Metoprolol succinate (tablet): Difference between revisions
No edit summary |
No edit summary |
||
| Line 191: | Line 191: | ||
Clinical laboratory findings may include elevated levels of serum [[transaminase]], [[alkaline phosphatase]], and [[lactate dehydrogenase]]. | Clinical laboratory findings may include elevated levels of serum [[transaminase]], [[alkaline phosphatase]], and [[lactate dehydrogenase]]. | ||
|postmarketing=The following adverse reactions have been identified during post-approval use of metoprolol succinate or immediate-release metoprolol. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure. | |postmarketing=The following adverse reactions have been identified during post-approval use of metoprolol succinate or immediate-release metoprolol. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure. | ||
* Cardiovascular: [[Cold extremities]], [[arterial insufficiency]] (usually of the [[Raynaud type]]), [[palpitations]], [[peripheral edema]], [[syncope]], [[chest pain]] and [[hypotension]]. | * Cardiovascular: [[Cold extremities]], [[arterial insufficiency]] (usually of the [[Raynaud type]]), [[palpitations]], [[peripheral edema]], [[syncope]], [[chest pain]] and [[hypotension]]. | ||
| Line 206: | Line 205: | ||
* Hematologic: [[Agranulocytosis]], [[nonthrombocytopenic purpura]], [[thrombocytopenic purpura]]. | * Hematologic: [[Agranulocytosis]], [[nonthrombocytopenic purpura]], [[thrombocytopenic purpura]]. | ||
* Hypersensitive Reactions: [[Laryngospasm]], [[respiratory distress]]. | * Hypersensitive Reactions: [[Laryngospasm]], [[respiratory distress]]. | ||
|drugInteractions= | |drugInteractions======Catecholamine Depleting Drugs===== | ||
===== | |||
( | Catecholamine depleting drugs (eg, [[reserpine]], [[monoamine oxidase inhibitors]]) may have an additive effect when given with beta-blocking agents. Observe patients treated with metoprolol succinate plus a [[catecholamine]] depletor for evidence of [[hypotension] or marked [[bradycardia]], which may produce [[vertigo]], [[syncope]], or [[orthostatic hypotension]]. | ||
===== | =====CYP2D6 Inhibitors===== | ||
Drugs that inhibit [[CYP2D6]] such as [[quinidine]], [[fluoxetine]], [[paroxetine]], and [[propafenone]] are likely to increase metoprolol concentration. In healthy subjects with [[CYP2D6]] extensive metabolizer phenotype, coadministration of [[quinidine]] 100 mg and immediate-release metoprolol 200 mg tripled the concentration of S-metoprolol and doubled the metoprolol elimination half-life. In four patients with cardiovascular disease, coadministration of [[propafenone]] 150 mg t.i.d. with immediate-release metoprolol 50 mg t.i.d. resulted in two- to five-fold increases in the steady-state concentration of metoprolol. These increases in plasma concentration would decrease the cardioselectivity of metoprolol. | |||
===== | =====Digitalis, Clonidine, and Calcium Channel Blockers===== | ||
[[Digitalis glycosides]], [[clonidine]], [[diltiazem]] and [[verapamil]] slow atrioventricular conduction and decrease [[heart rate]]. Concomitant use with [[beta blockers]] can increase the risk of [[bradycardia]]. | |||
If [[clonidine]] and a [[beta blocker]], such as metoprolol are coadministered, withdraw the [[beta-blocker]] several days before the gradual withdrawal of [[clonidine]] because [[beta-blockers]] may exacerbate the rebound [[hypertension]] that can follow the withdrawal of [[clonidine]]. If replacing [[clonidine]] by [[beta-blocker]] therapy, delay the introduction of [[beta-blockers]] for several days after [[clonidine]] administration has stopped | |||
|useInPregnancyFDA=(Description) | |useInPregnancyFDA=(Description) | ||
|useInPregnancyAUS=(Description) | |useInPregnancyAUS=(Description) | ||
Revision as of 14:15, 3 July 2014
{{DrugProjectFormSinglePage |authorTag=Alonso Alvarado, M.D. [1] |genericName=Metoprolol succinate |aOrAn=a |drugClass=beta-adrenergic blocker |indication=hypertension, angina pectoris, heart failure |hasBlackBoxWarning=Yes |adverseReactions=bradyarrhythmia, heart failure, hypotension, diarrhea, nausea, dizziness, fatigue, headache, depression, dyspnea, wheezing |blackBoxWarningTitle=WARNING |blackBoxWarningBody=ISCHEMIC HEART DISEASE Following abrupt cessation of therapy with certain beta-blocking agents, exacerbations of angina pectoris and, in some cases, myocardial infarction have occurred. When discontinuing chronically administered metoprolol succinate, particularly in patients with ischemic heart disease, the dosage should be gradually reduced over a period of 1 - 2 weeks and the patient should be carefully monitored. If angina markedly worsens or acute coronary insufficiency develops, metoprolol succinate administration should be reinstated promptly, at least temporarily, and other measures appropriate for the management of unstable angina should be taken. Warn patients against interruption or discontinuation of therapy without the physician’s advice. Because coronary artery disease is common and may be unrecognized, it may be prudent not to discontinue metoprolol succinate therapy abruptly even in patients treated only for hypertension |fdaLIADAdult======Hypertension=====
- Dosing Information
- The initial dosage is 25 to 100 mg daily in a single dose. The dosage may be increased at weekly (or longer) intervals until optimum blood pressure reduction is achieved. In general, the maximum effect of any given dosage level will be apparent after 1 week of therapy.
