Hemolytic-uremic syndrome pathophysiology
|
Hemolytic-uremic syndrome Microchapters |
|
Differentiating Hemolytic-uremic syndrome from other Diseases |
|---|
|
Diagnosis |
|
Treatment |
|
Case Studies |
|
Hemolytic-uremic syndrome pathophysiology On the Web |
|
American Roentgen Ray Society Images of Hemolytic-uremic syndrome pathophysiology |
|
Risk calculators and risk factors for Hemolytic-uremic syndrome pathophysiology |
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]; Associate Editor(s)-in-Chief: Sogand Goudarzi, MD [2], Anila Hussain, MD [3]Parth Vikram Singh, MBBS[4]
Overview
It is understood that hemolytic-uremic syndrome (HUS) is the result of microvascular endothelial cell damage characterized by thrombotic microangiopathy (TMA) in renal glomeruli, gastrointestinal tract, brain and pancreas in all of which the main lesion is the thickening of vessel wall (mainly in capillaries and arterioles), microthrombi in platelets and obstruction of vessel lumen ( partial or complete). Loss of physiological resistance to thrombus formation, complement consumption, leukocyte adhesion to damaged endothelium, the abnormal release of von Willibrand Factor (vWF) and fragmentation, and increased vascular shear stress lead to further amplification of microangiopathy. Typical/ Shiga-toxin-associated hemolytic uremic syndrome (HUS) is usually caused by E.Coli and serotype O157: H7 is most common while congenital predisposing conditions like complement factor abnormalities may play a role in recurrent and familial forms.
Shiga toxin production is the cardinal virulence trait of STEC. Shiga toxin 1 and Shiga toxin 2 each consist of a single A subunit and a pentameric B subunit. Disease begins when the B subunit binds globotriaosylceramide (Gb3) on eukaryotic cells. The holotoxin is internalized, traffics retrogradely through the Golgi apparatus to the endoplasmic reticulum, and releases an enzymatically active A1 fragment that cleaves 28S ribosomal RNA and inhibits protein synthesis. The pathogenicity of STEC is determined mainly by whether the organism expresses Shiga toxin 1, Shiga toxin 2, or both. STEC that produce Shiga toxin 2 usually cause bloody diarrhea and account for almost all cases of diarrhea-associated HUS. Because E. coli O157 almost universally produces Shiga toxin 2, it is generally considered a high-risk STEC.
Pathophysiology
Pathogenesis
- Circulating Shiga toxin is thought to underlie the vascular injury leading to HUS, with the most pronounced end-organ damage occurring in the kidneys. Organ-specific microvascular injury initiates thrombotic responses, including intraglomerular microthrombi and platelet adhesion, leading to thrombocytopenia, hemolysis, and end-organ damage. Activation of the microvascular endothelium is believed to contribute to the gastrointestinal manifestations of STEC infection. Early colonic histology shows superficial inflammation and focal necrosis with preserved deep crypts, findings that suggest ischemia. During the diarrheal phase, reported hematologic abnormalities include increased plasminogen activator inhibitor type 1 activity, elevated D-dimer and prothrombin activation fragments 1 and 2, increased platelet-activating factor, sheared von Willebrand factor, and dysregulated angiopoietin 1 and 2 activity.[1]
- It is understood that hemolytic-uremic syndrome (HUS) is the result of microvascular endothelial cell damage characterized by thrombotic microangiopathy (TMA) in renal glomeruli, gastrointestinal tract, brain and pancreas in all of which the main lesion is the thickening of vessel wall (mainly in capillaries and arterioles), microthrombi in platelets and obstruction of vessel lumen( partial or complete).
- Loss of physiological resistance to thrombus formation, complement consumption, leukocyte adhesion to damaged endothelium, the abnormal release of von Willibrand Factor (vWF) and fragmentation, and increased vascular shear stress lead to further amplification of microangiopathy.
- Congenital predisposing conditions like complement factor abnormalities may play a role in recurrent and familial forms.[2]
- Typical/ Shiga-toxin-associated hemolytic uremic syndrome (HUS) is usually caused by E.Coli.
