Sandbox:Dildar: Difference between revisions
No edit summary |
|||
| Line 6: | Line 6: | ||
|[[Image:Small-cell-lung-cancer-1.jpg|thumb|Chest x-ray: Small cell carcinoma of the lung. Antero-posterior view, Case courtesy of A.Prof Frank Gaillard, via,Radiopaedia.org <ref> href="https://radiopaedia.org/">Radiopaedia.org</a>. From the case <a href="https://radiopaedia.org/cases/10494">rID: 10494</ref>]] | |[[Image:Small-cell-lung-cancer-1.jpg|thumb|Chest x-ray: Small cell carcinoma of the lung. Antero-posterior view, Case courtesy of A.Prof Frank Gaillard, via,Radiopaedia.org <ref> href="https://radiopaedia.org/">Radiopaedia.org</a>. From the case <a href="https://radiopaedia.org/cases/10494">rID: 10494</ref>]] | ||
|[[Image: Left-hilar-mass-small-cell-lung-cancer-2.png.jpeg|thumb|Chest x-ray: Small cell carcinoma of the lung.Case courtesy of Dr Henry Knipe, via, Radiopaedia.org<ref> href="https://radiopaedia.org/">Radiopaedia.org</a>. From the case <a href="https://radiopaedia.org/cases/30005">rID: 30005</a></ref> | |[[Image: Left-hilar-mass-small-cell-lung-cancer-2.png.jpeg|thumb|Chest x-ray: Small cell carcinoma of the lung.Case courtesy of Dr Henry Knipe, via, Radiopaedia.org<ref> href="https://radiopaedia.org/">Radiopaedia.org</a>. From the case <a href="https://radiopaedia.org/cases/30005">rID: 30005</a></ref> | ||
|} | |||
==Lung cancer surgery == | ==Lung cancer surgery == | ||
{{CMG}}; {{AE}} | {{CMG}}; {{AE}} | ||
Revision as of 21:15, 23 February 2018
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]; Associate Editor(s)-in-Chief: Dildar Hussain, MBBS [2]
Small cell lung cancer X rays
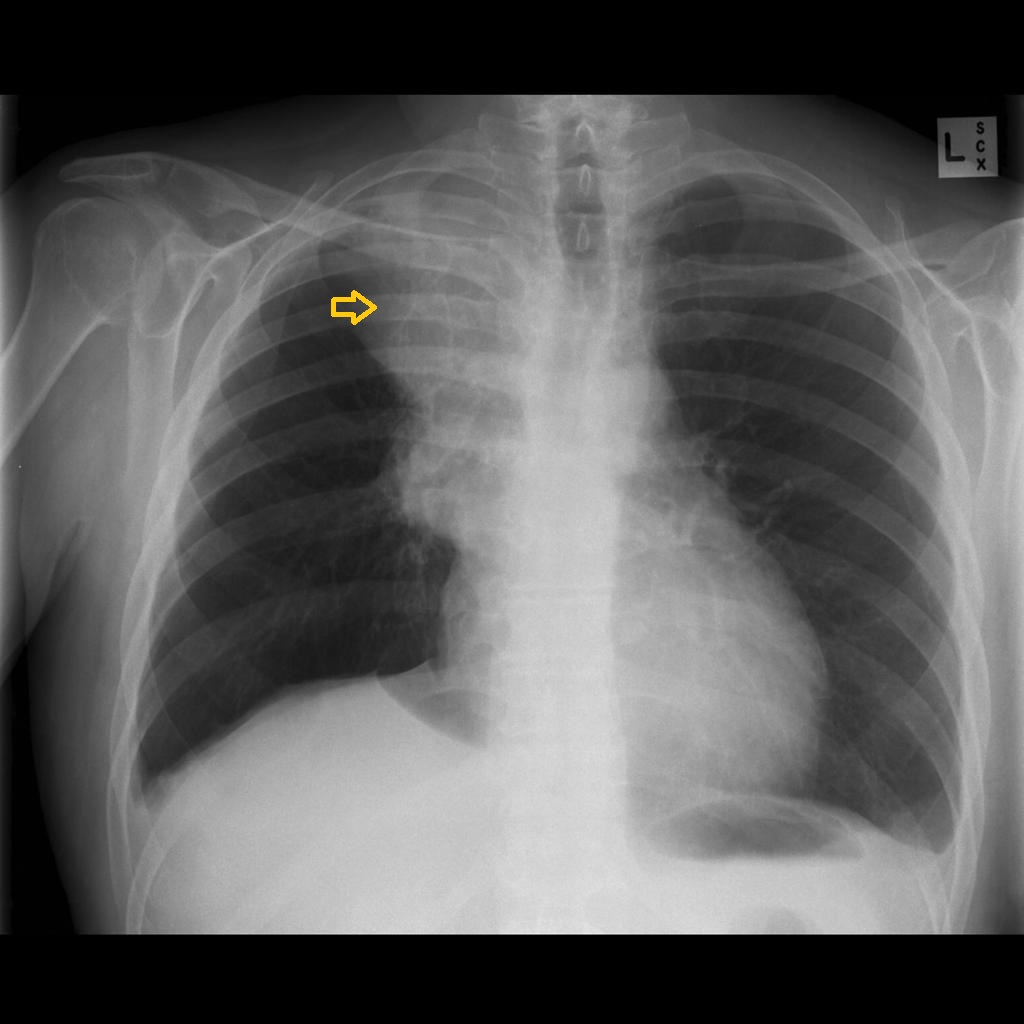 |
[[Image: Left-hilar-mass-small-cell-lung-cancer-2.png.jpeg|thumb|Chest x-ray: Small cell carcinoma of the lung.Case courtesy of Dr Henry Knipe, via, Radiopaedia.org[2] |
Lung cancer surgery
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [3]; Associate Editor(s)-in-Chief: If investigations confirm lung cancer, CT scan and often positron emission tomography (PET) are used to determine whether the disease is localised and amenable to surgery or whether it has spread to the point where it cannot be cured surgically.
Blood tests and spirometry (lung function testing) are also necessary to assess whether the patient is well enough to be operated on. If spirometry reveals poor respiratory reserve (often due to chronic obstructive pulmonary disease), surgery may be contraindicated.
Surgery itself has an operative death rate of about 4.4%, depending on the patient's lung function and other risk factors.[1] Surgery is usually only an option in non-small cell lung carcinoma limited to one lung, up to stage IIIA. This is assessed with medical imaging (computed tomography, positron emission tomography). A sufficient pre-operative respiratory reserve must be present to allow adequate lung function after the tissue is removed.
Pulmonary Reserve The American College of Chest Physicians established clinical practice guidelines for the physiologic evaluation of patients with lung cancer being considered for resectional surgery.[2] The preoperative physiologic assessment should include a cardiac evaluation and spirometry to measure the FEV1 and carbon monoxide diffusion capacity (DLCO). Depending on these results the patients can be stratified into different risk groups and further testing may be required or surgery can be initiated. Pulmonary reserve is measured by spirometry. The minimum forced vital capacity (FVC) for pneumonectomy in men is 2 liters. The minimum for lobectomy is 1.5 liters. In women, the minimum FVC values for pneumonectomy and lobectomy are 1.75 liters and 1.25 liters respectively.[3]
Surgery Procedures include wedge resection (removal of part of a lobe), lobectomy (one lobe), bilobectomy (two lobes) or pneumonectomy (whole lung). In patients with adequate respiratory reserve, lobectomy is the preferred option, as this minimizes the chance of local recurrence. If the patient does not have enough functional lung for this, wedge resection may be performed.[4] Radioactive iodine brachytherapy at the margins of wedge excision may reduce recurrence to that of lobectomy.[5]
Also, many times during lung cancer surgery, the doctor will remove some of the lymph nodes to test for cancer. If the lymph nodes test positive for cancer then that is indicative of the disease spreading beyond the lung. There will most likely be subsequent treatments to help eliminate the remaining cancer.