- Dosages above 400 mg per day have not been studied.
Angina Pectoris
- Dosing Information
- Individualize the dosage of metoprolol succinate. The usual initial dosage is 100 mg daily, given in a single dose. Gradually increase the dosage at weekly intervals until optimum clinical response has been obtained or there is a pronounced slowing of the heart rate.
- Dosages above 400 mg per day have not been studied. If treatment is to be discontinued, reduce the dosage gradually over a period of 1 - 2 weeks
Heart Failure
- Dosing information
- Dosage must be individualized and closely monitored during up-titration. Prior to initiation of metoprolol succinate, stabilize the dose of other heart failure drug therapy.
- The recommended starting dose of metoprolol succinate is 25 mg once daily for two weeks in patients with NYHA Class II heart failure and 12.5 mg once daily in patients with more severe heart failure. Double the dose every two weeks to the highest dosage level tolerated by the patient or up to 200 mg of metoprolol succinate.
- Initial difficulty with titration should not preclude later attempts to introduce metoprolol succinate. If patients experience symptomatic bradycardia, reduce the dose of metoprolol succinate. If transient worsening of heart failure occurs, consider treating with increased doses of diuretics, lowering the dose of metoprolol succinate or temporarily discontinuing it.
- The dose of metoprolol succinate should not be increased until symptoms of worsening heart failure have been stabilized.
|offLabelAdultGuideSupport======Condition 1=====
- Developed by: (Organisation)
- Class of Recommendation: (Class) (Link)
- Strength of Evidence: (Category A/B/C) (Link)
- Dosing Information/Recommendation
- (Dosage)
Condition 2
- Developed by: (Organisation)
- Class of Recommendation: (Class) (Link)
- Strength of Evidence: (Category A/B/C) (Link)
- Dosing Information/Recommendation
- (Dosage)
|offLabelAdultNoGuideSupport======Condition 1=====
- Dosing Information
- (Dosage)
Condition 2
- Dosing Information
- (Dosage)
Condition 3
- Dosing Information
- (Dosage)
|fdaLIADPed======Pediatric Hypertensive Patients ≥ 6 Years of Age=====
- Dosing Information
- A pediatric clinical hypertension study in patients 6 to 16 years of age did not meet its primary endpoint (dose response for reduction in SBP); however some other endpoints demonstrated effectiveness.
- If selected for treatment, the recommended starting dose of metoprolol succinate is 1 mg/kg once daily, but the maximum initial dose should not exceed 50 mg once daily. Dosage should be adjusted according to blood pressure response.
