Ceftazidime
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]; Associate Editor(s)-in-Chief: Alberto Plate [2]
WikiDoc MAKES NO GUARANTEE OF VALIDITY. WikiDoc is not a professional health care provider, nor is it a suitable replacement for a licensed healthcare provider. WikiDoc is intended to be an educational tool, not a tool for any form of healthcare delivery. The educational content on WikiDoc drug pages is based upon the FDA package insert, National Library of Medicine content and practice guidelines / consensus statements. WikiDoc does not promote the administration of any medication or device that is not consistent with its labeling. Please read our full disclaimer here.
Overview
Ceftazidime is a cephalosporin that is FDA approved for the treatment of bacterial infections of gram negative, gram positive aerobic bacterias and for anaerobic bacterias. Common adverse reactions include diarrhea.
Adult Indications and Dosage
FDA-Labeled Indications and Dosage (Adult)
Lower Respiratory Tract Infections
- Pneumonia: caused by Pseudomonas aeruginosa and other Pseudomonas spp.; Haemophilus influenzae, including ampicillin-resistant strains; Klebsiella spp.; Enterobacter spp; Proteus mirabilis; Escherichia coli; Serratia spp; Citrobacter spp; Streptococcus pneumoniae; and Staphylococcus aureus (methicillin-susceptible strains).
Skin and Skin-Structure Infections
- Caused by Pseudomonas aeruginosa; Klebsiella spp.; Escherichia coli; Proteus spp., including Proteus mirabilis and indole-positive Proteus; Enterobacter spp.; Serratia spp.; Staphylococcus aureus (methicillin-susceptible strains); and Streptococcus pyogenes (group A beta-hemolytic streptococci).
- Both complicated and uncomplicated, caused by Pseudomonas aeruginosa; Enterobacter spp; Proteus spp., including Proteus mirabilis and indole-positive Proteus; Klebsiella spp.; and Escherichia coli.
- Caused by Pseudomonas aeruginosa, Klebsiella spp., Haemophilus influenzae, Escherichia coli, Serratia spp., Streptococcus pneumoniae, and Staphylococcus aureus (methicillin-susceptible strains).
Bone and Joint Infections
- Caused by Pseudomonas aeruginosa, Klebsiella spp., Enterobacter spp., and Staphylococcus aureus (methicillin-susceptible strains).
Gynecologic Infections
- Including endometritis, pelvic cellulitis, and other infections of the female genital tract caused by Escherichia coli.
- Including peritonitis caused by Escherichia coli, Klebsiella spp., and Staphylococcus aureus (methicillin-susceptible strains) and polymicrobial infections caused by aerobic and anaerobic organisms and Bacteroides spp (many strains of Bacteroides fragilis are resistant).
Central Nervous System Infections
- Including meningitis, caused] by Haemophilus influenzae and Neisseria meningitidis. Ceftazidime has also been used successfully in a limited number of cases of meningitis due to Pseudomonas aeruginosa and Streptococcus pneumoniae.
- The usual adult dosage is 1 gram administered intravenously every 8 to 12 hours. The dosage should be determined by the susceptibility of the causative organisms, the severity of infection, and the condition and renal function of the patient.
- The guidelines for dosage of ceftazidime for injection are listed in the following table. The following dosage schedule is recommended:
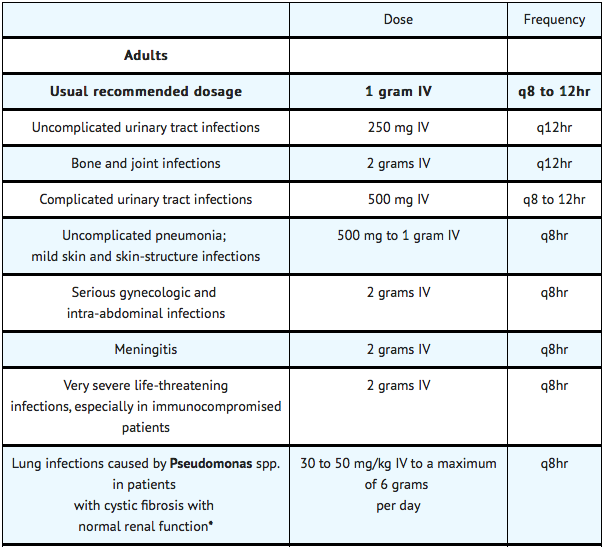
- In patients with severe infections who would normally receive 6 grams of ceftazidime for injection daily were it not for renal insufficiency, the unit dose given in the table above may be increased by 50% or the dosing frequency may be increased appropriately. Further dosing should be determined by therapeutic monitoring, severity of the infection, and susceptibility of the causative organism.