Patient Selection Not all patients are suitable for operation. The stage, location and cell type are important limiting factors. In addition, patients who are very ill with a poor performance status or who have inadequate pulmonary reserve would be unlikely to survive. Even with careful selection, the overall operative death rate is about 4.4%.[1]
Stage "Stage" refers to the degree of spread of the cancer.
See non-small cell lung cancer staging
In non-small cell lung cancer, stages IA, IB, IIA, and IIB are suitable for surgical resection.[6] Stages IIIA, IIIB, and IV tend to involve the spreading out of the cancer. In that case chemotherapy or radiation is usually deemed the appropriate action to take because surgery will not adequately solve the diseased lungs.
Types of Surgery Lobectomy (removal of a lobe of the lung) Pneumonectomy (removal of an entire lung) Wedge resection Sleeve resection
Overview
Surgery is the best treatment option of lung cancer for patients with resectable tumors. The feasibility of surgery depends on the stage of lung cancer at the time of diagnosis.
Indications
- Surgical intervention is not recommended for the management of [disease name].
OR
- Surgery is not the first-line treatment option for patients with [disease name]. Surgery is usually reserved for patients with either:
- [Indication 1]
- [Indication 2]
- [Indication 3]
- The mainstay of treatment for [disease name] is medical therapy. Surgery is usually reserved for patients with either:
- [Indication 1]
- [Indication 2]
- [Indication 3]
Surgery
- Surgery is the best treatment option of lung cancer for patients with resectable tumors.
- The feasibility of surgery depends on the stage of lung cancer at the time of diagnosis.
- The procedures for lung cancer imclude:[3][4]
- Wedge resection (removal of part of a lobe)
- Wedge resection is performed in the patients who do not have adequate respiratory reserve.
- Radioactive iodine brachytherapy at the margins of wedge resection may reduce recurrence to that of lobectomy.
- Lobectomy (one lobe)
- Lobectomy is the preferred option for patients with adequate respiratory reserve because it reduces the chances of local recurrence.
- Bilobectomy (two lobes)
- Pneumonectomy (whole lung)
- Wedge resection (removal of part of a lobe)
Contraindications
References
- ↑ href="https://radiopaedia.org/">Radiopaedia.org</a>. From the case <a href="https://radiopaedia.org/cases/10494">rID: 10494
- ↑ href="https://radiopaedia.org/">Radiopaedia.org</a>. From the case <a href="https://radiopaedia.org/cases/30005">rID: 30005</a>
- ↑ El-Sherif, A (Aug 2006). "Outcomes of sublobar resection versus lobectomy for stage I non-small cell lung cancer: a 13-year analysis". Annals of Thoracic Surgery. 82 (2): 408–415. PMID 16863738. Unknown parameter
|coauthors=ignored (help) - ↑ Fernando, HC (Feb 2005). "Lobar and sublobar resection with and without brachytherapy for small stage IA non-small cell lung cancer". Journal of Thoracic and Cardiovascular Surgery. 129 (2): 261–267. PMID 15678034. Unknown parameter
|coauthors=ignored (help)
Lung Cancer Differential
| Condition/disease | Signs/symptoms | Tests |
| Pneumonia/bronchitis | Typical symptoms include fever, cough, dyspnea, and chest pain; recurrent pneumonia or bronchitis in a smoker or former smoker should raise the suspicion of lung cancer | CXR is the first test performed; CT imaging can be helpful to evaluate pulmonary masses that might not be well visualised with chest x-ray; bronchoscopy can also be used to assess for endobronchial lesions or to biopsy suspicious pulmonary masses |
| Carcinoid tumor | Often asymptomatic with normal physical examination; may cause cough, dyspnea, hemoptysis, unilateral wheezing, or post-obstructive pneumonia if the tumor is endobronchial or compressing the central bronchi | CT chest: 80% of carcinoid tumors appear as an endobronchial nodule and 20% as a parenchymal nodule, with smooth, rounded borders and is highly vascularized; flexible bronchoscopy shows raised, pink, vascular, lobulated lesions; endobronchial forceps biopsy is usually required for pathology to be diagnostic; bronchial brushings, sputum specimens, and lavage fluid rarely provide sufficient tissue for a conclusive diagnosis |
| Metastatic cancer from a non-thoracic primary site | Signs and symptoms depend on the location of the primary tumor and distant disease and may include pain, weight loss, malaise, cough, dyspnea, clubbing, or focal wheezing; physical findings may be present depending on the location and extent of the disease | CT chest shows one or multiple nodules of variable sizes from diffuse micronodular opacities (miliary) to well-defined masses, lesions are often irregular and in the periphery of the lower lung zones; CT/MRI head, CT abdomen and pelvis: extrapulmonary cancers that commonly metastasis to the lung include melanoma, thyroid carcinoma, esophageal cancer; ovarian cancer; sarcomas; and adenocarcinomas of the colon, breast, kidney, and testis; PET-FDG scan shows increased uptake in both primary and distant sites, certain metastatic lesions, such as renal cell carcinoma, have a lower probability of 18-fluorodeoxyglucose (FDG) uptake; CT-guided transthoracic needle aspiration (TTNA) can reveal characteristic malignant cells, pneumothorax complicates 20% to 30% of TTNA procedures, the choice between bronchoscopy and TTNA is based on lesion size, location, risks, and local expertise; biopsy during flexible bronchoscopy and biopsy may show characteristic malignant cells, bronchoscopy has a 100% yield for endobronchial lesions (which are extremely rare in metastatic deposits from other primary tumors) |
| Infectious granuloma | History may include travel to endemic areas, pet/animal exposures, and specific leisure activities (e.g., caving); may feature cough, dyspnea, hemoptysis, weight loss, fever, joint aches, skin lesions, and night sweats, or no symptoms; many possible causes: Histoplasma capsulatum, Mycobacterium tuberculosis, Coccidioides immitis, Cryptococcus neoformans, Aspergillus, Pseudallescheria boydii, Fusarium species, zygomycetes, and others; non-specific skin findings may be seen in atypical mycobacteria and cryptococcosis; lymphadenopathy may be present with active disease | CT-guided TTNA can be used for diagnostic sampling, pneumothorax complicates 20% to 30% of TTNA procedures, the choice between bronchoscopy and TTNA is based on lesion size, location, risks, and local expertise; CT chest typically shows lesions <2 cm diameter and round with smooth borders, old granulomatous disease may feature central, laminated, or diffuse calcification pattern, mediastinal lymphadenopathy without calcifications is sometimes present, nodules from angioinvasive fungi (e.g., Aspergillus, Pseudallescheria boydii, Fusarium species, and zygomycetes) may demonstrate the "halo sign" (ground-glass opacity surrounding the nodule), occasionally, calcifications can be seen in the spleen or liver; fungal serologies: positive during active infection; flexible bronchoscopy and biopsy can sometimes provide sample for identification and culture and sensitivity of organism; PET: usually negative (<2.5 standardised uptake values), may be positive in active infectious processes |
| Sarcoidosis | Cough, dyspnea, fatigue weight loss, fever, night sweats, rash, eye pain, photophobia blurred vision, and red eye; pulmonary examination is usually unrevealing; can affect any organ, so physical findings depend on specific organs affected; skin lesions including maculopapular eruptions, subcutaneous nodular lesions, and red-purple skin lesions | CT chest: mediastinal adenopathy often present with sarcoid. Sarcoid nodules have predilection for upper zones, although can be located throughout the lung; flexible bronchoscopy and biopsy can demonstrate presence of non-caseating granulomas; CT-guided TTNA can provide access to material from some lesions inaccessible to flexible bronchoscopy; laboratory markers: ACE elevation may be seen in sarcoidosis but is non-specific |