- Doses above 2 mg/kg (or in excess of 200 mg) once daily have not been studied in pediatric patients.
|offLabelPedGuideSupport======Condition 1=====
- Developed by: (Organisation)
- Class of Recommendation: (Class) (Link)
- Strength of Evidence: (Category A/B/C) (Link)
- Dosing Information/Recommendation
- (Dosage)
Condition 2
- Developed by: (Organisation)
- Class of Recommendation: (Class) (Link)
- Strength of Evidence: (Category A/B/C) (Link)
- Dosing Information/Recommendation
- (Dosage)
|offLabelPedNoGuideSupport======Condition 1=====
- Dosing Information
- (Dosage)
Condition 2
- Dosing Information
- (Dosage)
Condition 3
- Dosing Information
- (Dosage)
|contraindications=Metoprolol succinate is contraindicated in severe bradycardia, second degree heart block and third degree heart block, cardiogenic shock, decompensated cardiac failure, sick sinus syndrome (unless a permanent pacemaker is in place), and in patients who are hypersensitive to any component of this product. |warnings======Ischemic Heart Disease=====
Following abrupt cessation of therapy with certain beta-blocking agents, exacerbations of angina pectoris and, in some cases, myocardial infarction have occurred. When discontinuing chronically administered metoprolol succinate, particularly in patients with ischemic heart disease, gradually reduce the dosage over a period of 1 - 2 weeks and monitor the patient. If angina markedly worsens or acute coronary ischemia develops, promptly reinstate metoprolol succinate, and take measures appropriate for the management of unstable angina. Warn patients not to interrupt therapy without their physician’s advice. Because coronary artery disease is common and may be unrecognized, avoid abruptly discontinuing metoprolol succinate in patients treated only for hypertension.
Heart Failure
Worsening cardiac failure may occur during up-titration of metoprolol succinate. If such symptoms occur, increase diuretics and restore clinical stability before advancing the dose of metoprolol succinate. It may be necessary to lower the dose of metoprolol succinate or temporarily discontinue it. Such episodes do not preclude subsequent successful titration of metoprolol succinate.
Bronchospastic Disease
PATIENTS WITH BRONCHOSPASTIC DISEASES SHOULD, IN GENERAL, NOT RECEIVE BETA-BLOCKERS. Because of its relative beta1 cardio-selectivity, however, metoprolol succinate may be used in patients with bronchospastic disease who do not respond to, or cannot tolerate, other antihypertensive treatment. Because beta1-selectivity is not absolute, use the lowest possible dose of metoprolol succinate. Bronchodilators, including beta2-agonists, should be readily available or administered concomitantly.
Pheochromocytoma
If metoprolol succinate is used in the setting of pheochromocytoma, it should be given in combination with an alpha blocker, and only after the alpha blocker has been initiated. Administration of beta-blockers alone in the setting of pheochromocytoma has been associated with a paradoxical increase in blood pressure due to the attenuation of beta-mediated vasodilatation in skeletal muscle.
Major Surgery
Avoid initiation of a high-dose regimen of extended-release metoprolol in patients undergoing non-cardiac surgery, since such use in patients with cardiovascular risk factors has been associated with bradycardia, hypotension, stroke and death. Chronically administered beta-blocking therapy should not be routinely withdrawn prior to major surgery, however, the impaired ability of the heart to respond to reflex adrenergic stimuli may augment the risks of general anesthesia and surgical procedures.
Diabetes and Hypoglycemia
Beta-blockers may mask tachycardia occurring with hypoglycemia, but other manifestations such as dizziness and sweating may not be significantly affected.
Hepatic Impairment
Consider initiating metoprolol succinate therapy at doses lower than those recommended for a given indication; gradually increase dosage to optimize therapy, while monitoring closely for adverse events.
Thyrotoxicosis
Beta-adrenergic blockade may mask certain clinical signs of hyperthyroidism, such as tachycardia. Abrupt withdrawal of beta-blockade may precipitate a thyroid storm.
Anaphylactic Reaction
While taking beta-blockers, patients with a history of severe anaphylactic reactions to a variety of allergens may be more reactive to repeated challenge and may be unresponsive to the usual doses of epinephrine used to treat an allergic reaction.
Peripheral Vascular Disease
Beta-blockers can precipitate or aggravate symptoms of arterial insufficiency in patients with peripheral vascular disease.
Calcium Channel Blockers
Because of significant inotropic and chronotropic effects in patients treated with beta-blockers and calcium channel blockers of the verapamil and diltiazem type, caution should be exercised in patients treated with these agents concomitantly. |clinicalTrials=The following adverse reactions are described elsewhere in labeling:
- Worsening angina or myocardial infarction.
- Worsening heart failure.
- Worsening AV block.
Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice. The adverse reaction information from clinical trials does, however, provide a basis for identifying the adverse events that appear to be related to drug use and for approximating rates.