- In pediatric patients as for adults, the creatinine clearance should be adjusted for body surface area or lean body mass, and the dosing frequency should be reduced in cases of renal insufficiency.
- In patients undergoing hemodialysis, a loading dose of 1 gram is recommended, followed by 1 gram after each hemodialysis period.
Ceftazidime for injection can also be used in patients undergoing intraperitoneal dialysis and continuous ambulatory peritoneal dialysis. In such patients, a loading dose of 1 gram of ceftazidime for injection may be given, followed by 500 mg every 24 hours. In addition to IV use, ceftazidime for injection can be incorporated in the dialysis fluid at a concentration of 250 mg for 2 L of dialysis fluid.
Off-Label Use and Dosage (Adult)
Guideline-Supported Use
Febrile Neutropenia[1]
- Single Therapy
- Double Therapy
- Ceftazidime + Amikacin
- Ceftazidine + Tobramycin
Non–Guideline-Supported Use
Melioidosis[2]
- Dosage
- Scheme 1: 4 milligrams/kilogram/hour after a 12 milligrams/kilogram (mg/kg) loading dose
- Scheme 2: 40 mg/kg every 8 hours
Pediatric Indications and Dosage
FDA-Labeled Indications and Dosage (Pediatric)

Off-Label Use and Dosage (Pediatric)
Guideline-Supported Use
Treatment and Prophylaxis of Infections in HIV Infected Patients
- An antipseudomonal drug, such as ceftazidime or imipenem, should be added for neutropenic patients, with the addition of an aminoglycoside if infection with pseudomonas is likely
- For patients with neutropenia, chronic lung disease other than asthma (lymphoid interstitial pneumonia, bronchiectasis), or indwelling venous catheter, consider regimens that include activity against P. aeruginosa, such as cefepime or ceftazidime instead of ceftriaxone [3]
Febrile Neutropenia[4]
- Single Therapy
- Double Therapy
- Ceftazidime + Amikacin
- Ceftazidine + Tobramycin
Non–Guideline-Supported Use
There is limited information regarding Off-Label Non–Guideline-Supported Use of Ceftazidime in pediatric patients.
Contraindications
- Ceftazidime for injection is contraindicated in patients who have shown hypersensitivity to ceftazidime or the cephalosporin group of antibiotics.
Warnings
Hypersensitivity
- Before therapy with Ceftazidime for injection is instituted, careful inquiry should be made to determine whether the patient has had previous hypersensitivity reactions to ceftazidime, cephalosporins, penicillins, or other drugs. if this product is to be given to penicillin-sensitive patients, caution should be exercised because cross-hypersensitivity among beta-lactam antibiotics has been clearly documented and may occur in up to 10% of patients with a history of penicillin allergy. if an allergic reaction to ceftazidime for injection occurs, discontinue the drug. Serious acute hypersensitivity reactions may require treatment with epinephrine and other emergency measures, including oxygen, IV fluids, iv antihistamines, corticosteroids, pressor amines, and airway management, as clinically indicated.
Clostridium Difficile Associated Diarrhea
- Clostridium difficile associated diarrhea (CDAD) has been reported with use of nearly all antibacterial agents, including ceftazidime for injection, and may range in severity from mild diarrhea to fatal colitis. Treatment with antibacterial agents alters the normal flora of the colon, leading to overgrowth of C. difficile.