| Rheumatoid arthritis | Arthralgias, pain, skin nodules, pleural effusions, pleuritis, joint pain, and deformity | CT chest typically shows lung nodule 3 mm to 7 cm, predominantly in peripheral upper and mid-lung zones, may show cavitation; flexible bronchoscopy and biopsy shows rheumatoid necrobiotic nodule, necrobiotic nodules demonstrate a central zone of eosinophilic fibrinoid necrosis surrounded by palisading fibroblasts, the nodule often centered on necrotic inflamed blood vessels; laboratory markers: patients with lung nodules due to rheumatoid arthritis frequently have high levels of rheumatoid factor, although seronegative cases have been reported |
| Wegener's granulomatosis | Cough, chest pain, dyspnea, hemoptysis, rhinorrhoea, epistaxis, ear/sinus pain, hoarseness, fever, fatigue, anorexia, weight loss, palpable purpura, painful ulcers, uveitis, upper airway inflammation, and sinus pain | CT chest shows solitary or multiple lung nodules, airways are frequently affected; Flexible bronchoscopy or CT-guided TTNA may show necrotising granulomatous inflammation; laboratory markers: anti-neutrophil cytoplasmic antibody (ANCA), ANCA testing results depend on the extent and severity of the disease |
| Arteriovenous malformation | Dyspnea is uncommon, may cause hemoptysis, pulmonary bruit, arteriovenous communications, or hemorrhagic telangiectasia in the skin, mucous membranes, and other organs, cyanosis and finger clubbing may be present, eurological symptoms from cerebral aneurysms, cerebral emboli | CT chest shows round or oval nodule(s) with feeding artery and draining vein often identified, most common in lower lobes, multiple lesions in 30% of cases, usually round or oval, ranging from 1 cm to several cm in diameter; pulmonary angiography confirms presence and location of AVMs, identifies feeding arterial and venous structures, in cases of significant hemoptysis, pulmonary angiogram is combined with bronchial artery embolisation; ABG analysis may show decreased pO2 and decreased oxygen saturation when AV flow is severe., in cases of severe systemic AVMs, chronic hypoxemia may cause polycythemia |
| Amyloidosis | Weight loss, paresthesias, dyspnea, and fatigue are the most common symptoms associated with amyloidosis and are common to all systemic forms; weight loss of >9 kg is common; small vessel involvement can cause jaw or limb claudication, and rarely angina; amyloid purpura is present in about 1 in 6 patients, typically peri-orbital; eyelid petechiae are common; hepatomegaly >5 cm below the right costal margin is seen in 10% of patients and splenomegaly is usually of modest degree | CT chest shows lung involvement characterised by focal pulmonary nodules, tracheobronchial lesions, or diffuse alveolar deposits; serum immunofixation shows presence of monoclonal protein; urine immunofixation shows presence of monoclonal protein; immunoglobulin free light chain assay shows abnormal kappa to lambda ratio |
| Bronchiolitis obliterans organizing pneumonia (BOOP) | Normally presents as a flu-like illness followed by a second illness lasting 1 to 4 months, with low-grade fever, non-productive cough, malaise, dyspnea, and weight loss; sometimes features pleuritic chest pain and hemoptysis; in most patients, auscultation reveals fine, dry lung crackles; finger clubbing is unusual | CT chest typical features include: patchy "ground-glass" opacities in a sub-pleural and/or peribronchovascular distribution; thickening of bronchial walls and cylindrical dilation; 3 to 5 mm diameter centrilobular nodules or other ill-defined nodules, mediastinal lymphadenopathy, pleural effusions; pulmonary function tests typically show a restrictive pattern; bronchoalveolar lavage (BAL shows a mixed cell pattern, with an increase in lymphocytes, neutrophils, eosinophils, mast cells, foamy macrophages, and occasional plasma cells, CD4+/CD8+ cell ratio is decreased, the ratio of lymphocytes to CD8+ cells is significantly increased; transbronchial lung biopsy in combination with BAL can be a useful approach, prior to possible open biopsy; open lung biopsy is often required for a definitive diagnosis |
| Pulmonary tuberculosis | Cough longer than 2 to 3 weeks, discolored or bloody sputum, night sweats, weight loss, loss of appetite, and/or pleuritic chest pain | Chest x-ray: primary disease commonly presents as middle and lower lung zone infiltrates, ipsilateral adenopathy, atelectasis from airway compression, and pleural effusion can be seen, reactivation-type (post-primary) pulmonary TB usually involves apical and/or posterior segment of right upper lobe, apicoposterior segment of left upper lobe, or superior segment of either lower lobe, with or without cavitation, as disease progresses it spreads to other segments/lobes; sputum smear: positive for acid-fast bacilli (AFB), sputum may be spontaneously expectorated or induced, and at least 3 specimens should be collected (minimum 8 hours apart, including an early morning specimen, which is the best way to detect Mycobacterium tuberculosis), organisms other than M. tuberculosis, especially on-tuberculous mycobacteria (e.g., M. kansasii and M. avium , may be positive for AFB stain; nucleic acid amplification tests (NAAT): positive for M. tuberculosis DNA or RNA amplification tests for rapid diagnosis, may be used on sputum or any sterile body fluid |
| Non-Hodgkin's lymphoma (NHL) | Aggressive NHL may present with fever, drenching night sweats, malaise, weight loss, cough, shortness of breath, abdominal discomfort, headache, change in mental status, dizziness, ataxia, pleural effusion, lymphadenopathy, pallor, purpura, jaundice, hepatomegaly, splenomegaly, skin nodules, and abnormal neurological examination, low-grade NHL patients often minimally symptomatic or asymptomatic | CT chest: frequently anterior mediastinum, can determine if mass is cystic or solid and whether it contains calcium or fat, contrast enhancement provides information concerning vascularisation of the mass and relationship to adjacent structures; FBC with differential: shows thrombocytopenia, pancytopenia; Blood smear: shows nucleated red blood cells, giant platelets; lymph node biopsy with immunohistochemistry: shows characteristic cells, preferably obtain excisional or core biopsy to provide information on lymph node architecture; mediastinoscopy: used to sample mediastinal nodes |
| Hodgkin's lymphoma | Predominantly a disease of young adults; most patients present with a several-month history of persistent adenopathy, most commonly of the cervical chain | Plain chest x-ray: typically shows mediastinal mass/large mediastinal adenopathy; PET scan: involved sites appear fluorodeoxyglucose (FDG)-avid (bright) with PET imaging; lymph node biopsy with immunohistochemistry: the Hodgkin's cell can be a characteristic Reed-Sternberg cell, or one of its variants, such as the lacunar cell in the nodular sclerosis subtype; in nodular lymphocyte-predominant Hodgkin's lymphoma, the characteristic cell is the lymphocytic and histiocytic (L&H) cell, also referred to as a popcorn cell |
| Thymoma/thymic carcinoma | Approximately 30% of patients with thymoma are asymptomatic at the time of diagnosis; may also present with cough, chest pain, signs of upper airway congestion, superior vena cava syndrome, dysphagia, or hoarseness; may have features of paraneoplastic syndromes associated with thymoma including myasthenia gravis, polymyositis, lupus erythematosus, rheumatoid arthritis, thyroiditis, and Sjogren's syndrome; about 30% of patients have symptoms suggestive of myasthenia gravis (e.g., ptosis, double vision) | Plain chest x-ray: in 50% of the patients, thymomas are detected by chance with plain-film chest radiography; CT chest: 90% occur in anterior mediastinum; Positron emission tomography (PET): may be of value in determining malignancy and extramediastinal involvement; pre-operative biopsy: indicated if there are atypical features or if imaging suggests invasive tumor and patient is under consideration for induction therapy |
