Eribulin
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]; Associate Editor(s)-in-Chief: Alberto Plate [2]
Disclaimer
WikiDoc MAKES NO GUARANTEE OF VALIDITY. WikiDoc is not a professional health care provider, nor is it a suitable replacement for a licensed healthcare provider. WikiDoc is intended to be an educational tool, not a tool for any form of healthcare delivery. The educational content on WikiDoc drug pages is based upon the FDA package insert, National Library of Medicine content and practice guidelines / consensus statements. WikiDoc does not promote the administration of any medication or device that is not consistent with its labeling. Please read our full disclaimer here.
Overview
Eribulin is an antineoplastic agent and mitotic Inhibitor that is FDA approved for the treatment of patients with metastatic breast cancer who have previously received at least two chemotherapeutic regimens for the treatment of metastatic disease. Prior therapy should have included an anthracycline and a taxane in either the adjuvant or metastatic setting. Common adverse reactions include alopecia, Weight loss, constipation, anorexia, nausea, anemia, neutropenia, ALT/SGPT, arthralgia, myalgia, asthenia, headache, peripheral neuropathy, fatigue, fever.
Adult Indications and Dosage
FDA-Labeled Indications and Dosage (Adult)
Recommended Dose
The recommended dose of Eribulin is 1.4 mg/m2 administered intravenously over 2 to 5 minutes on Days 1 and 8 of a 21-day cycle.
Dose Modification
Assess for peripheral neuropathy and obtain complete blood cell counts prior to each dose.
Recommended dose delays
- Do not administer Eribulin on Day 1 or Day 8 for any of the following:
- The Day 8 dose may be delayed for a maximum of 1 week.
- If toxicities do not resolve or improve to ≤ Grade 2 severity by Day 15, omit the dose.
- If toxicities resolve or improve to ≤ Grade 2 severity by Day 15, administer Eribulin at a reduced dose and initiate the next cycle no sooner than 2 weeks later.
Recommended dose reductions
If a dose has been delayed for toxicity and toxicities have recovered to Grade 2 severity or less, resume Eribulin at a reduced dose as set out in Table 1. Do not re-escalate Eribulin dose after it has been reduced.

Instructions for Preparation and Administration
- Aseptically withdraw the required amount of Eribulin from the single-use vial and administer undiluted or diluted in 100 mL of 0.9% Sodium Chloride Injection, USP.
- Do not dilute in or administer through an intravenous line containing solutions with dextrose. Do not administer in the same intravenous line concurrent with the other medicinal products.
- Store undiluted Eribulin in the syringe for up to 4 hours at room temperature or for up to 24 hours under refrigeration (40°F or/ 4°C). Store diluted solutions of Eribulin for up to 4 hours at room temperature or up to 24 hours under refrigeration.
- Discard unused portions of the vial.
Off-Label Use and Dosage (Adult)
Guideline-Supported Use
There is limited information regarding Off-Label Guideline-Supported Use of Eribulin in adult patients.
Non–Guideline-Supported Use
There is limited information regarding Off-Label Non–Guideline-Supported Use of Eribulin in adult patients.
Pediatric Indications and Dosage
FDA-Labeled Indications and Dosage (Pediatric)
There is limited information regarding Eribulin FDA-Labeled Indications and Dosage (Pediatric) in the drug label.
Off-Label Use and Dosage (Pediatric)
Guideline-Supported Use
There is limited information regarding Off-Label Guideline-Supported Use of Eribulin in pediatric patients.
Non–Guideline-Supported Use
There is limited information regarding Off-Label Non–Guideline-Supported Use of Eribulin in pediatric patients.
Contraindications
None
Warnings
Neutropenia
Severe neutropenia (ANC < 500/mm3) lasting more than one week occurred in 12% (62/503) of patients in Study 1, leading to discontinuation in <1% of patients. Patients with alanine aminotransferase or aspartate aminotransferase > 3 × ULN (upper limit of normal) experienced a higher incidence of Grade 4 neutropenia and febrile neutropenia than patients with normal aminotransferase levels. Patients with bilirubin > 1.5 × ULN also had a higher incidence of Grade 4 neutropenia and febrile neutropenia.
Monitor complete blood counts prior to each dose; increase the frequency of monitoring in patients who develop Grade 3 or 4 cytopenias. Delay administration of Eribulin and reduce subsequent doses in patients who experience febrile neutropenia or Grade 4 neutropenia lasting longer than 7 days. Clinical studies of Eribulin did not include patients with baseline neutrophil counts below 1,500/mm3.
