Nephrotic syndrome pathophysiology
|
Nephrotic Syndrome Microchapters |
|
Diagnosis |
|---|
|
Treatment |
|
Case Studies |
|
Nephrotic syndrome pathophysiology On the Web |
|
American Roentgen Ray Society Images of Nephrotic syndrome pathophysiology |
|
Risk calculators and risk factors for Nephrotic syndrome pathophysiology |
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]; Associate Editor(s)-in-Chief: Yazan Daaboul, Serge Korjian
Overview
The pathophysiology of hypoalbuminemia in nephrotic syndrome is multifactorial. Proteinuria plays an important role in the pathogenesis of hyperlipidemia in nephrotic syndrome. Neurohormonal changes in the renin-angiotensin-aldosterone system, vasopressin, atrial natriuretic peptide (ANP), and sympathetic nervous system are is implicated in edema formation in nephrotic syndrome.
Pathophysiology
Hypoalbuminemia
The pathophysiology of hypoalbuminemia in nephrotic syndrome is multifactorial. The glomerular basement membrane (GBM) is normally formed of heparan sulfate and proteoglycans that contain anion sites.[1] These sites occupy the lamina rara externa of the GBM. Quantitative loss of the anion sites and neutralization of the GBM charge-selective barrier are directly related to albuminuria.[2] In nephrotic syndrome, the kidneys lose the size and charge perm-selectivity of the glomerular capillary wall and protein leakage will occur.[3][4] As such, the glomerulus will lose the ability to prevent the passage of macromolecules, such as albumin through the barrier, and albumin excretion will ensue.[4][5][6][7] In addition to the increased renal loss of albumin, increased renal catabolism always contributed to hypoalbuminemia.[7][8] Although initially believed that increased albumin intake would increase serum levels of albumin, results showed that following ingestion of albumin, albuminuria was exacerbated due to protein trafficking along the renal tubules.[8] It is thus common practice to restrict protein intake in nephrotic syndrome.[8][9] The use of ACE-inhibitors to reduce the degree of proteinuria suggested that neurohormonal changes related to angiotensin II must have a significant role in hypoalbuminemia as well.[1]
In addition, circulating factors, such as vascular permeability factor (VPF) and other lymphokines produced by T cells, are believed to play a role in binding to polyanions in the GBM and neutralizing the barrier.[10][11][12] Inflammatory changes have been related to proteinuria and related ultrastructural abnormalities may also be part of the multifactorial process behind hypoalbuminemia.[1] Hemodynamic factors, such as a reduction in glomerular plasma flow, were also shown to play a role in the urinary loss of proteins. Consequently, an increase in the clearance of proteins along the glomerular capillary is observed due to removal of water and the creation of a diffusion gradient along the Bowman's space. When the intravascular pool of albumin is lost, interstitial stores are shifted into the intravascular compartment and are similarly lost to eventually deplete all albumin stores.[1]
Hyperlipidemia