| Bronchogenic cyst | Usually diagnosed in infancy and childhood, although 50% are diagnosed after 15 years of age; Approximately 50% of patients are asymptomatic; in adults, chest pain (often pleuritic) and dysphagia (due to esophageal compression) are the most common symptoms; may also feature recurrent cough and chest infection/pneumonia, superior vena cava syndrome, tracheal compression, and pneumothorax | Two-view chest radiography: typically shows a sharply demarcated spherical mass of variable size, most commonly located in the middle mediastinum around the carina, can appear as a solid tumor or show air-fluid level if cyst is infected or contains secretions; CT chest: frequently middle mediastinum, typically at level of the mediastinum, calcifications may also be seen; MRI: frequently middle mediastinum, typically at level of the mediastinum, T2-weighted images show a homogeneous mass of moderate-to-bright intensity, on T1-weighted images, lesions may vary in intensity depending on protein content of the cyst |
| Tracheal tumors | Common symptoms include dyspnea, cough, hemoptysis, wheeze, and stridor; less commonly, hoarseness and dysphagia may be present | Plain chest radiographs are generally insensitive for detection of tracheal tumors, clues that may indicate the presence of a tracheal tumour include abnormal calcification, tracheal narrowing, post-obstructive pneumonia, and/or atelectasis; helical CT enables accurate calculation of tumor volumes and can help differentiate mucosal lesions from submucosal lesions; MRI can be useful in assessing extension into surrounding tissue and vascular anatomy; bronchoscopy allows direct visualisation, opportunity for biopsy, and potential for laser treatment |
| Thyroid mass | Symptoms and signs depend on size of mass; may be visible/palpable as lump on anterior aspect of neck; may present with dysphagia, hoarseness, difficulty breathing, and pain in neck or throat; may also be signs and symptoms of hyper- or hypothyroidism depending on the nature of the mass | Laboratory testing should include thyroid function panel, with TSH, free T4, free T3; I-123 thyroid scan is ordered for patients with overt or subclinical hyperthyroidism a hyperfunctioning (hot) nodule is almost always benign, most nodules are hypofunctioning (cold) (most of these are benign, but malignant nodules are also cold); ultrasound and doppler can be used to define dimensions of thyroid nodules and solid/cystic component(s), features suspicious of malignancy include microcalcifications, a more tall-than-wide shape, hypervascularity, marked hypoechogenicity, or irregular margins, it can also guide fine-needle aspiration, which can reveal malignant cells or cyst fluid; CT neck can evaluate cervical lymph nodes in cases of medullary thyroid cancer, and extension of the scan into the chest can help evaluate a retrosternal thyroid mass |
Other conditions that can be mistaken for lung cancer including the following:
- Pneumomediastinum
- Empyema
- Abscess
- Pneumothorax (tension and traumatic)
- Pleural effusion
- Pneumothorax
- Superior Vena Cava Syndrome
Differential Diagnosis
Lung cancer must be differentiated from other cavitary lung lesions.
| Causes of
lung cavities |
Differentiating Features | Differentiating radiological findings | Diagnosis
confirmation |
|---|---|---|---|
|
|
| |
|
|
|
|
|
|
| |
|
|
|
|
|
|
| |
|
|
| |
|
|
||
|
|
| |
|
|
|
PBC USG
There are no ultrasound findings associated with primary biliary cirrhosis. However, the ultrasound is mandatory for liver and biliary tree for all cholestatic patients for the differentiation of intrahepatic from extrahepatic cholestasis.
- The ultrasound findings may include:[14]
- Cholestasis
- Abdominal lymphadenopathy
Ultrasound examination of the liver and biliary tree is obligatory in all cholestatic patients in order to differentiate intrahepatic from extrahepatic . When the biliary system appears normal and serum AMA are present, no further radiologic workup is necessary. , particularly in the hilar region of the liver, is seen in 80% of patients with PBC
PBC CT
- Findings on CT scan suggestive of advanced primary biliary cirrhosis include:[15]
- Small heterogeneously attenuating liver
- Varices
- Splenomegaly
- Lymphadenopathy
- Findings on CT scan suggestive of less advanced disease include:
- Enlarged or normal size liver
- Smooth contour liver
- Little atrophy
- Lacelike fibrosis
- Regenerative nodules
- Varices
- Ascites
- Lymphadenopathy
Synonoms
- Solitary hyperplastic nodule
- Hepatic hamartoma
- Focal cirrhosis
- Hamartomatous cholangiohepatoma
- Hepatic pseudotumor
Historical Perspective
- In early 1900s,Focal nodular hyperplasia was first described.
- Between 1918-1982,96.625 autopsy studies were conducted out of which 8 percent of nonhemangiomatous lesions were focal nodular hyperplasia.
- In 1994,Working party of the world congresses of gastroenterology suggested a standardized terminology of nodular hepatic lesions that placed Focal noduldar carcinoma in the group of regenerative nodules, as opposed to dysplastic or neoplastic nodules.[16]
Differentiating Focal nodular hyperplasia from Other diseases
Focal nodular hyperplasia must be differentiated from:
- Hepatocellular carcinoma
- Cholangiocarcinoma
- Pancreatic carcinoma
- Liver hemangioma
- Liver abscess
- Cirrhosis
- Inflammatory lesions
Abbreviations: RUQ= Right upper quadrant of the abdomen, LUQ= Left upper quadrant, LLQ= Left lower quadrant, RLQ= Right lower quadrant, LFT= Liver function test, SIRS= Systemic inflammatory response syndrome, ERCP= Endoscopic retrograde cholangiopancreatography, IV= Intravenous, N= Normal, AMA= Anti mitochondrial antibodies, LDH= Lactate dehydrogenase, GI= Gastrointestinal, CXR= Chest X ray, IgA= Immunoglobulin A, IgG= Immunoglobulin G, IgM= Immunoglobulin M, CT= Computed tomography, PMN= Polymorphonuclear cells, ESR= Erythrocyte sedimentation rate, CRP= C-reactive protein, TS= Transferrin saturation, SF= Serum Ferritin, SMA= Superior mesenteric artery, SMV= Superior mesenteric vein, ECG= Electrocardiogram
| Disease | Clinical manifestations | Diagnosis | Comments | |||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Symptoms | Signs | |||||||||||||||
| Abdominal Pain | Fever | Rigors and chills | Nausea or vomiting | Jaundice | Constipation | Diarrhea | Weight loss | GI bleeding | Hypo-
tension |
Guarding | Rebound Tenderness | Bowel sounds | Lab Findings | Imaging | ||
| Focal nodular hyperplasia | Diffuse | ± | − | − | ± | − | − | + | + | − | − | − | Normal |
|
|
|
| Hepatocellular carcinoma/Metastasis | RUQ | + | − | + | + | + | + | + | + | + | − | + |
|
|
Other symptoms: | |
| Cholangiocarcinoma | RUQ | + | − | + | + | − | − | + | − | − | − | + | Normal |
|
| |
| Pancreatic carcinoma | MidEpigastric | − | − | + | + | + | − | + | − | − | − | + | Normal |
Skin manifestations may include: | ||
| Disease | Abdominal Pain | Fever | Rigors and chills | Nausea or vomiting | Jaundice | Constipation | Diarrhea | Weight loss | GI bleeding | Hypo-
tension |
Guarding | Rebound Tenderness | Bowel sounds | Lab Findings | Imaging | Comments |
| Gallbladder cancer | Midepigastric | − | − | + | + | − | + | + | − | − | − | − | Normal |
|
||
| Liver hemangioma | Intermittent RUQ | − | − | + | + | − | − | − | − | − | − | − | Normal |
|
| |
| Liver abscess | RUQ | + | − | + | + | − | − | + | − | − | − | − | Normal |
|
|
|
| Cirrhosis | RUQ+Bloating | + | − | + | + | − | − | + | − | − | − | − | Normal |
|
US
|
|
| Inflammatory lesions | RUQ | ± | − | + | + | − | − | − | − | − | − | − | Normal |
|
US
|
|
Focal Nodular Hyperplasia
FNH is typically benign, and usually no treatment is needed. Hemangiomas are the most common and are entirely benign. Treatment is unnecessary unless their expansion causes symptoms
Focal nodular hyperplasia (FNH) is a non-malignant hepatic tumor that is not of vascular origin. In one autopsy series of 96,625 patients, 8 percent of non-hemangiomatous lesions were FNH, representing 66 percent of all benign non-hemangiomatous lesions seen between 1918 and 1982 [1]. In large retrospective studies of patients referred for ultrasound and multidetector computed tomography, the prevalence of focal nodular hyperplasia was 0.2 percent and 1.6 percent, respectively [2,3].