- Serotype O157: H7 is most commonly seen in the USA and Europe, although other serotypes less commonly associated include O26:H11, O103:H2, O121:H19, O145:NM and O111:NM. Other strains, especially O111:H-serotype is frequently found in other countries as well.
- EHEC produce several virulence factors including Shiga-Toxin and that gain access to the blood circulation after damaging the intestinal endothelium and later affect the target organs
- Pathogen is usually transmitted via the ingestion of undercooked ground meat to the human host.
- Following transmission/ingestion, the EHEC is assumed to bind to the small intestine followed by colonization of colon.[3]
- EHEC interacts with intestinal microflora as well as host hormonal response thus leading to the activation of several virulence factors including Shiga-Toxin (Stx) and others that enable attachment of pathogen to the instestinal epithelial cell and enhancing the mobility of flagella thus leading to induction of Stx which adheres to the endothelium of the intestine and lead to ulceration and hemorrhaging[4][5][6]
- Intestinal epithelial damage allows bacterial virulence factors to enter the circulation after which Stx in circulation binds to the platelets, neutrophils, and monocytes as well as to platelet-monocyte and platelet-neutrophils in complexes leading to tissue-factor (TF) expressing microparticle release.[7]
- Aggregates are formed between monocyte and platelets and also between neutrophils and platelets. Stx can also bind to the blood cells via G3b receptors in addition to other glycolipid receptors where as lipopolysaccharide or LPS binds via TLR-4 or Toll like receptor, which is in complex with CD62 on platelets.[8]
- Platelet activation lead to prothrombotic state and microthrombi lead to thrombocytopenia. In presence of a circulation with high resistance like renal microcirculations, these effects are enhanced. Other G3b expressing organs like including brain can also be affected.
- Stx induces cell death by inhibiting the protein synthesis or by apoptosis.[9]
- Neutrophils, monocytes and IgM-producing B lymphocytes show resistance to cytotoxic effects of shiga toxin. In macrophage-like THP-1 cells, both apoptotic and cell survival signaling pathways were activated after they were exposed to Shiga toxin-1, hence, most leukocytes being exposed to Shiga toxin will not undergo cell death, allowing the toxin to circulate bound to their cell membrane.[10]
- Endothelial cell damage of glomerular capillaries is the main feature in the pathogenesis of HUS.
- Stx exerts cytotoxic and apoptotic effects on glomerular endothelial and epithelial cells[11][12].
- The pathogenesis in complement mediated or atypical HUS may include complement mediated platelet activation and endothelial damage and usually have low complement levels.
Genetics
Mutations in the genes associated with atypical HUS can cause uncontrolled complement system activation which attacks endothelial cells leading to inflammation and thrombi formation and may lead to kidney injury and renal failure. Examples include:[13][14][15]
- Complement factor H (CFH) mutation/ factor H deficiency (autosomal dominant)
- Membrane co-factor protein deficiency (MCP; CD46)
- Factor B overactivity (Complement Factor B mutation)
- Diacylglycerol kinase epsilon gene mutations
- Factor I (IF) mutation
Other genetic conditions predisposing to atypical HUS include:
- Mutations in the MMACHC (methyl malonic aciduria and homocystinuria type C) gene[16]
- Genetic disorders of ADAMTS13[17]
Associated Conditions
Conditions associated with HUS include:
- Malignancy, cancer chemotherapy and ionizing radiation
- Calcineurin inhibitors and transplantation
- Pregnancy, HELLP syndrome, and oral contraceptive pill
- Systemic lupus erythematosus and antiphospholipid antibody syndrome
- Glomerulopathy
Gross Pathology
On gross pathology, [feature2], and [feature3] are characteristic findings of HUS.