Proteinuria plays an important role in the pathogenesis of hyperlipidemia in nephrotic syndrome.[8] When hypoalbuminemia is present, the liver increases lipoprotein synthesis due to the decreased low plasma oncotic pressure. The process by which the liver increases the production of its products is believed to be regulated by a substance that is not yet identified. Animal studies have shown that in nephrotic syndrome, enzymes like hepatic 3-hydroxy-3-methylglutaryl-coenzyme A (HMG-CoA) reductase - the enzyme responsible for the rate limiting step in lipid synthesis, and acyl-coenzyme-A-cholesterol acyltransferase are both increased.[13][14][15] In parallel, activities cholesterol-7-alpha-hydroxylase and lipoprotein lipase are decreased.[13][14][15] Both processes validate that lipid synthesis is increased in nephrotic syndrome. Hyperlipidemia due to increase apo-B containing lipoproteins occurs secondary to the decrease in oncotic pressure, not to hypoalbuminemia per se.[1] The severity of lipid abnormalities directly correlates with the severity of proteinuria, but is not associated with the identity of glomerular lesions or primary etiology of nephrotic syndrome.[16][17] Increased triglycerides, LDL, and VLDL are increased and also contribute to the glomerular damage in mechanisms that remain unclear.[18] It is perhaps that abnormalities in the metabolism of triglycerides, rather than increased synthesis, are responsible for hypertriglyceridemia. It is believed that lipoprotein-mediated conversion of VLDL to LDL (via IDL) may be slowed down in nephrotic syndrome.[19][20] In contrast, HDL2 is generally reduced whereas HDL3 is elevated due to a reduction in the lecithin-cholesterol acyltransferase activity.[16]
Edema Formation
The pathophysiology of edema formation in nephrotic syndrome is still controversial. Researchers hypothesize that the "underfill" mechanism is responsible for edema formation due to loss of plasma albumin and colloid oncotic pressure, followed by increased filtration from the intravascular space to the interstitial space.[21] Eventually, hypovolemia and renal hypoperfusion will lead to activation of the renin-angiotensin-aldosterone system (RAAS), vasopressin,[22][23][24] atrial natriuretic peptide (ANP),[25][26][27][28] and the sympathetic nervous system[29], causing sodium retention.[21]
The Starling equation adequately explains the net fluid movement between the intravascular and the interstitial compartments:
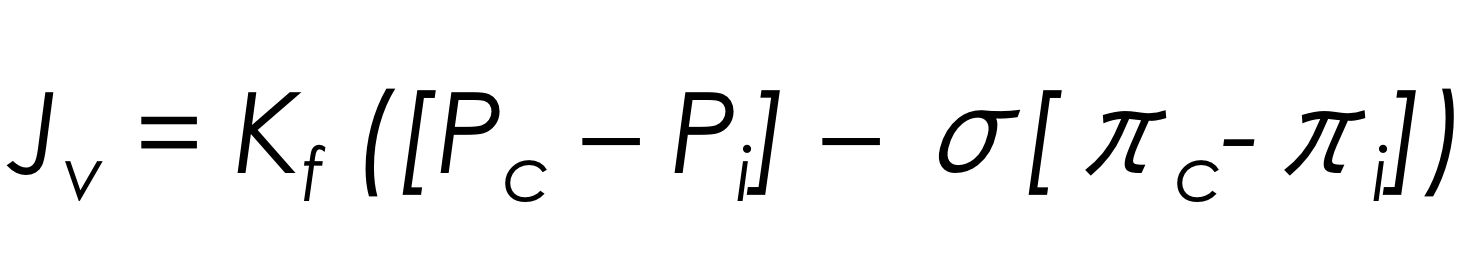
Jv is the net fluid movement between compartments; Pc is the capillary hydrostatic pressure; Pi is the interstitial hydrostatic pressure; σ is the reflection coefficient to proteins. It is a measure of vascular permeability; πc is the capillary oncotic pressure; πi is the interstitial oncotic pressure; Kf is the overall filtration permeability constant to volume flow; it is a product of the hydraulic conductance and capillary surface area. It is a measure of vascular permeability.
Protective mechanisms against the formation of edema in hypoalbuminemia are[21]:
- "Washdown" of interstitial protein concentration; Increase in fluid filtration from the intravascular space into the interstitial space.
- "Washout" of interstitial proteins; Increase in fluid delivery to interstitial space to increase lymphatic flow.