FNH is seen in both sexes and throughout the age spectrum, although it is found predominantly in women (in a ratio of 8 or 9:1) between the ages of 20 and 50 years [4]. FNH comprises up to 2 percent of liver tumors in children [5].
This topic review will focus on the pathogenesis, clinical manifestations and management of FNH. An approach to patients presenting with a focal liver lesion is discussed separately. (See "Solid liver lesions: Differential diagnosis and evaluation".)
PATHOGENESIS — FNH has various labels: solitary hyperplastic nodule, hepatic hamartoma, focal cirrhosis, hamartomatous cholangiohepatoma, and hepatic pseudotumor. This profusion of terms epitomizes the confusion surrounding our understanding of the pathogenesis of the many conditions in which nodules of benign appearing hepatocytes are found. The International Working Party of the World Congresses of Gastroenterology proposed a standardized nomenclature in 1994, which placed FNH in the group of regenerative nodules, as opposed to dysplastic or neoplastic nodules [6]. This fits well with our current understanding of the pathogenesis of FNH. The contention that this lesion is non-neoplastic has been bolstered by the reported polyclonal origin of the hepatocytes [7], although this is disputed by others [8].
Previously considered to be a hamartoma, a neoplasm, a response to ischemia or other injury, or a focal area of regeneration, FNH is now generally accepted to be a hyperplastic (regenerative) response to hyperperfusion by the characteristic anomalous arteries found in the center of these nodules [4,9,10]. Whether vascular injury is also involved is less clear, but FNH is occasionally supplied primarily by portal venous blood due to thrombosis of the anomalous central artery [11].
The association of FNH with hereditary hemorrhagic telangiectasia (Osler-Weber-Rendu disease [12]) and hepatic hemangiomas strengthens the hypothesis that FNH is a congenital vascular anomaly. Two pathology studies found cavernous hemangiomas in 6.5 and 2.3 percent of patients with FNH [13,14] and an imaging study, using ultrasound and dynamic CT, found that 23 percent of FNH patients had associated hemangiomas [15]. Multiple FNH lesions have also been noted in association with hemihypertrophy and vascular malformations (Klippel-Trénaunay-Weber syndrome) [16]. FNH with similar clinical and radiographic features has been documented in identical twins supporting a role of congenital vascular anomalies in its pathogenesis and a possible genetic predisposition to the disease [17].
PATHOLOGY — FNH is most often solitary (80 to 95 percent), and usually less than 5 cm in diameter. Only 3 percent are larger than 10 cm, although FNH as large as 19 cm have been reported [1,13,18]. It has a sharp margin with no capsule and may be pedunculated. The characteristic finding is the presence of a central stellate scar (picture 1) containing an inappropriately large artery with multiple branches radiating through the fibrous septa to the periphery. These branches divide the mass into multiple small nodules or cords of normal appearing hepatocytes (picture 2). The scar-like tissues within FNH nodules are composed of abnormally large portal tracts including large feeding arteries, portal veins, and bile ducts [10].
The arteries drain into adjacent hepatic veins. This radiating, branching pattern produces the spoke and wheel image typically seen on angiography. Although normal bile ducts are absent, bile ductules derived from hepatocyte metaplasia are usually prominent, traveling along the fibrous septa (picture 3). Sinusoids and Kupffer cells are typically present, distinguishing it from hepatocellular adenoma (HA), which usually lacks bile ducts and Kupffer cells [1,13,14,18,19]. The minimal microscopic criteria for the diagnosis of classical FNH are nodular architecture, abnormal vessels, and proliferation of bile ductules [13]. Lymphocyte infiltration, canalicular bile plugs, copper deposition, and feathery degeneration of hepatocytes may suggest cholestasis and/or inactive cirrhosis. Irregular intimal fibrosis or hypertrophy of the media may be seen in large arteries and veins, at times even occluding the lumen [13,14,19]. When present, portal veins are dilated and/or stenotic [10].
Non-classical variants — Non-classical forms of FNH lack either the typical nodular architecture or vascular malformations, but always contain bile ductular proliferation. They almost always lack the characteristic central scar [13]. Three variants have been recognized:
●The most common of these, the telangiectatic type, often presents with multiple FNH. In addition to the lack of a central scar, the mass is characterized by the absence of nodular architecture and the presence of single, quite regular plates of hepatocytes separated by sinusoids fed directly by anomalous arteries [13,20]. The risk of bleeding appears to be similar to the risk observed in patients with a hepatic adenoma [21].
●A mixed hyperplastic and adenomatous form may be difficult to distinguish from HA due to its subtle vascular and bile ductular findings [13,20].
●A third histologic variant consisting of FNH with cytologic atypia resembling dysplasia of large cell type has been proposed [13].
A comprehensive pathological study of 305 lesions failed to identify a macroscopic central stellate scar in 50 percent and noted non-classical histology in 20 percent of the lesions, most showing a telangiectatic variant [13]. The surprisingly high number of lesions without a central scar was almost exclusively due to the large number of masses that had non-classical histology. Ninety-five percent of those with non-classical histology did not have a scar, whereas only 18 percent of those with classical histology lacked a scar [13]. The overall prevalence and clinical significance of these variants remains to be determined.
DIAGNOSIS — The diagnosis of FNH is usually made by demonstrating its characteristic features on imaging tests and excluding other lesions. The latter can typically be accomplished by assessment of the context in which FNH is detected and by obtaining specific radiologic and laboratory testing (table 1). The differential diagnosis includes hepatic adenoma, hepatocellular carcinoma, fibrolamellar carcinoma, cirrhosis, large regenerative nodules, hemangioma, and hypervascular metastases. (See "Solid liver lesions: Differential diagnosis and evaluation".)
Symptoms — The majority of reports have found that symptoms or signs directly attributable to FNH are infrequent. Two-thirds to three-fourths of patients are identified incidentally [18], with the mass noted at the time of surgery, on an abdominal imaging study, or at autopsy. Unlike hepatic adenomas, FNH rarely presents with acute onset of hemorrhage, necrosis, or infarction [22,23].
However, symptomatic presentations have been described. In one series, for example, abdominal discomfort or a palpable liver mass was observed in 25 percent of 41 patients [24]. Another series that included 168 patients found that 60 percent had abdominal pain and 4 percent had an abdominal mass [13]. The high number of symptomatic patients in the second report probably reflects selection bias since all of the patients were identified from pathology specimens obtained at the time of surgical resection [13].
Laboratory tests — Liver tests are most often normal although minor elevations in aspartate and alanine aminotransferase, alkaline phosphatase and gamma glutamyl transpeptidase levels may be seen [13,14,24]. The alpha-fetoprotein is normal.
Imaging tests — A confident diagnosis can usually be made through a combination of imaging modalities; tissue diagnosis is usually not required.
Ultrasonography — Although often first identified on ultrasound examination, FNH is variably hyper, hypo, or isoechoic [24] and US is able to identify the central scar in only 20 percent of cases [25]. The ultrasound characteristics are difficult to distinguish from an adenoma or malignant lesions. Power Doppler ultrasound may help differentiate the arterial flow in FNH from the venous flow in HA [24,26,27].
Contrast-enhanced ultrasonography — Several reports have described improved characterization of focal liver lesions using contrast-enhanced ultrasonography compared with standard ultrasonography [28-30]. While the approach is not approved in the United States, it is available in other countries. Test characteristics compared with other imaging modalities remain incompletely defined, although emerging data suggest its ability for differentiation among solid liver lesions is comparable with MRI [31]. (See "Contrast-enhanced ultrasound for the evaluation of liver lesions".)