- C. difficile produces toxins A and B which contribute to the development of CDAD. Hypertoxin producing strains of C. difficile cause increased morbidity and mortality, as these infections can be refractory to antimicrobial therapy and may require colectomy. CDAD must be considered in all patients who present with diarrhea following antibiotic use. Careful medical history is necessary since CDAD has been reported to occur over two months after the administration of antibacterial agents.
- If CDAD is suspected or confirmed, ongoing antibiotic use not directed against C. difficile may need to be discontinued. Appropriate fluid and electrolyte management, protein supplementation, antibiotic treatment of C. difficile, and surgical evaluation should be instituted as clinically indicated. Elevated levels of ceftazidime in patients with renal insufficiency can lead to seizures, encephalopathy, coma, asterixis, neuromuscular excitability, and myoclonia.
Adverse Reactions
Clinical Trials Experience
Ceftazidime is generally well tolerated. The incidence of adverse reactions associated with the administration of ceftazidime was low in clinical trials. The most common were local reactions following IV injection and allergic and gastrointestinal reactions. Other adverse reactions were encountered infrequently. No disulfiram-like reactions were reported. The following adverse effects from clinical trials were considered to be either related to ceftazidime therapy or were of uncertain etiology:
Local Effects
- Phlebitis
- Inflammation at the site of injection (1 in 69 patients).
Hypersensitivity Reactions
- Pruritus
- Rash
- Fever
- Toxic epidermal necrolysis
- Stevens-Johnson syndrome
- Erythema multiform
- Angioedema
- Anaphylaxis (bronchospasm and/or hypotension) have been reported very rarely.
Gastrointestinal Symptoms
- Diarrhea
- Nausea
- Vomiting
- Abdominal pain
- The onset of pseudomembranous colitis symptoms may occur during or after treatment
Central Nervous System Reactions
- Headache
- Dizziness and
- Paresthesia
- Seizures
- Encephalopathy
- Coma
- Asterixis
- Neuromuscular excitability
- Myoclonia have been reported in renally impaired patients treated with unadjusted dosing regimens of ceftazidime.
Less Frequent Adverse Events
Hematologic
- Rare cases of hemolytic anemia
- Transient leukopenia, neutropenia, agranulocytosis, thrombocytopenia, and lymphocytosis were seen very rarely.
Laboratory Test Changes
- Eosinophilia
- Positive Coombs test without hemolysis
- Thrombocytosis
- Liver enzyme elevation:
- As with some other cephalosporins, transient elevations of blood urea, blood urea nitrogen, and/or serum creatinine were observed occasionally.
Postmarketing Experience
In addition to the adverse events reported during clinical trials, the following events have been observed during clinical practice in patients treated with ceftazidime and were reported spontaneously. For some of these events, data are insufficient to allow an estimate of incidence or to establish causation.
General
Hepatobiliary Tract
Renal and Genitourinary
Cephalosporin-Class Adverse Reactions
In addition to the adverse reactions listed above that have been observed in patients treated with ceftazidime, the following adverse reactions and altered laboratory tests have been reported for cephalosporin-class antibiotics:
- Adverse Reactions: Colitis, toxic nephropathy, hepatic dysfunction including cholestasis, aplastic anemia, hemorrhage.
- Altered Laboratory Tests: Prolonged prothrombin time, false-positive test for urinary glucose, pancytopenia.
Drug Interactions
- Nephrotoxicity has been reported following concomitant administration of cephalosporins with aminoglycoside antibiotics or potent diuretics such as furosemide. Renal function should be carefully monitored, especially if higher dosages of the aminoglycosides are to be administered or if therapy is prolonged, because of the potential nephrotoxicity and ototoxicity of aminoglycosidic antibiotics. Nephrotoxicity and ototoxicity were not noted when ceftazidime was given alone in clinical trials.
- Chloramphenicol has been shown to be antagonistic to beta-lactam antibiotics, including ceftazidime, based on in vitro studies and time kill curves with enteric gram-negative bacilli. Due to the possibility of antagonism in vivo, particularly when bactericidal activity is desired, this drug combination should be avoided.
- In common with other antibiotics, ceftazidime may affect the gut flora, leading to lower estrogen reabsorption and reduced efficacy of combined oral estrogen/progesterone contraceptives.