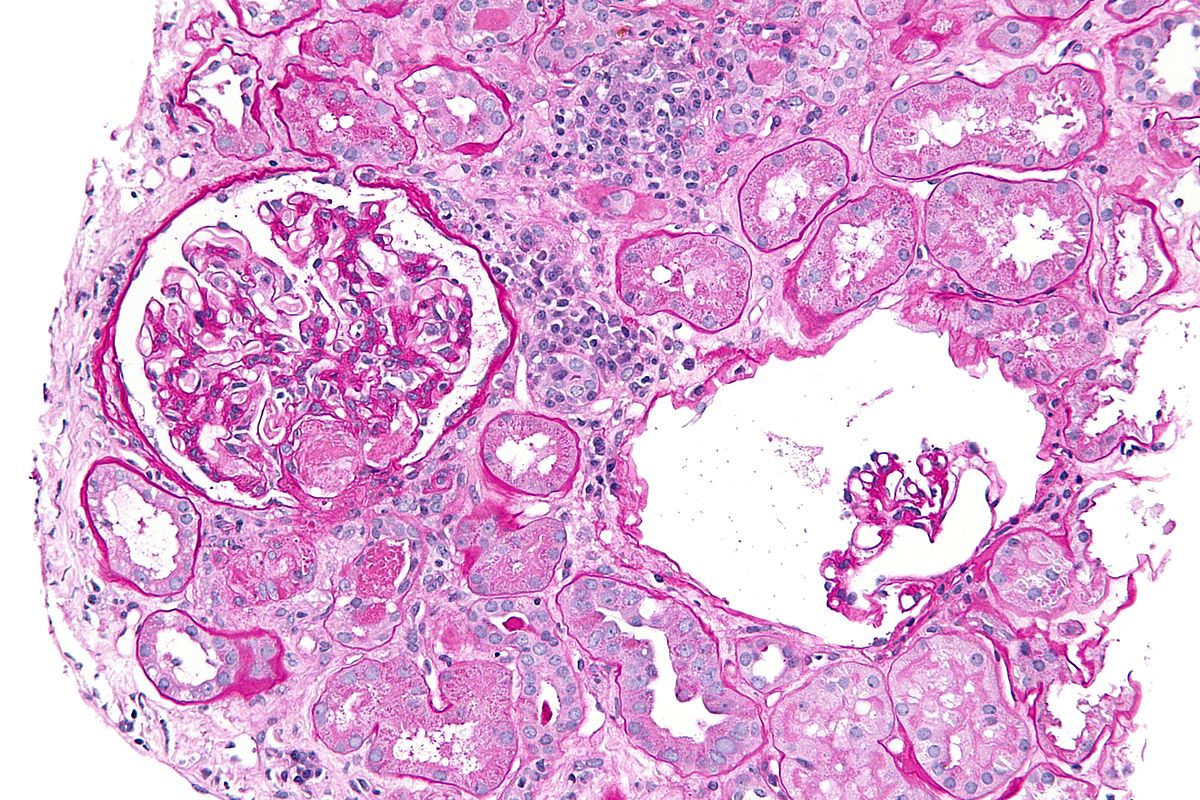
Microscopic Pathology
On microscopic histopathological analysis finding of HUS.
- Granular (muddy brown) casts
References
- ↑ Freedman SB, van de Kar NC, Tarr PI (October 2023). "Shiga Toxin-Producing Escherichia coli and the Hemolytic-Uremic Syndrome". N Engl J Med. 389 (15): 1402–1414. doi:10.1056/NEJMra2108739. PMID 37819955 Check
|pmid=value (help). - ↑ Ruggenenti P, Noris M, Remuzzi G (2001). "Thrombotic microangiopathy, hemolytic uremic syndrome, and thrombotic thrombocytopenic purpura". Kidney Int. 60 (3): 831–46. doi:10.1046/j.1523-1755.2001.060003831.x. PMID 11532079.
- ↑ Malyukova I, Murray KF, Zhu C, Boedeker E, Kane A, Patterson K; et al. (2009). "Macropinocytosis in Shiga toxin 1 uptake by human intestinal epithelial cells and transcellular transcytosis". Am J Physiol Gastrointest Liver Physiol. 296 (1): G78–92. doi:10.1152/ajpgi.90347.2008. PMC 2636932. PMID 18974311.
- ↑ Pacheco AR, Sperandio V (2009). "Inter-kingdom signaling: chemical language between bacteria and host". Curr Opin Microbiol. 12 (2): 192–8. doi:10.1016/j.mib.2009.01.006. PMC 4852728. PMID 19318290.