Hypertension and Angina: Most adverse reactions have been mild and transient. The most common (>2%) adverse reactions are tiredness, dizziness, depression, diarrhea, shortness of breath, bradycardia, and rash.
Heart Failure
In the MERIT-HF study comparing metoprolol succinate in daily doses up to 200 mg (mean dose 159 mg once-daily; n=1990) to placebo (n=2001), 10.3% of metoprolol succinate patients discontinued for adverse reactions vs. 12.2% of placebo patients.
The table below lists adverse reactions in the MERIT-HF study that occurred at an incidence of ≥ 1% in the metoprolol succinate group and greater than placebo by more than 0.5%, regardless of the assessment of causality.
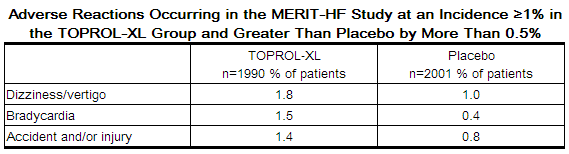
Post-operative Adverse Events
In a randomized, double-blind, placebo-controlled trial of 8351 patients with or at risk for atherosclerotic disease undergoing non-vascular surgery and who were not taking beta–blocker therapy, metoprolol succinate 100 mg was started 2 to 4 hours prior to surgery then continued for 30 days at 200 mg per day. metoprolol succinate use was associated with a higher incidence of bradycardia (6.6% vs. 2.4%; HR, 2.74; 95% CI 2.19, 3.43), hypotension (15% vs. 9.7%; HR 1.55; 95% CI 1.37, 1.74), stroke (1.0% vs. 0.5%; HR 2.17; 95% CI 1.26, 3.74) and death (3.1% vs. 2.3%; HR 1.33; 95% CI 1.03, 1.74) compared to placebo.
Laboratory Test Findings
Clinical laboratory findings may include elevated levels of serum transaminase, alkaline phosphatase, and lactate dehydrogenase. |postmarketing=The following adverse reactions have been identified during post-approval use of metoprolol succinate or immediate-release metoprolol. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
- Cardiovascular: Cold extremities, arterial insufficiency (usually of the Raynaud type), palpitations, peripheral edema, syncope, chest pain and hypotension.
- Respiratory: Wheezing (bronchospasm), dyspnea.
- Central Nervous System: Confusion, short-term memory loss, headache, somnolence, nightmares, insomnia, anxiety/nervousness, hallucinations, paresthesia.
- Gastrointestinal: Nausea, dry mouth, constipation, flatulence, heartburn, hepatitis, vomiting.
- Hypersensitive Reactions: Pruritus.
- Miscellaneous: Musculoskeletal pain, arthralgia, blurred vision, decreased libido, male impotence, tinnitus, reversible alopecia, agranulocytosis, dry eyes, worsening of psoriasis, Peyronie’s disease, sweating, photosensitivity, taste disturbance.
Potential Adverse Reactions
- In addition, there are adverse reactions not listed above that have been reported with other beta-adrenergic blocking agents and should be considered potential adverse reactions to metoprolol succinate.
- Central Nervous System: Reversible mental depression progressing to catatonia; an acute reversible syndrome characterized by disorientation for time and place, short-term memory loss, emotional lability, clouded sensorium, and decreased performance on neuropsychometrics.
- Hematologic: Agranulocytosis, nonthrombocytopenic purpura, thrombocytopenic purpura.
- Hypersensitive Reactions: Laryngospasm, respiratory distress.
|drugInteractions======Catecholamine Depleting Drugs=====
Catecholamine depleting drugs (eg, reserpine, monoamine oxidase inhibitors) may have an additive effect when given with beta-blocking agents. Observe patients treated with metoprolol succinate plus a catecholamine depletor for evidence of [[hypotension] or marked bradycardia, which may produce vertigo, syncope, or orthostatic hypotension.
CYP2D6 Inhibitors
Drugs that inhibit CYP2D6 such as quinidine, fluoxetine, paroxetine, and propafenone are likely to increase metoprolol concentration. In healthy subjects with CYP2D6 extensive metabolizer phenotype, coadministration of quinidine 100 mg and immediate-release metoprolol 200 mg tripled the concentration of S-metoprolol and doubled the metoprolol elimination half-life. In four patients with cardiovascular disease, coadministration of propafenone 150 mg t.i.d. with immediate-release metoprolol 50 mg t.i.d. resulted in two- to five-fold increases in the steady-state concentration of metoprolol. These increases in plasma concentration would decrease the cardioselectivity of metoprolol.