CT scan — A properly timed dynamic, triphasic, helical CT scan performed without contrast, and with contrast during the hepatic arterial and portal venous phases, will often be highly suggestive of the diagnosis [32,33]. The lesion may be hypo or isodense on non-contrast imaging with the central scar identified in one-third of patients. The lesion becomes hyperdense during the hepatic arterial phase due to the arterial origin of its blood supply (image 1). FNH is generally isodense during the portal venous phase, although the central scar may become hyperdense as contrast diffuses into the scar. While characteristic of FNH, a central scar may be present in the fibrolamellar variant of HCC. (See "Epidemiology, clinical manifestations, diagnosis, and treatment of fibrolamellar carcinoma", section on 'Imaging'.)
MRI — There may be little to distinguish FNH from normal liver on standard MRI, since it is composed of the same elements as normal liver. An isointense lesion is noted on T1-weighted images, while an isointense to slightly hyperintense mass appears on T2-weighted images (image 2 and image 3) [34]. The scar typically shows high signal intensity on T2-weighted images due to vessels or edema in the scar (image 3) [35]. Gadolinium infusion produces rapid enhancement of the FNH mass due to its arterial blood supply, producing a hyperintense lesion on early films (image 2). On delayed images it becomes more isointense with respect to normal liver. The central scar enhances on delayed imaging as contrast gradually diffuses into the fibrous center of the mass [36-39]. In one study, gadolinium enhanced MRI had a sensitivity and specificity of 70 and 98 percent, respectively [24].
A relatively new MR contrast agent has been introduced into clinical use. Unlike currently used gadolinium-based contrast agents for MRI, this agent, a Gd-BOPTA chelate of Gadobenate Dimeglumine, has a dual route of elimination, through both renal and hepatobiliary excretion (image 4). Thus, it can be useful for distinguishing hepatic adenomas from focal nodular hyperplasia. (See "Solid liver lesions: Differential diagnosis and evaluation".)
Angiography — Although angiography may reveal the diagnostic "spoked wheel" appearance of FNH, its use is rarely indicated [32,40,41].
ROLE OF ORAL CONTRACEPTIVES — FNH was first described in the early 1900s, long before the advent of oral contraceptives (OCPs). It is seen in men and children who do not use OCPs and its incidence remained steady after the introduction of OCPs in 1960, in sharp contrast to the dramatic rise in the incidence of HA with the widespread use of OCPs. Thus use of OCPs is not required for the development of FNH [42-44].
On the other hand, FNH may be responsive to estrogens [11]. Patients taking OCPs tend to have larger, more vascular tumors, have more symptoms, and reports of hemorrhage or rupture in patients with FNH have all occurred in patients taking OCPs [45-48]. However, the magnitude of the risk associated with OCPs is uncertain. In a study of 216 women with FNH, use of OCPs did not appear to influence the size or number of FNH lesions or size changes (which were rare) during follow-up for an average of two years [49]. A case control trial comparing 23 women with histologically confirmed FNH to 94 controls estimated the odds ratio of OCP use to be 2.8 (95% CI, 0.8 to 9.4) for those who had ever used OCPs and 4.5 (95% CI, 1.2 to 16.9) for those who had ≥3 years of use [50].
We generally do not insist that oral contraceptives and other estrogen containing preparations should be discontinued. However, it is reasonable to obtain a follow-up imaging study in 6 to 12 months in women who continue taking these drugs.
MANAGEMENT — The natural history of FNH is one of stability and lack of complications. Lesions generally do not change over time, although they occasionally become smaller [49,51-54]. However, as mentioned above, enlargement of FNH in the setting of OCPs and during pregnancy have been reported [55]. There is no evidence for malignant transformation of FNH [13,24,56,57].
Patients who are suspected of having FNH based upon the evaluation described above should be managed conservatively [24,35,49,51,52,54,58,59]. If a diagnosis remains unclear, a liver biopsy may be helpful, but may also be misleading since only resection will be definitive [60]. Follow-up studies at three and six months will often be sufficient to confirm the stability of the lesion and its benign nature, after which no long-term follow-up is required routinely. Surgery should be reserved for the rare, very symptomatic FNH lesion, and the highly suspicious lesion, which has eluded diagnosis by all other modalities.
We generally do not insist that oral contraceptives and other estrogen containing preparations should be discontinued. However, it is reasonable to obtain a follow-up imaging study in 6 to 12 months in women who continue taking these drugs. Small FNH do not appear to pose a significant risk to a successful pregnancy [49,61], although close observation is strongly recommended and resection may be prudent for large (>8 cm) FNH.
SUMMARY AND RECOMMENDATIONS
●Focal nodular hyperplasia (FNH) is a non-malignant hepatic tumor that is not of vascular origin. It is now generally accepted to be a hyperplastic (regenerative) response to hyperperfusion by the characteristic anomalous arteries found in the center of these nodules. (See 'Pathogenesis' above.)
●FNH is most often solitary (80 to 95 percent) and usually less than 5 cm in diameter. Only 3 percent are larger than 10 cm. (See 'Pathology' above.)
●The majority of reports have found that symptoms or signs directly attributable to FNH are infrequent. (See 'Symptoms' above.)
●The diagnosis of FNH is usually made by demonstrating its characteristic features on imaging tests and excluding other lesions. The latter can typically be accomplished by assessing the context in which FNH is detected and by obtaining specific radiologic and laboratory testing (table 1). (See 'Diagnosis' above and "Solid liver lesions: Differential diagnosis and evaluation".)
●The natural history of FNH is one of stability and a lack of complications. Thus, we suggest that patients who are suspected of having FNH based upon the evaluation described above be managed conservatively (Grade 2B). (See 'Management' above.)
●FNH may be responsive to exogenous estrogens. We generally do not insist that oral contraceptives and other estrogen-containing preparations should be discontinued. However, it is reasonable to obtain a follow-up imaging study in 6 to 12 months in women who continue taking these drugs. (See 'Role of oral contraceptives' above.)
Use of UpToDate is subject to the Subscription and License Agreement.
REFERENCES
1.Craig J, Peters R, Edmundson H. Tumors of the Liver and Intrahepatic Bile Ducts, Fasc 26, 2nd ed, DC Armed Forces Institute of Pathology, Washington, DC 1989. p.6.
2.Kaltenbach TE, Engler P, Kratzer W, et al. Prevalence of benign focal liver lesions: ultrasound investigation of 45,319 hospital patients. Abdom Radiol (NY) 2016; 41:25.
3.Horta G, López M, Dotte A, et al. [Benign focal liver lesions detected by computed tomography: Review of 1,184 examinations]. Rev Med Chil 2015; 143:197.
4.Wanless IR, Mawdsley C, Adams R. On the pathogenesis of focal nodular hyperplasia of the liver. Hepatology 1985; 5:1194.
5.Reymond D, Plaschkes J, Lüthy AR, et al. Focal nodular hyperplasia of the liver in children: review of follow-up and outcome. J Pediatr Surg 1995; 30:1590.
6.International Working Party. Terminology of nodular hepatocellular lesions. Hepatology 1995; 22:983.
7.Paradis V, Laurent A, Flejou JF, et al. Evidence for the polyclonal nature of focal nodular hyperplasia of the liver by the study of X-chromosome inactivation. Hepatology 1997; 26:891.
8.Gaffey MJ, Iezzoni JC, Weiss LM. Clonal analysis of focal nodular hyperplasia of the liver. Am J Pathol 1996; 148:1089.
9.Fukukura Y, Nakashima O, Kusaba A, et al. Angioarchitecture and blood circulation in focal nodular hyperplasia of the liver. J Hepatol 1998; 29:470.