- ↑ Walker WA. Pediatric Gastrointestinal Disease: Pathophysiology, Diagnosis, Management. 4th ed. Hamilton, Ont.: BC Decker, 2004
- ↑ Hughes DT, Clarke MB, Yamamoto K, Rasko DA, Sperandio V (2009). "The QseC adrenergic signaling cascade in Enterohemorrhagic E. coli (EHEC)". PLoS Pathog. 5 (8): e1000553. doi:10.1371/journal.ppat.1000553. PMC 2726761. PMID 19696934.
- ↑ Ståhl AL, Sartz L, Nelsson A, Békássy ZD, Karpman D (2009). "Shiga toxin and lipopolysaccharide induce platelet-leukocyte aggregates and tissue factor release, a thrombotic mechanism in hemolytic uremic syndrome". PLoS One. 4 (9): e6990. doi:10.1371/journal.pone.0006990. PMC 2735777. PMID 19750223.
- ↑ Ståhl AL, Svensson M, Mörgelin M, Svanborg C, Tarr PI, Mooney JC; et al. (2006). "Lipopolysaccharide from enterohemorrhagic Escherichia coli binds to platelets through TLR4 and CD62 and is detected on circulating platelets in patients with hemolytic uremic syndrome". Blood. 108 (1): 167–76. doi:10.1182/blood-2005-08-3219. PMC 1895830. PMID 16514062.
- ↑ Cherla RP, Lee SY, Tesh VL (2003). "Shiga toxins and apoptosis". FEMS Microbiol Lett. 228 (2): 159–66. PMID 14638419.
- ↑ Brigotti M, Carnicelli D, Ravanelli E, Barbieri S, Ricci F, Bontadini A; et al. (2008). "Interactions between Shiga toxins and human polymorphonuclear leukocytes". J Leukoc Biol. 84 (4): 1019–27. doi:10.1189/jlb.0308157. PMID 18625912.
- ↑ Pijpers AH, van Setten PA, van den Heuvel LP, et al. Verocytotoxin-induced apoptosis of human microvascular endothelial cells. J Am Soc Nephrol 2001;12:767- 778
- ↑ Hughes AK, Stricklett PK, Schmid D, Kohan DE (2000). "Cytotoxic effect of Shiga toxin-1 on human glomerular epithelial cells". Kidney Int. 57 (6): 2350–9. doi:10.1046/j.1523-1755.2000.00095.x. PMID 10844605.
- ↑ Frémeaux-Bacchi V (2013). "[Pathophysiology of atypical hemolytic uremic syndrome. Ten years of progress, from laboratory to patient]". Biol Aujourdhui. 207 (4): 231–40. doi:10.1051/jbio/2013027. PMID 24594571.
- ↑ Loirat C, Noris M, Fremeaux-Bacchi V (2008). "Complement and the atypical hemolytic uremic syndrome in children". Pediatr Nephrol. 23 (11): 1957–72. doi:10.1007/s00467-008-0872-4. PMID 18594873.
- ↑ Jessica Caprioli, Marina Noris, Simona Brioschi, Gaia Pianetti, Federica Castelletti, Paola Bettinaglio, Caterina Mele, Elena Bresin, Linda Cassis, Sara Gamba, Francesca Porrati, Sara Bucchioni, Giuseppe Monteferrante, Celia J. Fang, M. K. Liszewski, David Kavanagh, John P. Atkinson & Giuseppe Remuzzi (2006). "Genetics of HUS: the impact of MCP, CFH, and IF mutations on clinical presentation, response to treatment, and outcome". Blood. 108 (4): 1267–1279. doi:10.1182/blood-2005-10-007252. PMID 16621965. Unknown parameter
|month=ignored (help) - ↑ Adrovic A, Canpolat N, Caliskan S, Sever L, Kıykım E, Agbas A; et al. (2016). "Cobalamin C defect-hemolytic uremic syndrome caused by new mutation in MMACHC". Pediatr Int. 58 (8): 763–5. doi:10.1111/ped.12953. PMID 27324188.
- ↑ Feng S, Eyler SJ, Zhang Y, Maga T, Nester CM, Kroll MH; et al. (2013). "Partial ADAMTS13 deficiency in atypical hemolytic uremic syndrome". Blood. 122 (8): 1487–93. doi:10.1182/blood-2013-03-492421. PMC 3750341. PMID 23847193.