In summary, the role of the following neurohormonal changes is implicated in edema formation in nephrotic syndrome:[21]
- Renin-angiotensin-aldosterone system
- Vasopressin
- Atrial natriuretic peptide (ANP)
- Sympathetic nervous system
Although the mechanism seemed straightforward, newer studies showed that the pathophysiology is not as simple as once thought. Even the inhibition of the RAAS pathway using ACE-inhibitors did not seem to inhibit sodium retention as expected.[30][31][32][33][34][35] Volume depletion associated with nephrotic syndrome is only seen in a minority of patients.[21] In fact, the rate at which protein loss occurs is equally important. When plasmapheresis was conducted in rats to control the rate of plasma protein reduction, rapid loss of proteins was associated with increase in RAAS and positive sodium balance with decrease in plasma and blood volumes.[36][37][38] These changes were not seen in rats that received moderate continuous plasmapheresis. It is postulated that in some glomerular diseases, such as minimal change disease, where proteinuria occurs very rapidly, the rate of protein loss might mimic rat studies and edema formation occurs due to changes unseen in moderate protein loss.[36][37][38]
It is believed that excessive proteinuria, as seen in patients with minimal change disease, and depletion of serum albumin creates a disequilibrium between plasma and extravascular stores of albumin in attempt to restore the plasma-to-interstitial difference in colloid oncotic pressure (COP).[32] The disequilibrium creates a state of uncompensated hypovolemia when COP becomes < 8 mmHg.[32] The dropping pressure temporarily stimulates aldosterone and other sodium-handling indices to retain sodium.[32][39][40] Following sodium retention, a steady-state is reached and sodium is no longer actively retained.[41][42][35] If a stable steady-state is not reached in cases when COP cannot be maintained above 8 mmHg, massive proteinuria persists.[32]
Edema formation is not simply due to a sodium retention following a decrease in systemic volume and fall in plasma colloid pressure.[43][44] Recent evidence has shown that edema formation and sodium retention may thus be less related to hypovolemia itself, but rather to a primary intrinsic dysfunction of the renal handling of sodium followed by superimposing hypovolemia.[30][45][46] Tubular absorption is increased in patients with nephrotic syndrome, even in segments beyond the distal convoluted tubule, due to mechanisms that remain obscured.[47][32] Nonetheless, an increase in the sodium/potassium/ATPase activity and amount associated with an increase of aldosterone-dependent expression of epithelial sodium channels (ENaC)[48][49][50] were noted distally in the cortical collecting duct of nephrotic kidneys, emphasizing the role of the kidneys themselves in the pathogenesis of edema formation.[48][51][52][53]
Finally, it is important to recognize that the pathology of edema formation is not homogeneous and that sodium retention alone cannot explain edema formation in nephrotic syndrome.[21] On the contrary, the extent of edema may be very different even with the same degree of proteinuria.[32] Several hypotheses suggest that patient and disease characteristics may account for the varying degree of edema in patients with similar amounts of proteinuria.
References
- ↑ 1.0 1.1 1.2 1.3 1.4 Rodrigo R, Bravo I, Pino M (1996). "Proteinuria and albumin homeostasis in the nephrotic syndrome: effect of dietary protein intake". Nutr Rev. 54 (11 Pt 1): 337–47. PMID 9110562.
- ↑ Levin M, Gascoine P, Turner MW, Barratt TM (1989). "A highly cationic protein in plasma and urine of children with steroid-responsive nephrotic syndrome". Kidney Int. 36 (5): 867–77. PMID 2615193.
- ↑ Ota Z, Shikata K, Ota K (1994). "Nephrotic tunnels in glomerular basement membrane as revealed by a new electron microscopic method". J Am Soc Nephrol. 4 (12): 1965–73. PMID 7919150.
- ↑ 4.0 4.1 Deen WM, Bridges CR, Brenner BM, Myers BD (1985). "Heteroporous model of glomerular size selectivity: application to normal and nephrotic humans". Am J Physiol. 249 (3 Pt 2): F374–89. PMID 4037090.
- ↑ Myers BD, Guasch A (1994). "Mechanisms of proteinuria in nephrotic humans". Pediatr Nephrol. 8 (1): 107–12. PMID 8142207.
- ↑ Savin VJ (1993). "Mechanisms of proteinuria in noninflammatory glomerular diseases". Am J Kidney Dis. 21 (4): 347–62. PMID 8465812.
- ↑ 7.0 7.1 Myers BD (1990). "Pathophysiology of proteinuria in immune glomerular injury". Am J Nephrol. 10 Suppl 1: 19–23. PMID 1701613.
- ↑ 8.0 8.1 8.2 8.3 Kaysen GA, Kirkpatrick WG, Couser WG (1984). "Albumin homeostasis in the nephrotic rat: nutritional considerations". Am J Physiol. 247 (1 Pt 2): F192–202. PMID 6742202.