10.Kondo F, Nagao T, Sato T, et al. Etiological analysis of focal nodular hyperplasia of the liver, with emphasis on similar abnormal vasculatures to nodular regenerative hyperplasia and idiopathic portal hypertension. Pathol Res Pract 1998; 194:487.
11.Nakanuma Y. Non-neoplastic nodular lesions in the liver. Pathol Int 1995; 45:703.
12.Wanless IR, Gryfe A. Nodular transformation of the liver in hereditary hemorrhagic telangiectasia. Arch Pathol Lab Med 1986; 110:331.
13.Nguyen BN, Fléjou JF, Terris B, et al. Focal nodular hyperplasia of the liver: a comprehensive pathologic study of 305 lesions and recognition of new histologic forms. Am J Surg Pathol 1999; 23:1441.
14.Ishak KG, Rabin L. Benign tumors of the liver. Med Clin North Am 1975; 59:995.
15.Mathieu D, Zafrani ES, Anglade MC, Dhumeaux D. Association of focal nodular hyperplasia and hepatic hemangioma. Gastroenterology 1989; 97:154.
16.Haber M, Reuben A, Burrell M, et al. Multiple focal nodular hyperplasia of the liver associated with hemihypertrophy and vascular malformations. Gastroenterology 1995; 108:1256.
17.Mindikoglu AL, Regev A, Levi JU, et al. Focal nodular hyperplasia in identical twins. Am J Gastroenterol 2005; 100:1616.
18.Goodman ZD. Benign Tumors of the Liver. In: Neoplasms of the Liver, Okuda K, Ishak KD (Eds), Springer, Tokyo 1987. p.105.
19.Klatskin G, Conn H. Neoplasms of the Liver and Intrahepatic Bile Ducts. In: Histopathology of the Liver, Oxford University Press, New York 1993. p.367.
20.Wanless IR, Albrecht S, Bilbao J, et al. Multiple focal nodular hyperplasia of the liver associated with vascular malformations of various organs and neoplasia of the brain: a new syndrome. Mod Pathol 1989; 2:456.
21.Bioulac-Sage P, Rebouissou S, Sa Cunha A, et al. Clinical, morphologic, and molecular features defining so-called telangiectatic focal nodular hyperplasias of the liver. Gastroenterology 2005; 128:1211.
22.Brunt EM, Flye MW. Infarction in focal nodular hyperplasia of the liver. A case report. Am J Clin Pathol 1991; 95:503.
23.Lee MJ, Saini S, Hamm B, et al. Focal nodular hyperplasia of the liver: MR findings in 35 proved cases. AJR Am J Roentgenol 1991; 156:317.
24.Cherqui D, Rahmouni A, Charlotte F, et al. Management of focal nodular hyperplasia and hepatocellular adenoma in young women: a series of 41 patients with clinical, radiological, and pathological correlations. Hepatology 1995; 22:1674.
25.Shamsi K, De Schepper A, Degryse H, Deckers F. Focal nodular hyperplasia of the liver: radiologic findings. Abdom Imaging 1993; 18:32.
26.Bartolozzi C, Lencioni R, Paolicchi A, et al. Differentiation of hepatocellular adenoma and focal nodular hyperplasia of the liver: comparison of power Doppler imaging and conventional color Doppler sonography. Eur Radiol 1997; 7:1410.
27.Wang LY, Wang JH, Lin ZY, et al. Hepatic focal nodular hyperplasia: findings on color Doppler ultrasound. Abdom Imaging 1997; 22:178.
28.Quaia E. The real capabilities of contrast-enhanced ultrasound in the characterization of solid focal liver lesions. Eur Radiol 2011; 21:457.
29.Kang HS, Kim BK, Shim CS. Focal nodular hyperplasia: with a focus on contrast enhanced ultrasound. Korean J Hepatol 2010; 16:414.
30.Bernatik T, Seitz K, Blank W, et al. Unclear focal liver lesions in contrast-enhanced ultrasonography--lessons to be learned from the DEGUM multicenter study for the characterization of liver tumors. Ultraschall Med 2010; 31:577.
31.Seitz K, Bernatik T, Strobel D, et al. Contrast-enhanced ultrasound (CEUS) for the characterization of focal liver lesions in clinical practice (DEGUM Multicenter Trial): CEUS vs. MRI--a prospective comparison in 269 patients. Ultraschall Med 2010; 31:492.
32.Mergo PJ, Ros PR. Benign Lesions of the Liver. In: The Radiologic Clinics of North America, 2, W.B. Saunders, Philadelphia 1998. Vol 36, p.319.
33.Carlson SK, Johnson CD, Bender CE, Welch TJ. CT of focal nodular hyperplasia of the liver. AJR Am J Roentgenol 2000; 174:705.
34.Mattison GR, Glazer GM, Quint LE, et al. MR imaging of hepatic focal nodular hyperplasia: characterization and distinction from primary malignant hepatic tumors. AJR Am J Roentgenol 1987; 148:711.
35.Buetow PC, Pantongrag-Brown L, Buck JL, et al. Focal nodular hyperplasia of the liver: radiologic-pathologic correlation. Radiographics 1996; 16:369.
36.Irie H, Honda H, Kaneko K, et al. MR imaging of focal nodular hyperplasia of the liver: value of contrast-enhanced dynamic study. Radiat Med 1997; 15:29.
37.Mahfouz AE, Hamm B, Taupitz M, Wolf KJ. Hypervascular liver lesions: differentiation of focal nodular hyperplasia from malignant tumors with dynamic gadolinium-enhanced MR imaging. Radiology 1993; 186:133.
38.Rummeny E, Weissleder R, Sironi S, et al. Central scars in primary liver tumors: MR features, specificity, and pathologic correlation. Radiology 1989; 171:323.
39.Mathieu D, Rahmouni A, Anglade MC, et al. Focal nodular hyperplasia of the liver: assessment with contrast-enhanced TurboFLASH MR imaging. Radiology 1991; 180:25.
40.Welch TJ, Sheedy PF 2nd, Johnson CM, et al. Focal nodular hyperplasia and hepatic adenoma: comparison of angiography, CT, US, and scintigraphy. Radiology 1985; 156:593.
41.Rogers JV, Mack LA, Freeny PC, et al. Hepatic focal nodular hyperplasia: angiography, CT, sonography, and scintigraphy. AJR Am J Roentgenol 1981; 137:983.
42.Fechner RE. Benign hepatic lesions and orally administered contraceptives. A report of seven cases and a critical analysis of the literature. Hum Pathol 1977; 8:255.
43.Ishak KG. Hepatic Neoplasms Associated with Contraceptive and Anabolic Steroids in Carcinogenic Hormones. In: Recent Results in Cancer Research, Lingeman CH (Ed), Springer-Verlag, New York 1979. p.73.
44.Geders JM, Haque S, Tesi RJ, et al. A young man with a solitary hepatic mass. Hepatology 1995; 22:655.
45.Nime F, Pickren JW, Vana J, et al. The histology of liver tumors in oral contraceptive users observed during a national survey by the American College of Surgeons Commission on Cancer. Cancer 1979; 44:1481.
46.Aldinger K, Ben-Menachem Y, Whalen G. Focal nodular hyperplasia of the liver associated with high-dosage estrogens. Arch Intern Med 1977; 137:357.
47.Klatskin G. Hepatic tumors: possible relationship to use of oral contraceptives. Gastroenterology 1977; 73:386.
48.Shortell CK, Schwartz SI. Hepatic adenoma and focal nodular hyperplasia. Surg Gynecol Obstet 1991; 173:426.
49.Mathieu D, Kobeiter H, Maison P, et al. Oral contraceptive use and focal nodular hyperplasia of the liver. Gastroenterology 2000; 118:560.
50.Scalori A, Tavani A, Gallus S, et al. Oral contraceptives and the risk of focal nodular hyperplasia of the liver: a case-control study. Am J Obstet Gynecol 2002; 186:195.