Drug/Laboratory Test Interactions
- The administration of ceftazidime may result in a false-positive reaction for glucose in the urine when using CLINITEST® tablets, Benedict's solution, or Fehling's solution. It is recommended that glucose tests based on enzymatic glucose oxidase reactions (such as CLINISTIX®) be used.
Use in Specific Populations
Pregnancy
- Reproduction studies have been performed in mice and rats at doses up to 40 times the human dose and have revealed no evidence of impaired fertility or harm to the fetus due to ceftazidime for injection. There are, however, no adequate and well-controlled studies in pregnant women. Because animal reproduction studies are not always predictive of human response, this drug should be used during pregnancy only if clearly needed.
Pregnancy Category (AUS):
There is no Australian Drug Evaluation Committee (ADEC) guidance on usage of Ceftazidime in women who are pregnant.
Labor and Delivery
There is no FDA guidance on use of Ceftazidime during labor and delivery.
Nursing Mothers
- Ceftazidime is excreted in human milk in low concentrations. Caution should be exercised when ceftazidime is administered to a nursing woman.
Pediatric Use
There is no FDA guidance on the use of Ceftazidime in pediatric settings.
Geriatic Use
- Of the 2,221 subjects who received ceftazidime in 11 clinical studies, 824 (37%) were 65 and over while 391 (18%) were 75 and over. No overall differences in safety or effectiveness were observed between these subjects and younger subjects, and other reported clinical experience has not identified differences in responses between the elderly and younger patients, but greater susceptibility of some older individuals to drug effects cannot be ruled out. This drug is known to be substantially excreted by the kidney, and the risk of toxic reactions to this drug may be greater in patients with impaired renal function. Because elderly patients are more likely to have decreased renal function, care should be taken in dose selection, and it may be useful to monitor renal function.
Gender
There is no FDA guidance on the use of Ceftazidime with respect to specific gender populations.
Race
There is no FDA guidance on the use of Ceftazidime with respect to specific racial populations.
Renal Impairment
- Ceftazidime is excreted by the kidneys, almost exclusively by glomerular filtration. Therefore, in patients with impaired renal function (glomerular filtration rate [GFR] <50 mL/min), it is recommended that the dosage of ceftazidime be reduced to compensate for its slower excretion. In patients with suspected renal insufficiency, an initial loading dose of 1 gram of ceftazidime may be given. An estimate of GFR should be made to determine the appropriate maintenance dosage.
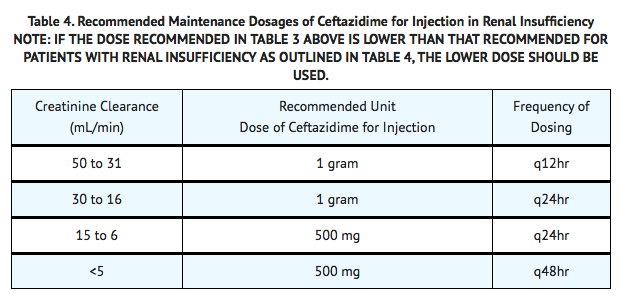
Hepatic Impairment
- No adjustment in dosage is required for patients with hepatic dysfunction.
Females of Reproductive Potential and Males
- Long-term studies in animals have not been performed to evaluate carcinogenic potential. However, a mouse Micronucleus test and an Ames test were both negative for mutagenic effects.
Immunocompromised Patients
There is no FDA guidance one the use of Ceftazidime in patients who are immunocompromised.
Administration and Monitoring
Administration
- Ceftazidime for injection may be given intravenously. Intra-arterial administration should be avoided. The IV route is preferable for patients with bacterial septicemia, bacterial meningitis, peritonitis, or other severe or life-threatening infections, or for patients who may be poor risks because of lowered resistance resulting from such debilitating conditions as malnutrition, trauma, surgery, diabetes, heart failure, or malignancy, particularly if shock is present or pending.