Digitalis, Clonidine, and Calcium Channel Blockers
Digitalis glycosides, clonidine, diltiazem and verapamil slow atrioventricular conduction and decrease heart rate. Concomitant use with beta blockers can increase the risk of bradycardia.
If clonidine and a beta blocker, such as metoprolol are coadministered, withdraw the beta-blocker several days before the gradual withdrawal of clonidine because beta-blockers may exacerbate the rebound hypertension that can follow the withdrawal of clonidine. If replacing clonidine by beta-blocker therapy, delay the introduction of beta-blockers for several days after clonidine administration has stopped
|useInPregnancyFDA=(Description) |useInPregnancyAUS=(Description) |useInLaborDelivery=(Description) |useInNursing=(Description) |useInPed=(Description) |useInGeri=(Description) |useInGender=(Description) |useInRace=(Description) |useInRenalImpair=(Description) |useInHepaticImpair=(Description) |useInReproPotential=(Description) |useInImmunocomp=(Description) |othersTitle=Others |useInOthers=(Description) |administration=(Oral/Intravenous/etc) |monitoring======Condition 1=====
(Description regarding monitoring, from Warnings section)
Condition 2
(Description regarding monitoring, from Warnings section)
Condition 3
(Description regarding monitoring, from Warnings section) |IVCompat====Solution===
Compatible
- Solution 1
- Solution 2
- Solution 3
Not Tested
- Solution 1
- Solution 2
- Solution 3
Variable
- Solution 1
- Solution 2
- Solution 3
Incompatible
- Solution 1
- Solution 2
- Solution 3
Y-Site
Compatible
- Solution 1
- Solution 2
- Solution 3
Not Tested
- Solution 1
- Solution 2
- Solution 3
Variable
- Solution 1
- Solution 2
- Solution 3
Incompatible
- Solution 1
- Solution 2
- Solution 3
Admixture
Compatible
- Solution 1
- Solution 2
- Solution 3
Not Tested
- Solution 1
- Solution 2
- Solution 3
Variable
- Solution 1
- Solution 2
- Solution 3
Incompatible
- Solution 1
- Solution 2
- Solution 3
Syringe
Compatible
- Solution 1
- Solution 2
- Solution 3
Not Tested
- Solution 1
- Solution 2
- Solution 3
Variable
- Solution 1
- Solution 2
- Solution 3
Incompatible
- Solution 1
- Solution 2
- Solution 3
TPN/TNA
Compatible
- Solution 1
- Solution 2
- Solution 3
Not Tested
- Solution 1
- Solution 2
- Solution 3
Variable
- Solution 1
- Solution 2
- Solution 3
Incompatible
- Solution 1
- Solution 2
- Solution 3
|overdose====Acute Overdose===
Signs and Symptoms
(Description)
Management
(Description)
Chronic Overdose
Signs and Symptoms
(Description)
Management
(Description) |drugBox=
Metoprolol succinate (tablet)
| |
| Systematic (IUPAC) name | |
| ? | |
| Identifiers | |
| CAS number | ? |
| ATC code | ? |
| PubChem | ? |
| Chemical data | |
| Formula | ? |
| Mol. mass | ? |
| Pharmacokinetic data | |
| Bioavailability | ? |
| Metabolism | ? |
| Half life | ? |
| Excretion | ? |
| Therapeutic considerations | |
| Pregnancy cat. |
? |
| Legal status | |
| Routes | ? |
|mechAction=(Description) |structure=(Description with picture) |PD=(Description) |PK=(Description) |nonClinToxic=(Description) |clinicalStudies======Condition 1=====
(Description)
Condition 2
(Description)
Condition 3
(Description) |howSupplied=(Description) |fdaPatientInfo=(Patient Counseling Information) |alcohol=Alcohol-Metoprolol succinate interaction has not been established. Talk to your doctor about the effects of taking alcohol with this medication. |lookAlike=* (Paired Confused Name 1a) — (Paired Confused Name 1b)
- (Paired Confused Name 2a) — (Paired Confused Name 2b)
- (Paired Confused Name 3a) — (Paired Confused Name 3b)
|nlmPatientInfo=(Link to patient information page) |drugShortage=Drug Shortage }}