Peripheral Neuropathy
Grade 3 peripheral neuropathy occurred in 8% (40/503) of patients, and Grade 4 in 0.4% (2/503) of patients in Study 1. Peripheral neuropathy was the most common toxicity leading to discontinuation of Eribulin (5% of patients; 24/503). Neuropathy lasting more than one year occurred in 5% (26/503) of patients. Twenty-two percent (109/503) of patients developed a new or worsening neuropathy that had not recovered within a median follow-up duration of 269 days (range 25-662 days). Monitor patients closely for signs of peripheral motor neuropathy and sensory neuropathy. Withhold Eribulin in patients who experience Grade 3 or 4 peripheral neuropathy until resolution to Grade 2 or less.
Embryo-Fetal Toxicity
There are no adequate and well-controlled studies of Eribulin in pregnant women. Eribulin is a microtubule inhibitor; therefore, it is expected to cause fetal harm when administered to a pregnant woman. Embryo-fetal toxicity and teratogenicity occurred in rats that received eribulin mesylate at approximately half of the recommended human dose based on body surface area. If this drug is used during pregnancy, or if a patient becomes pregnant while taking this drug, she should be apprised of the potential hazard to the fetus.
Prolongation
In an uncontrolled open-label ECG study in 26 patients, QT prolongation was observed on Day 8, independent of eribulin concentration, with no QT prolongation observed on Day 1. ECG monitoring is recommended if therapy is initiated in patients with congestive heart failure, bradyarrhythmias, drugs known to prolong the QT interval, including Class Ia antiarrhythmics and Class III antiarrhythmics, and electrolyte abnormalities. Correct hypokalemia or hypomagnesemia prior to initiating Eribulin and monitor these electrolytes periodically during therapy. Avoid Eribulin in patients with congenital long QT syndrome.
Adverse Reactions
Clinical Trials Experience
The following adverse reactions are discussed in detail in other sections of the labeling:
The most common adverse reactions (≥25%) reported in patients receiving Eribulin were neutropenia, anemia, asthenia/fatigue, alopecia, peripheral neuropathy, nausea, and constipation. The most common serious adverse reactions reported in patients receiving Eribulin were febrile neutropenia (4%) and neutropenia (2%). The most common adverse reaction resulting in discontinuation of Eribulin was peripheral neuropathy (5%).
Because clinical trials are conducted under widely varying conditions, the adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in other clinical trials and may not reflect the rates observed in clinical practice.
In clinical trials, Eribulin has been administered to 1,222 patients with multiple tumor types, including 240 patients exposed to Eribulin for 6 months or longer. The majority of the 1,222 patients were women (82%) with a median age of 58 years (range: 26 to 91 years). The racial and ethnic distribution was Caucasian (83%), Black (5%), Asian (2%), and other (5%).
The adverse reactions described in Table 2 were identified in 750 patients treated in Study 1. In Study 1, patients were randomized (2:1) to receive either Eribulin (1.4 mg/m2 on Days 1 and 8 of a 21-day cycle) or single agent treatment chosen by their physician (control group). A total of 503 patients received Eribulin and 247 patients in the control group received therapy consisting of chemotherapy [total 97% (anthracyclines 10%, capecitabine 18%, gemcitabine 19%, taxanes 15%, vinorelbine 25%, other chemotherapies 10%)] or hormonal therapy (3%). The median duration of exposure was 118 days for patients receiving Eribulin and 63 days for patients receiving control therapy. Table 2 reports the most common adverse reactions occurring in at least 10% of patients in either group.

Cytopenias
Grade 3 neutropenia occurred in 28% (143/503) of patients who received Eribulin in Study 1, and 29% (144/503) of patients experienced Grade 4 neutropenia. Febrile neutropenia occurred in 5% (23/503) of patients; two patients (0.4%) died from complications of febrile neutropenia. Dose reduction due to neutropenia was required in 12% (62/503) of patients and discontinuation was required in <1% of patients. The mean time to nadir was 13 days and the mean time to recovery from severe neutropenia (<500/mm3) was 8 days. Grade 3 or greater thrombocytopenia occurred in 1% (7/503) of patients. G-CSF (granulocyte colony-stimulating factor) or GM-CSF (granulocyte–macrophage colony-stimulating factor) was used in 19% of patients who received Eribulin.
Peripheral Neuropathy
In Study 1, 17 % of enrolled patients had Grade 1 peripheral neuropathy and 3% of patients had Grade 2 peripheral neuropathy at baseline. Dose reduction due to peripheral neuropathy was required by 3% (14/503) of patients who received Eribulin. Four percent (20/503) of patients experienced peripheral motor neuropathy of any grade and 2% (8/503) of patients developed Grade 3 peripheral motor neuropathy.