Directions for Proper Use of a Pharmacy Bulk Package
- Not for direct infusion. This Pharmacy Bulk Package is for use in a hospital pharmacy admixture service, only in a suitable work area, such as a laminar flow hood. Using aseptic technique, the container closure may be penetrated only one time using a suitable sterile dispensing set or transfer device that allows measured dispensing of the contents. Use of a syringe and needle is not recommended as it may cause leakage. The withdrawal of container contents should be accomplished without delay. However, should this not be possible, a maximum time of 4 HOURS from initial closure entry is permitted to complete fluid transfer operations. This time limit should begin with the introduction of the solvent or diluent into the Pharmacy Bulk Package. Discard any unused portion after 4 hours. Not for direct infusion. This pharmacy bulk package is not intended to be dispensed as a unit.
- For IV infusion, constitute the 6 g Pharmacy Bulk Package bottle with Sterile Water for Injection and add an appropriate quantity of the resulting solution to an IV container with one of the compatible IV fluids.
- Intermittent IV infusion with a Y-type administration set can be accomplished with compatible solutions. However, during infusion of a solution containing ceftazidime, it is desirable to discontinue the other solution.

Monitoring
There is limited information regarding Ceftazidime Monitoring in the drug label.
IV Compatibility
Intravenous
- Ceftazidime for injection, when constituted as directed with Sterile Water for Injection should have the contents withdrawn within 4 hours. Solutions in Sterile Water for Injection in the infusion vial or in 0.9% Sodium Chloride Injection in VIAFLEX® small-volume containers that are frozen immediately after constitution are stable for 6 months when stored at -20°C. Do not force thaw by immersion in water baths or by microwave irradiation. Once thawed, solutions should not be refrozen. Thawed solutions may be stored for up to 24 hours at room temperature or for 7 days in a refrigerator.
- Ceftazidime is compatible with the more commonly used IV infusion fluids. Solutions at concentrations between 1 and 40 mg/mL in 0.9% Sodium Chloride Injection; 1/6 M Sodium Lactate Injection; 5% Dextrose Injection; 5% Dextrose and 0.225% Sodium Chloride Injection; 5% Dextrose and 0.45% Sodium Chloride Injection; 5% Dextrose and 0.9% Sodium Chloride Injection; 10% Dextrose Injection; Ringer's Injection, USP; Lactated Ringer's Injection, USP; 10% Invert Sugar in Water for Injection; and NORMOSOL®-M in 5% Dextrose Injection may be stored for up to 24 hours at room temperature or for 7 days if refrigerated.
- Ceftazidime for injection is less stable in Sodium Bicarbonate Injection than in other IV fluids. It is not recommended as a diluent. Solutions of ceftazidime for injection in 5% Dextrose Injection and 0.9% Sodium Chloride Injection are stable for at least 6 hours at room temperature in plastic tubing, drip chambers, and volume control devices of common IV infusion sets.
- Ceftazidime at a concentration of 4 mg/mL has been found compatible for 24 hours at room temperature or for 7 days under refrigeration in 0.9% Sodium Chloride Injection or 5% Dextrose Injection when admixed with: cefuroxime sodium 3 mg/mL, heparin 10 or 50 U/mL, or potassium chloride 10 or 40 mEq/L.
- Vancomycin solution exhibits a physical incompatibility when mixed with a number of drugs, including ceftazidime. The likelihood of precipitation with ceftazidime is dependent on the concentrations of vancomycin and ceftazidime present. It is therefore recommended, when both drugs are to be administered by intermittent IV infusion, that they be given separately, flushing the IV lines (with 1 of the compatible IV fluids) between the administration of these 2 agents.
Note: Parenteral drug products should be inspected visually for particulate matter before administration whenever solution and container permit. As with other cephalosporins, ceftazidime for injection powder, as well as solutions, tend to darken depending on storage conditions; within the stated recommendations, however, product potency is not adversely affected.
Overdosage
- Ceftazidime overdosage has occurred in patients with renal failure. Reactions have included seizure activity, encephalopathy, asterixis, neuromuscular excitability, and coma. Patients who receive an acute overdosage should be carefully observed and given supportive treatment. In the presence of renal insufficiency, hemodialysis or peritoneal dialysis may aid in the removal of ceftazidime from the body.
