross-reactivity rate with penicillin is much lower, having no significantly increased risk of reactivity in the studies examined.[2]
Several cephalosporins are associated with hypoprothrombinemia and a disulfiram-like reaction.[3][4] These include latamoxef, cefmenoxime, moxalactam, cefoperazone, cefamandole, cefmetazole, and cefotetan. This is thought to be due to the N-methylthiotetrazole (NMTT) side chain of these cephalosporins which blocks the enzyme vitamin K epoxide reductase.
Classification
The cephalosporin nucleus can be modified to gain different properties. Cephalosporins are sometimes grouped into "generations" by their antimicrobial properties. The first cephalosporins were designated first generation while later, more extended spectrum cephalosporins were classified as second generation cephalosporins. Each newer generation of cephalosporins has significantly greater Gram-negative antimicrobial properties than the preceding generation, in most cases with decreased activity against Gram-positive organisms. Fourth generation cephalosporins, however, have true broad spectrum activity.
The classification of cephalosporins into "generations" is commonly practised, although the exact categorisation of cephalosporins is often imprecise. For example, the fourth generation of cephalosporins is not yet recognized in Japan. In Japan, cefaclor is classed as a first generation cephalosporin even though in the United States it is a 2nd generation; and cefbuperazone, cefminox and cefotetan are classed as second generation cephalosporins. Cefmetazole and cefoxitin are classed as third generation cephems. Flomoxef, latamoxef are in a new class called oxacephems.
Most first generation cephalosporins were originally spelled "ceph-" in English-speaking countries. This continues to be the preferred spelling in the US and Australia, while European countries have adopted International Nonproprietary Names, which are usually spelt "cef-". Newer first-generation cephalosporins and all cephalosporins of later generations are spelled "cef-".

First generation
First generation cephalosporins are moderate spectrum agents, with a spectrum of activity that includes penicillinase-producing, methicillin-susceptible staphylococci and streptococci, though they are not the drugs of choice for such infections. They also have activity against some Escherichia coli, Klebsiella pneumoniae and Proteus mirabilis, but have no activity against Bacteroides fragilis, enterococci, methicilllin-resistant staphylococci, Pseudomonas, Acinetobacter, Enterobacter, indole-positive Proteus or Serratia.
- Cefacetrile (cephacetrile)
- Cefadroxil (cefadroxyl; Duricef®)
- Cefalexin (cephalexin; Keflex®)
- Cephaloglycin
- Cefalonium (cephalonium)
- Cefaloridine (cephaloradine)
- Cefalotin (cephalothin; Keflin®)
- Cefapirin (cephapirin; Cefadryl®)
- Cefatrizine
- Cefazaflur
- Cefazedone
- Cefazolin (cephazolin; Ancef®, Kefzol®)
- Cefradine (cephradine; Velosef®)
- Cefroxadine
- Ceftezole
Second generation
The second generation cephalosporins have a greater Gram-negative spectrum while retaining some activity against Gram-positive cocci. They are also more resistant to beta-lactamase.
- Cefonicid (Monocid®)
- Cefprozil (cefproxil; Cefzil®)
- Cefuroxime (Zinnat®, Zinacef®, Ceftin®, Biofuroksym®[5])
- Cefuzonam
- Cefaclor (Ceclor®, Distaclor®, Keflor®, Raniclor®)
Second generation cephalosporins with antianaerobe activity
The following cephems are also sometimes grouped with second-generation cephalosporins:
- Carbacephems: loracarbef (Lorabid®)
- Cephamycins: cefbuperazone, cefmetazole (Zefazone®), cefminox, cefotetan (Cefotan®), cefoxitin (Mefoxin®)
Third generation
Third generation cephalosporins have a broad spectrum of activity and further increased activity against Gram-negative organisms. Some members of this group (particularly those available in an oral formulation, and those with anti-pseudomonal activity) have decreased activity against Gram-positive organisms. They may be particularly useful in treating hospital-acquired infections, although increasing levels of extended-spectrum beta-lactamases are reducing the clinical utility of this class of antibiotics.