Liver Function Test Abnormalities
Among patients with Grade 0 or 1 ALT levels at baseline, 18% of Eribulin-treated patients experienced Grade 2 or greater ALT elevation. One Eribulin-treated patient without documented liver metastases had concomitant Grade 2 elevations in bilirubin and ALT; these abnormalities resolved and did not recur with re-exposure to Eribulin.
Less Common Adverse Reactions
The following additional adverse reactions were reported in ≥5% to <10% of the Eribulin-treated group:
- Eye Disorders: increased lacrimation
- Gastrointestinal Disorders: dyspepsia, abdominal pain, stomatitis, dry mouth
- General Disorders and Administration Site Conditions: peripheral edema
- Infections and Infestations: upper respiratory tract infection
- Metabolism and Nutrition Disorders: hypokalemia
- Musculoskeletal and Connective Tissue Disorders: muscle spasms, muscular weakness
- Nervous System Disorders: dysgeusia, dizziness
- Psychiatric Disorders: insomnia, depression
- Skin and Subcutaneous Tissue Disorders: rash
Postmarketing Experience
The following adverse drug reactions have been identified during post-approval of Eribulin Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
- Blood and Lymphatic System Disorders: lymphopenia
- Gastrointestinal Disorders: pancreatitis
- Hepatobiliary Disorders: hepatotoxicity
- Immune System Disorders: drug hypersensitivity
- Infections and Infestations: pneumonia, sepsis/neutropenic sepsis
- Metabolism and Nutrition Disorders: hypomagnesemia, dehydration
- Respiratory, thoracic and mediastinal disorders: interstitial lung disease
- Skin and Subcutaneous Tissue Disorders: pruritus
Drug Interactions
Effects of Other Drugs on Eribulin
No drug-drug interactions are expected with CYP3A4 inhibitors, CYP3A4 inducers or P-glycoprotein (P-gp) inhibitors. Clinically meaningful differences in exposure (AUC) were not observed in patients with advanced solid tumors when Eribulin was administered with or without ketoconazole (a strong inhibitor of CYP3A4 and a P-gp inhibitor) and when Eribulin was administered with or without rifampin (a CYP3A4 inducer).
Effects of Eribulin on Other Drugs
Eribulin does not inhibit CYP1A2, CYP2C9, CYP2C19, CYP2D6, CYP2E1 or CYP3A4 enzymes or induce CYP1A2, CYP2C9, CYP2C19 or CYP3A4 enzymes at relevant clinical concentrations. Eribulin is not expected to alter the plasma concentrations of drugs that are substrates of these enzymes.
Use in Specific Populations
Pregnancy
Pregnancy Category (FDA): D There are no adequate and well-controlled studies with HALAVEN in pregnant women. HALAVEN is a microtubule inhibitor, therefore, it is expected to cause fetal harm when administered to a pregnant woman. Embryo-fetal toxicity and teratogenicity occurred in rats that received eribulin mesylate at approximately half of the recommended human dose based on body surface area. If this drug is used during pregnancy, or if the patient becomes pregnant while taking this drug, the patient should be apprised of the potential hazard to the fetus.
In a developmental toxicity study, pregnant rats received intravenous infusion of eribulin mesylate during organogenesis (Gestation Days 8, 10, and 12) at doses approximately 0.04, 0.13, 0.43 and 0.64 times the recommended human dose, based on body surface area (mg/m2). Increased abortion and severe external or soft tissue malformations were observed in offspring at doses 0.64 times the recommended human dose based on body surface area (mg/m2), including the absence of a lower jaw, tongue, stomach and spleen. Increased embryo-fetal death/resorption, reduced fetal weights, and minor skeletal anomalies consistent with developmental delay were also reported at or above doses of 0.43 times the recommended human dose.
Maternal toxicity of eribulin mesylate was reported in rats at or above doses of 0.43 times the recommended human dose (mg/m²), and included enlarged spleen, reduced maternal weight gain and decreased food consumption.
Pregnancy Category (AUS):
There is no Australian Drug Evaluation Committee (ADEC) guidance on usage of Eribulin in women who are pregnant.
Labor and Delivery
There is no FDA guidance on use of Eribulin during labor and delivery.
Nursing Mothers
It is not known whether HALAVEN is excreted into human milk. No studies in humans or animals were conducted to determine if HALAVEN is excreted into milk. Because many drugs are excreted into human milk and because of the potential for serious adverse reactions in human milk fed infants from HALAVEN, a decision should be made whether to discontinue nursing or to discontinue HALAVEN taking into account the importance of the drug to the mother.
Pediatric Use
The safety and effectiveness of HALAVEN in pediatric patients below the age of 18 years have not been established.