- ↑ Hutchinson FN, Schambelan M, Kaysen GA (1987). "Modulation of albuminuria by dietary protein and converting enzyme inhibition". Am J Physiol. 253 (4 Pt 2): F719–25. PMID 2821830.
- ↑ Bakker WW, Roskam G, Hardonk MJ, Vos JT, Bleumink E (1985). "The glomerular polyanion (GPA) of the rat kidney. III. Further characterization of a vaso-active serum factor which reduces GPA". Br J Exp Pathol. 66 (1): 47–55. PMC 2041028. PMID 3882117.
- ↑ Zimmerman SW (1984). "Increased urinary protein excretion in the rat produced by serum from a patient with recurrent focal glomerular sclerosis after renal transplantation". Clin Nephrol. 22 (1): 32–8. PMID 6383672.
- ↑ Schnaper HW (1989). "The immune system in minimal change nephrotic syndrome". Pediatr Nephrol. 3 (1): 101–10. PMID 2702078.
- ↑ 13.0 13.1 Vaziri ND, Liang K (2002). "Up-regulation of acyl-coenzyme A:cholesterol acyltransferase (ACAT) in nephrotic syndrome". Kidney Int. 61 (5): 1769–75. doi:10.1046/j.1523-1755.2002.00319.x. PMID 11967026.
- ↑ 14.0 14.1 Shearer GC, Kaysen GA (2001). "Proteinuria and plasma compositional changes contribute to defective lipoprotein catabolism in the nephrotic syndrome by separate mechanisms". Am J Kidney Dis. 37 (1 Suppl 2): S119–22. PMID 11158876.
- ↑ 15.0 15.1 Shearer GC, Stevenson FT, Atkinson DN, Jones H, Staprans I, Kaysen GA (2001). "Hypoalbuminemia and proteinuria contribute separately to reduced lipoprotein catabolism in the nephrotic syndrome". Kidney Int. 59 (1): 179–89. doi:10.1046/j.1523-1755.2001.00478.x. PMID 11135070.
- ↑ 16.0 16.1 Wheeler DC, Bernard DB (1994). "Lipid abnormalities in the nephrotic syndrome: causes, consequences, and treatment". Am J Kidney Dis. 23 (3): 331–46. PMID 8128933.
- ↑ Newmark SR, Anderson CF, Donadio JV, Ellefson RD (1975). "Lipoprotein profiles in adult nephrotics". Mayo Clin Proc. 50 (7): 359–64. PMID 1152529.
- ↑ Wheeler DC, Varghese Z, Moorhead JF (1989). "Hyperlipidemia in nephrotic syndrome". Am J Nephrol. 9 Suppl 1: 78–84. PMID 2646930.
- ↑ Warwick GL, Packard CJ, Demant T, Bedford DK, Boulton-Jones JM, Shepherd J (1991). "Metabolism of apolipoprotein B-containing lipoproteins in subjects with nephrotic-range proteinuria". Kidney Int. 40 (1): 129–38. PMID 1921148.
- ↑ Vega GL, Toto RD, Grundy SM (1995). "Metabolism of low density lipoproteins in nephrotic dyslipidemia: comparison of hypercholesterolemia alone and combined hyperlipidemia". Kidney Int. 47 (2): 579–86. PMID 7723244.
- ↑ 21.0 21.1 21.2 21.3 21.4 21.5 Siddall EC, Radhakrishnan J (2012). "The pathophysiology of edema formation in the nephrotic syndrome". Kidney Int. 82 (6): 635–42. doi:10.1038/ki.2012.180. PMID 22718186.
- ↑ Usberti M, Federico S, Meccariello S, Cianciaruso B, Balletta M, Pecoraro C; et al. (1984). "Role of plasma vasopressin in the impairment of water excretion in nephrotic syndrome". Kidney Int. 25 (2): 422–9. PMID 6727137.
- ↑ Rascher W, Tulassay T (1987). "Hormonal regulation of water metabolism in children with nephrotic syndrome". Kidney Int Suppl. 21: S83–9. PMID 3306110.
- ↑ Tulassay T, Rascher W, Lang RE, Seyberth HW, Schärer K (1987). "Atrial natriuretic peptide and other vasoactive hormones in nephrotic syndrome". Kidney Int. 31 (6): 1391–5. PMID 2956451.