- Cefcapene
- Cefdaloxime
- Cefdinir (Omnicef®)
- Cefditoren
- Cefetamet
- Cefixime (Suprax®)
- Cefmenoxime
- Cefodizime
- Cefoperazone (Cefobid®)
- Cefotaxime (Claforan®)
- Cefpimizole
- Cefpodoxime (Vantin®)
- Cefteram
- Ceftibuten (Cedax®)
- Ceftiofur
- Ceftiolene
- Ceftizoxime (Cefizax®)
- Ceftriaxone (Rocephin®)
Cephalosporins with antipseudomonal activity
- Ceftazidime (Fortum®, Fortaz®) (3rd Generation Cephalosporin)
- Cefoperazone (3rd Generation Cephalosporin)
- Cefipime (4th Generation Cephalosporin)
The following cephems are also sometimes grouped with third-generation cephalosporins:
- Oxacephems: latamoxef (moxalactam)
Fourth generation
Fourth generation cephalosporins are extended-spectrum agents with similar activity against Gram-positive organisms as first-generation cephalosporins. They also have a greater resistance to beta-lactamases than the third generation cephalosporins. Many can cross blood brain barrier and are effective in meningitis. They are also used against Pseudomonas aeruginosa.
- Cefclidine
- Cefepime (Maxipime®)
- Cefluprenam
- Cefoselis
- Cefozopran
- Cefpirome
- Cefquinome
The following cephems are also sometimes grouped with third-generation cephalosporins:
Yet to be classified
These cephems have progressed far enough to be named, but have not been assigned to a particular generation. Ceftobiprole (and the soluble prodrug medocaril) are on the FDA fast track. Ceftobiprole has powerful antipseudomonal characteristics and appears to be less susceptible to development of resistance.
- Cefaclomezine
- Cefaloram
- Cefaparole
- Cefcanel
- Cefedrolor
- Cefempidone
- Cefetrizole
- Cefivitril
- Cefmatilen
- Cefmepidium
- Cefovecin
- Cefoxazole
- Cefrotil
- Cefsumide
- Ceftioxide
- Ceftobiprole (previously BAL 9141 and RO 63-9141)
- Ceftobiprole (previously BAL 5788)
- Cefuracetime
Cephalosporins in Popular Culture
In the fourth season premiere of House, MD, a patient suffers from an allergy to cephalosporin.
References
- ↑ Rossi S, editor. Australian Medicines Handbook 2006. Adelaide: Australian Medicines Handbook; 2006.
- ↑ Pichichero ME. Cephalosporins can be prescribed safely for penicillin-allergic patients. J Fam Pract. 2006 Feb;55(2):106-12. PMID 16451776
- ↑ Kitson TM. The effect of cephalosporin antibiotics on alcohol metabolism: a review. Alcohol. 1987 May-Jun;4(3):143-8. PMID 3593530.
- ↑ Shearer MJ, Bechtold H, Andrassy K, Koderisch J, McCarthy PT, Trenk D, Jahnchen E, Ritz E. Mechanism of cephalosporin-induced hypoprothrombinemia: relation to cephalosporin side chain, vitamin K metabolism, and vitamin K status. J Clin Pharmacol. 1988 Jan;28(1):88-95. PMID 3350995.
- ↑ Jędrzejczyk, Tadeusz. "Internetowa Encyklopedia Leków". leki.med.pl. Retrieved 2007-03-03.
See also
External links
- MedlinePlus Drug Information: Cephalosporins (systemic) – information from USP DI Advice for the Patient (Broken Link)
- Cephalosporins College of Health and Life Sciences, Fort Hays State University.
- Cephalosporins "Family Practice Notebook" page on Cephalosporins.
- Cephalosporins can be prescribed safely for penicillin allergic patients, full article
- Chemical Land 21 Cephalosporin class antibiotics
- Structure Activity Relationships "Antibacterial Agents; Structure Activity Relationships," André Bryskier MD; beginning at pp83
ar:سيفالوسبورين
de:Cephalosporine
he:צפלוספורין
nl:Cefalosporine