Geriatic Use
Study 1 did not include sufficient numbers of subjects aged 65 years and older to determine whether they respond differently from younger subjects. Of the 827 subjects who received the recommended dose and schedule of HALAVEN in clinical studies, 15% (121/827) were 65 and older, and 2% (17/827) patients were 75 and older. No overall differences in safety were observed between these subjects and younger subjects.
Gender
There is no FDA guidance on the use of Eribulin with respect to specific gender populations.
Race
There is no FDA guidance on the use of Eribulin with respect to specific racial populations.
Renal Impairment
For patients with moderate renal impairment (CrCl 30-50 mL/min), the geometric mean dose-normalized systemic exposure increased 2-fold compared to patients with normal renal function. A lower starting dose of 1.1 mg/m2 is recommended for patients with moderate renal impairment. The safety of HALAVEN was not studied in patients with severe renal impairment (CrCl < 30 mL/min)
Hepatic Impairment
Administration of HALAVEN at a dose of 1.1 mg/m2 to patients with mild hepatic impairment and 0.7 mg/m2 to patients with moderate hepatic impairment resulted in similar exposure to eribulin as a dose of 1.4 mg/m2 to patients with normal hepatic function. Therefore, a lower starting dose of 1.1 mg/m2 is recommended for patients with mild hepatic impairment (Child-Pugh A) and of 0.7 mg/m2 is recommended for patients with moderate hepatic impairment (Child-Pugh B). HALAVEN was not studied in patients with severe hepatic impairment (Child-Pugh C)
Females of Reproductive Potential and Males
There is no FDA guidance on the use of Eribulin in women of reproductive potentials and males.
Immunocompromised Patients
There is no FDA guidance one the use of Eribulin in patients who are immunocompromised.
Administration and Monitoring
Administration
There is limited information regarding Eribulin Administration in the drug label.
Monitoring
There is limited information regarding Eribulin Monitoring in the drug label.
IV Compatibility
There is limited information regarding the compatibility of Eribulin and IV administrations.
Overdosage
Overdosage of Eribulin has been reported at approximately 4 times the recommended dose, which resulted in Grade 3 neutropenia lasting seven days and a Grade 3 hypersensitivity reaction lasting one day. There is no known antidote for Eribulin overdose.
Pharmacology
Mechanism of Action
Eribulin inhibits the growth phase of microtubules without affecting the shortening phase and sequesters tubulin into nonproductive aggregates. Eribulin exerts its effects via a tubulin-based antimitotic mechanism leading to G2/M cell-cycle block, disruption of mitotic spindles, and, ultimately, apoptotic cell death after prolonged mitotic blockage.
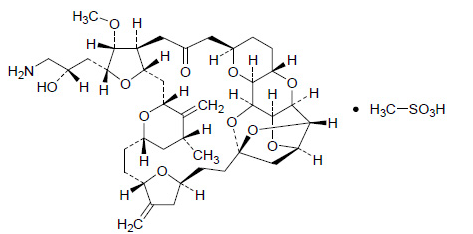
Structure
It has a molecular weight of 826.0 (729.9 for free base). The empirical formula is C40H59NO11•CH4O3S. Eribulin mesylate has the following structural formula:
Pharmacodynamics
There is limited information regarding Eribulin Pharmacodynamics in the drug label.
Pharmacokinetics
The pharmacokinetics (PK) of eribulin is linear with a mean elimination half-life of approximately 40 hours, a mean volume of distribution of 43 L/m2 to 114 L/m2 and mean clearance of 1.16 L/hr/m2 to 2.42 L/hr/m2 over the dose range of 0.25 mg/m2 to 4.0 mg/m2. The human plasma protein binding of eribulin at concentrations of 100 ng/mL to 1,000 ng/mL ranges from 49% to 65% Eribulin exposure after multiple dosing is comparable to that following a single dose. No accumulation of eribulin is observed with weekly administration.
Metabolism
Unchanged eribulin was the major circulating species in plasma following administration of 14C‑eribulin to patients. Metabolite concentrations represented <0.6% of parent compound, confirming that there are no major human metabolites of eribulin. Cytochrome P450 3A4 (CYP3A4) negligibly metabolizes eribulin in vitro.
Elimination
Eribulin is eliminated primarily in feces unchanged. After administration of 14C-eribulin to patients, approximately 82% of the dose was eliminated in feces and 9% in urine. Unchanged eribulin accounted for approximately 88% and 91% of the dose in feces and urine, respectively.
Nonclinical Toxicology
Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenicity studies have not been conducted with eribulin mesylate.