- ↑ Perico N, Delaini F, Lupini C, Benigni A, Galbusera M, Boccardo P; et al. (1989). "Blunted excretory response to atrial natriuretic peptide in experimental nephrosis". Kidney Int. 36 (1): 57–64. PMID 2554049.
- ↑ Hildebrandt DA, Banks RO (1988). "Effect of atrial natriuretic factor on renal function in rats with nephrotic syndrome". Am J Physiol. 254 (2 Pt 2): F210–6. PMID 2964203.
- ↑ Radin MJ, McCune SA (1993). "The effect of atrial natriuretic peptide infusion on renal haemodynamics and plasma lipoproteins in puromycin aminonucleoside nephrosis in rats". Clin Exp Pharmacol Physiol. 20 (4): 245–51. PMID 8485924.
- ↑ Keeler R, Feuchuk D, Wilson N (1987). "Atrial peptides and the renal response to hypervolemia in nephrotic rats". Can J Physiol Pharmacol. 65 (10): 2071–5. PMID 2962709.
- ↑ DiBona GF (1990). "Role of the renal nerves in renal sodium retention and edema formation". Trans Am Clin Climatol Assoc. 101: 38–44, discussion 44-5. PMC 2376505. PMID 2486445.
- ↑ 30.0 30.1 Meltzer JI, Keim HJ, Laragh JH, Sealey JE, Jan KM, Chien S (1979). "Nephrotic syndrome: vasoconstriction and hypervolemic types indicated by renin-sodium profiling". Ann Intern Med. 91 (5): 688–96. PMID 496101.
- ↑ Vande Walle J, Donckerwolcke R, Boer P, van Isselt HW, Koomans HA, Joles JA (1996). "Blood volume, colloid osmotic pressure and F-cell ratio in children with the nephrotic syndrome". Kidney Int. 49 (5): 1471–7. PMID 8731116.
- ↑ 32.0 32.1 32.2 32.3 32.4 32.5 32.6 Vande Walle JG, Donckerwolcke RA, Koomans HA (1999). "Pathophysiology of edema formation in children with nephrotic syndrome not due to minimal change disease". J Am Soc Nephrol. 10 (2): 323–31. PMID 10215332.
- ↑ Hammond TG, Whitworth JA, Saines D, Thatcher R, Andrews J, Kincaid-Smith P (1984). "Renin-angiotensin-aldosterone system in nephrotic syndrome". Am J Kidney Dis. 4 (1): 18–23. PMID 6377881.
- ↑ Shapiro MD, Hasbargen J, Hensen J, Schrier RW (1990). "Role of aldosterone in the sodium retention of patients with nephrotic syndrome". Am J Nephrol. 10 (1): 44–8. PMID 2188506.
- ↑ 35.0 35.1 Bohlin AB, Berg U (1984). "Renal sodium handling in minimal change nephrotic syndrome". Arch Dis Child. 59 (9): 825–30. PMC 1628730. PMID 6486860.
- ↑ 36.0 36.1 Manning RD, Guyton AC (1983). "Effects of hypoproteinemia on fluid volumes and arterial pressure". Am J Physiol. 245 (2): H284–93. PMID 6881362.
- ↑ 37.0 37.1 Manning RD (1987). "Effects of hypoproteinemia on renal hemodynamics, arterial pressure, and fluid volume". Am J Physiol. 252 (1 Pt 2): F91–8. PMID 3544869.
- ↑ 38.0 38.1 Joles JA, Koomans HA, Kortlandt W, Boer P, Dorhout Mees EJ (1988). "Hypoproteinemia and recovery from edema in dogs". Am J Physiol. 254 (6 Pt 2): F887–94. PMID 3132859.
- ↑ Koomans HA, Kortlandt W, Geers AB, Dorhout Mees EJ (1985). "Lowered protein content of tissue fluid in patients with the nephrotic syndrome: observations during disease and recovery". Nephron. 40 (4): 391–5. PMID 4022206.
