Cefacetrile
|
WikiDoc Resources for Cefacetrile |
|
Articles |
|---|
|
Most recent articles on Cefacetrile Most cited articles on Cefacetrile |
|
Media |
|
Powerpoint slides on Cefacetrile |
|
Evidence Based Medicine |
|
Clinical Trials |
|
Ongoing Trials on Cefacetrile at Clinical Trials.gov Clinical Trials on Cefacetrile at Google
|
|
Guidelines / Policies / Govt |
|
US National Guidelines Clearinghouse on Cefacetrile
|
|
Books |
|
News |
|
Commentary |
|
Definitions |
|
Patient Resources / Community |
|
Patient resources on Cefacetrile Discussion groups on Cefacetrile Patient Handouts on Cefacetrile Directions to Hospitals Treating Cefacetrile Risk calculators and risk factors for Cefacetrile
|
|
Healthcare Provider Resources |
|
Causes & Risk Factors for Cefacetrile |
|
Continuing Medical Education (CME) |
|
International |
|
|
|
Business |
|
Experimental / Informatics |
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]
Overview
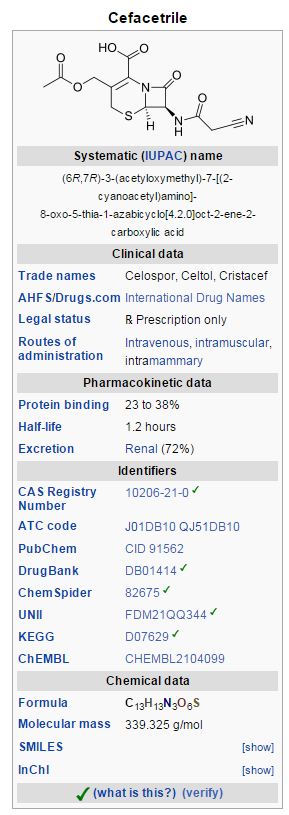
Cefacetrile (INN, also spelled cephacetrile) is a broad-spectrum first generation cephalosporin antibiotic effective in gram-positive and gram-negative bacterial infections. It is a bacteriostatic antibioticCefacetrile is marketed under the trade names Celospor, Celtol, and Cristacef, and as Vetimast for the treatment of mammary infections in lactating cows.