Pharmacology
Mechanism of Action
- Ceftazidime is bactericidal in action, exerting its effect by inhibition of enzymes responsible for cell-wall synthesis. A wide range of gram-negative organisms is susceptible to ceftazidime in vitro, including strains resistant to gentamicin and other aminoglycosides. In addition, ceftazidime has been shown to be active against gram-positive organisms. It is highly stable to most clinically important beta-lactamases, plasmid or chromosomal, which are produced by both gram-negative and gram-positive organisms and, consequently, is active against many strains resistant to ampicillin and other cephalosporins.
Structure
- Ceftazidime is a semisynthetic, broad-spectrum, beta-lactam antibiotic for parenteral administration. It is the pentahydrate of pyridinium, 1-[[7-[[(2-amino-4-thiazolyl)[(1-carboxy-1-methylethoxy)imino]acetyl]amino]-2-carboxy-8-oxo-5-thia-1-azabicyclo[4.2.0]oct-2-en-3-yl]methyl]-, hydroxide, inner salt, [6R-[6α,7β(Z)]]]]. It has the following structural formula:
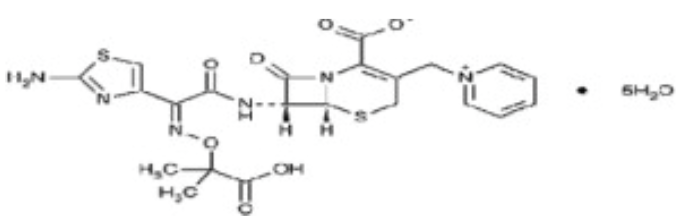
The molecular formula is C22H32N6O12S2, representing a molecular weight of 636.6.
Pharmacodynamics
There is limited information regarding Ceftazidime Pharmacodynamics in the drug label.
Pharmacokinetics
Absorption
- After IV administration of 500 mg and 1 g doses of ceftazidime over 5 minutes to normal adult male volunteers, mean peak serum concentrations of 45 and 90 mcg/mL, respectively, were achieved. After IV infusion of 500 mg, 1 g, and 2 g doses of ceftazidime over 20 to 30 minutes to normal adult male volunteers, mean peak serum concentrations of 42, 69, and 170 mcg/mL, respectively, were achieved. The average serum concentrations following IV infusion of 500 mg, 1 g, and 2 g doses to these volunteers over an 8-hour interval are given in the following table:
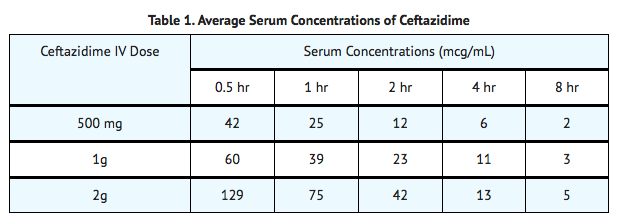
Distribution
- The absorption and elimination of ceftazidime were directly proportional to the size of the dose. The half-life following IV administration was approximately 1.9 hours. Less than 10% of ceftazidime was protein bound. The degree of protein binding was independent of concentration. There was no evidence of accumulation of ceftazidime in the serum in individuals with normal renal function following multiple IV doses of 1 and 2 g every 8 hours for 10 days.
- The presence of hepatic dysfunction had no effect on the pharmacokinetics of ceftazidime in individuals administered 2 g intravenously every 8 hours for 5 days. Therefore, a dosage adjustment from the normal recommended dosage is not required for patients with hepatic dysfunction, provided renal function is not impaired.
Excretion
- Approximately 80% to 90% of an IV dose of ceftazidime is excreted unchanged by the kidneys over a 24-hour period. After the IV administration of single 500 mg or 1 g doses, approximately 50% of the dose appeared in the urine in the first 2 hours. An additional 20% was excreted between 2 and 4 hours after dosing, and approximately another 12% of the dose appeared in the urine between 4 and 8 hours later. The elimination of ceftazidime by the kidneys resulted in high therapeutic concentrations in the urine.