- ↑ Koomans HA, Braam B, Geers AB, Roos JC, Dorhout Mees EJ (1986). "The importance of plasma protein for blood volume and blood pressure homeostasis". Kidney Int. 30 (5): 730–5. PMID 3784303.
- ↑ Vande Walle JG, Donckerwolcke RA, van Isselt JW, Derkx FH, Joles JA, Koomans HA (1995). "Volume regulation in children with early relapse of minimal-change nephrosis with or without hypovolaemic symptoms". Lancet. 346 (8968): 148–52. PMID 7603230.
- ↑ Van de Walle JG, Donckerwolcke RA, Greidanus TB, Joles JA, Koomans HA (1996). "Renal sodium handling in children with nephrotic relapse: relation to hypovolaemic symptoms". Nephrol Dial Transplant. 11 (11): 2202–8. PMID 8941579.
- ↑ BROWN E, HOPPER J, WENNESLAND R (1957). "Blood volume and its regulation". Annu Rev Physiol. 19: 231–54. doi:10.1146/annurev.ph.19.030157.001311. PMID 13412057.
- ↑ YAMAUCHI H, HOPPER J (1964). "HYPOVOLEMIC SHOCK AND HYPOTENSION AS A COMPLICATION IN THE NEPHROTIC SYNDROME. REPORT OF TEN CASES". Ann Intern Med. 60: 242–54. PMID 14114444.
- ↑ Dorhout EJ, Roos JC, Boer P, Yoe OH, Simatupang TA (1979). "Observations on edema formation in the nephrotic syndrome in adults with minimal lesions". Am J Med. 67 (3): 378–84. PMID 474584.
- ↑ Brown EA, Markandu ND, Sagnella GA, Squires M, Jones BE, MacGregor GA (1982). "Evidence that some mechanism other than the renin system causes sodium retention in nephrotic syndrome". Lancet. 2 (8310): 1237–40. PMID 6128546.
- ↑ Ichikawa I, Rennke HG, Hoyer JR, Badr KF, Schor N, Troy JL; et al. (1983). "Role for intrarenal mechanisms in the impaired salt excretion of experimental nephrotic syndrome". J Clin Invest. 71 (1): 91–103. PMC 436841. PMID 6848563.
- ↑ 48.0 48.1 Lourdel S, Loffing J, Favre G, Paulais M, Nissant A, Fakitsas P; et al. (2005). "Hyperaldosteronemia and activation of the epithelial sodium channel are not required for sodium retention in puromycin-induced nephrosis". J Am Soc Nephrol. 16 (12): 3642–50. doi:10.1681/ASN.2005040363. PMID 16267158.
- ↑ de Seigneux S, Kim SW, Hemmingsen SC, Frøkiaer J, Nielsen S (2006). "Increased expression but not targeting of ENaC in adrenalectomized rats with PAN-induced nephrotic syndrome". Am J Physiol Renal Physiol. 291 (1): F208–17. doi:10.1152/ajprenal.00399.2005. PMID 16403831.
- ↑ Kim SW, Wang W, Nielsen J, Praetorius J, Kwon TH, Knepper MA; et al. (2004). "Increased expression and apical targeting of renal ENaC subunits in puromycin aminonucleoside-induced nephrotic syndrome in rats". Am J Physiol Renal Physiol. 286 (5): F922–35. doi:10.1152/ajprenal.00277.2003. PMID 15075188.
- ↑ Vogt B, Favre H (1991). "Na+,K(+)-ATPase activity and hormones in single nephron segments from nephrotic rats". Clin Sci (Lond). 80 (6): 599–604. PMID 1647923.
- ↑ Deschênes G, Doucet A (2000). "Collecting duct (Na+/K+)-ATPase activity is correlated with urinary sodium excretion in rat nephrotic syndromes". J Am Soc Nephrol. 11 (4): 604–15. PMID 10752519.
- ↑ Deschênes G, Gonin S, Zolty E, Cheval L, Rousselot M, Martin PY; et al. (2001). "Increased synthesis and avp unresponsiveness of Na,K-ATPase in collecting duct from nephrotic rats". J Am Soc Nephrol. 12 (11): 2241–52. PMID 11675400.