Eribulin mesylate was not mutagenic in in vitro bacterial reverse mutation assays (Ames test). Eribulin mesylate was positive in mouse lymphoma mutagenesis assays, and was clastogenic in an in vivo rat bone marrow micronucleus assay.
The effects of Eribulin on human fertility are unknown. Fertility studies have not been conducted with eribulin mesylate in humans or animals. However, nonclinical findings in repeated-dose dog and rat toxicology studies suggest that male fertility may be compromised by treatment with eribulin mesylate. Rats exhibited testicular toxicity (hypocellularity of seminiferous epithelium with hypospermia/aspermia) following dosing with eribulin mesylate at or above 0.43 times the recommended human dose (mg/m2) given once weekly for 3 weeks, or at or above 0.21 times the recommended human dose (mg/m2) given once weekly for 3 out of 5 weeks, repeated for 6 cycles. Testicular toxicity was also observed in dogs given 0.64 times the recommended human dose (mg/m2) weekly for 3 out of 5 weeks, repeated for 6 cycles.
Clinical Studies
Study 1 was an open-label, randomized, multicenter trial of 762 patients with metastatic breast cancer who received at least two chemotherapeutic regimens for the treatment of metastatic disease and experienced disease progression within 6 months of their last chemotherapeutic regimen. Patients were required to receive prior anthracycline- and taxane- based chemotherapy for adjuvant or metastatic disease. Patients were randomized (2:1) to receive Eribulin (n=508) or a single agent therapy selected prior to randomization (control arm, n=254). Randomization was stratified by geographic region, HER2/neu status, and prior capecitabine exposure. Eribulin was administered at a dose of 1.4 mg/m2 on Days 1 and 8 of a 21-day cycle. Eribulin-treated patients received a median of 5 cycles (range: 1 to 23 cycles) of therapy. Control arm therapy consisted of 97% chemotherapy (26% vinorelbine, 18% gemcitabine, 18% capecitabine, 16% taxane, 9% anthracycline, 10% other chemotherapy), and 3% hormonal therapy. The main efficacy outcome was overall survival.
Patient demographic and baseline characteristics were comparable between the treatment arms. The median age was 55 (range: 27 to 85 years) and 92% were White. Sixty-four percent of patients were enrolled in North America/Western Europe/Australia, 25% in Eastern Europe/Russia, and 11% in Latin America/South Africa. Ninety-one percent of patients had a baseline ECOG performance status of 0 or 1. Tumor prognostic characteristics, including estrogen receptor status (positive: 67%, negative: 28%), progesterone receptor status (positive: 49%, negative: 39%), HER2/neu receptor status (positive: 16%, negative: 74%), triple negative status (ER-,PR-,HER2/neu-: 19%), presence of visceral disease (82%, including 60% liver and 38% lung) and bone disease (61%), and number of sites of metastases (greater than two: 50%), were also similar in the Eribulin and control arms. Patients received a median of four prior chemotherapy regimens in both arms.
In Study 1, a statistically significant improvement in overall survival was observed in patients randomized to the Eribulin arm compared to the control arm (see Table 3). An updated, unplanned survival analysis, conducted when 77% of events had been observed (see Figure 1), was consistent with the primary analysis. In patients randomized to Eribulin, the objective response rate by the RECIST criteria was 11% (95% CI: 8.6%, 14.3%) and the median response duration was 4.2 months (95% CI: 3.8, 5.0 months).

How Supplied
Eribulin mesylate injection, 1 mg/2 mL, in a single-use vial. One vial per carton.
Storage
Store at 25°C (77°F); excursions permitted to 15° – 30° C (59° -86° F). Do not freeze. Store the vials in their original cartons.
Images
Drug Images
{{#ask: Page Name::Eribulin |?Pill Name |?Drug Name |?Pill Ingred |?Pill Imprint |?Pill Dosage |?Pill Color |?Pill Shape |?Pill Size (mm) |?Pill Scoring |?NDC |?Drug Author |format=template |template=DrugPageImages |mainlabel=- |sort=Pill Name }}
Package and Label Display Panel


{{#ask: Label Page::Eribulin |?Label Name |format=template |template=DrugLabelImages |mainlabel=- |sort=Label Page }}
Patient Counseling Information
There is limited information regarding Eribulin Patient Counseling Information in the drug label.
Precautions with Alcohol
Alcohol-Eribulin interaction has not been established. Talk to your doctor about the effects of taking alcohol with this medication.
Brand Names
Look-Alike Drug Names
There is limited information regarding Eribulin Look-Alike Drug Names in the drug label.
Drug Shortage Status
Price
References
The contents of this FDA label are provided by the National Library of Medicine.