- The mean renal clearance of ceftazidime was approximately 100 mL/min. The calculated plasma clearance of approximately 115 mL/min indicated nearly complete elimination of ceftazidime by the renal route. Administration of probenecid before dosing had no effect on the elimination kinetics of ceftazidime. This suggested that ceftazidime is eliminated by glomerular filtration and is not actively secreted by renal tubular mechanisms. Since ceftazidime is eliminated almost solely by the kidneys, its serum half-life is significantly prolonged in patients with impaired renal function. Consequently, dosage adjustments in such patients as described in the "Dosage and Administration" section are suggested. Therapeutic concentrations of ceftazidime are achieved in the following body tissues and fluids.
Nonclinical Toxicology
There is limited information regarding Ceftazidime Nonclinical Toxicology in the drug label.
Clinical Studies
There is limited information regarding Ceftazidime Clinical Studies in the drug label.
How Supplied
- Ceftazidime for Injection, USP is a white to cream-colored crystalline powder supplied in Pharmacy Bulk Package Bottles as follows:

Storage
- Ceftazidime for injection in the dry state should be stored at 20° to 25°C (68° to 77°F) and protected from light.
Images
Drug Images
{{#ask: Page Name::Ceftazidime |?Pill Name |?Drug Name |?Pill Ingred |?Pill Imprint |?Pill Dosage |?Pill Color |?Pill Shape |?Pill Size (mm) |?Pill Scoring |?NDC |?Drug Author |format=template |template=DrugPageImages |mainlabel=- |sort=Pill Name }}
Package and Label Display Panel
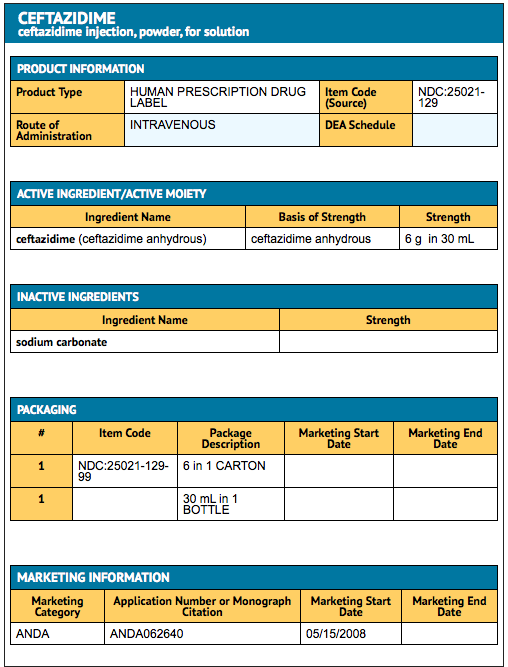
{{#ask: Label Page::Ceftazidime |?Label Name |format=template |template=DrugLabelImages |mainlabel=- |sort=Label Page }}
Patient Counseling Information
There is limited information regarding Ceftazidime Patient Counseling Information in the drug label.
Precautions with Alcohol
Alcohol-Ceftazidime interaction has not been established. Talk to your doctor about the effects of taking alcohol with this medication.
Brand Names
Look-Alike Drug Names
There is limited information regarding Ceftazidime Look-Alike Drug Names in the drug label.
Price
References
The contents of this FDA label are provided by the National Library of Medicine.
- ↑ "Therapy in febrile Neutropenia".
- ↑ Angus BJ, Smith MD, Suputtamongkol Y, Mattie H, Walsh AL, Wuthiekanun V; et al. (2000). "Pharmacokinetic-pharmacodynamic evaluation of ceftazidime continuous infusion vs intermittent bolus injection in septicaemic melioidosis". Br J Clin Pharmacol. 50 (2): 184–91. PMC 2014399. PMID 10930972.
- ↑ (PDF) http://aidsinfo.nih.gov/contentfiles/lvguidelines/oi_guidelines_pediatrics.pdf. Unknown parameter
|Title=ignored (|title=suggested) (help); Missing or empty|title=(help) - ↑ "Therapy in febrile Neutropenia".
{{#subobject:
|Label Page=Ceftazidime |Label Name=Package Ceftazidime.png
}}