51.Weimann A, Ringe B, Klempnauer J, et al. Benign liver tumors: differential diagnosis and indications for surgery. World J Surg 1997; 21:983.
52.Heinemann LA, Weimann A, Gerken G, et al. Modern oral contraceptive use and benign liver tumors: the German Benign Liver Tumor Case-Control Study. Eur J Contracept Reprod Health Care 1998; 3:194.
53.Di Stasi M, Caturelli E, De Sio I, et al. Natural history of focal nodular hyperplasia of the liver: an ultrasound study. J Clin Ultrasound 1996; 24:345.
54.Leconte I, Van Beers BE, Lacrosse M, et al. Focal nodular hyperplasia: natural course observed with CT and MRI. J Comput Assist Tomogr 2000; 24:61.
55.Scott LD, Katz AR, Duke JH, et al. Oral contraceptives, pregnancy, and focal nodular hyperplasia of the liver. JAMA 1984; 251:1461.
56.Wanless IR. Nodular regenerative hyperplasia, dysplasia, and hepatocellular carcinoma. Am J Gastroenterol 1996; 91:836.
57.Rubin RA, Mitchell DG. Evaluation of the solid hepatic mass. Med Clin North Am 1996; 80:907.
58.Belghiti J, Pateron D, Panis Y, et al. Resection of presumed benign liver tumours. Br J Surg 1993; 80:380.
59.De Carlis L, Pirotta V, Rondinara GF, et al. Hepatic adenoma and focal nodular hyperplasia: diagnosis and criteria for treatment. Liver Transpl Surg 1997; 3:160.
60.Fabre A, Audet P, Vilgrain V, et al. Histologic scoring of liver biopsy in focal nodular hyperplasia with atypical presentation. Hepatology 2002; 35:414.
61.Weimann A, Mössinger M, Fronhoff K, et al. Pregnancy in women with observed focal nodular hyperplasia of the liver. Lancet 1998; 351:1251.
HCC Differnetial Table
Abbreviations: RUQ= Right upper quadrant of the abdomen, LUQ= Left upper quadrant, LLQ= Left lower quadrant, RLQ= Right lower quadrant, LFT= Liver function test, SIRS= Systemic inflammatory response syndrome, ERCP= Endoscopic retrograde cholangiopancreatography, IV= Intravenous, N= Normal, AMA= Anti mitochondrial antibodies, LDH= Lactate dehydrogenase, GI= Gastrointestinal, CXR= Chest X ray, IgA= Immunoglobulin A, IgG= Immunoglobulin G, IgM= Immunoglobulin M, CT= Computed tomography, PMN= Polymorphonuclear cells, ESR= Erythrocyte sedimentation rate, CRP= C-reactive protein, TS= Transferrin saturation, SF= Serum Ferritin, SMA= Superior mesenteric artery, SMV= Superior mesenteric vein, ECG= Electrocardiogram
| Disease | Clinical manifestations | Diagnosis | Comments | |||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Symptoms | Signs | |||||||||||||||
| Abdominal Pain | Fever | Rigors and chills | Nausea or vomiting | Jaundice | Constipation | Diarrhea | Weight loss | GI bleeding | Hypo-
tension |
Guarding | Rebound Tenderness | Bowel sounds | Lab Findings | Imaging | ||
| Hepatocellular carcinoma/Metastasis | RUQ | + | − | + | + | + | + | + | + | + | − | + |
|
|
Other symptoms: | |
| Cholangiocarcinoma | RUQ | + | − | + | + | − | − | + | − | − | − | + | Normal |
|
| |
| Pancreatic carcinoma | MidEpigastric | − | − | + | + | + | − | + | − | − | − | + | Normal |
Skin manifestations may include: | ||
| Focal nodular hyperplasia | Diffuse | ± | − | − | ± | − | − | + | + | − | − | − | Normal |
|
|
|
| Disease | Abdominal Pain | Fever | Rigors and chills | Nausea or vomiting | Jaundice | Constipation | Diarrhea | Weight loss | GI bleeding | Hypo-
tension |
Guarding | Rebound Tenderness | Bowel sounds | Lab Findings | Imaging | Comments |
| Gallbladder cancer | Midepigastric | − | − | + | + | − | + | + | − | − | − | − | Normal |
|
||
| Liver hemangioma | Intermittent RUQ | − | − | + | + | − | − | − | − | − | − | − | Normal |
|
| |
| Liver abscess | RUQ | + | − | + | + | − | − | + | − | − | − | − | Normal |
|
|
|
| Cirrhosis | RUQ+Bloating | + | − | + | + | − | − | + | − | − | − | − | Normal |
|
US
|
|
| Inflammatory lesions | RUQ | ± | − | + | + | − | − | − | − | − | − | − | Normal |
|
US
|
|
Classification
Historical Perspective
Pathophysiology
Causes
Differentiating Splenic Rupture from Other Diseases
Epidemiology and Demographics
Risk Factors
Screening
Natural History, Complications and Prognosis
Diagnosis
Diagnostic Study of Choice
History and Symptoms
Physical Examination
Laboratory Findings
Electrocardiogram
X-Ray
MRI
Other Imaging Findings
Other Diagnostic Studies
Algorithms
| Major molecular events in the pathogenesis of HCC | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Genomic alterations | Epigenetic modifications | Growthfactor pathway alterations | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Gene Mutations | Gene Amplification | DNA methylation micro RNA | Micro RNA | LNC RNA | Major Signaling pathways | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| •TERT promoter •TP53 •CTNNB1 •AXIN1 •AXIN2 •ATM •RPS6KA3 •JAK1 •IL6R •IL6ST •ARID1 •ARID2 | •CCND1 •FGF19 •CDKNA2A •CDKNA2B •AXIN1 •IRF2 •MET | GSTP1 •E-Cadherin •CDKNA2 •RASSF1A •SOCS-3 •MIGMT | •MiR-155 •Mir-122 •Mir-224 •Mir-21 | •HULC •HEIH •Dreh •MVIH •HOTAIR •MDIG •LINE1 | •Wnt/β –catenin •Tyrosine kinase pathways EGF HGF/c-MET FGF VEGF •IGF •HIF •TGF β •Hedgehog | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
The incidence of HCC has almost tripled since the early 1980s in the United States where it is the fastest rising cause of cancer-related deaths1. According to population based Surveillance Epidemiology and End Results registry data, the overall HCC age adjusted incidence rates for liver and intrahepatic ducts cancer is as high as 8 per 100,000 underling population in 2010 (Fig. 1) of which at least 6 per 100,000 related to HCC. Men are at approximately three times higher risk than women. Asian men (i.e., Chinese, Korean, Filipino, and Japanese) have the highest age-adjusted incidence rates. However, the largest proportional increases have occurred among Hispanics followed by blacks and non-Hispanic whites, whereas the lowest proportional increases have occurred among Asians. In contrast to Asians/Pacific Islanders, HCC incidence rates are reported to be higher among Hispanics born in the United States than among foreign-born Hispanics2. HCC incidence rates have increased in each successive birth cohort born between 1900 and 19593 (Fig. 2). In addition, the age distribution of HCC patients has shifted to younger ages, with the greatest proportional increases among individuals 45–60 years old (Fig. 2). There is a south to north gradient in the incidence and mortality of HCC; Southern states including Texas, Louisiana, and Mississippi have some of the highest HCC incidence rates in the nation (Fig. 3). In one study, Texas Latino and especially South Texas Latinos had the highest age-adjusted HCC incidence rates (as high as 10.6/100,000)4.
Video codes
Normal video
{{#ev:youtube|dU26cGlmkRg}} {{#ev:youtube|4uSSvD1BAHg}} {{#ev:youtube|PQXb5D-5UZw}} {{#ev:youtube|UVJYQlUm2A8}}
Video in table
Floating video
| Title |
| https://https://www.youtube.com/watch?v=o1VTHP_oYD8}} |
Redirect
- REDIRECTEsophageal web
synonym website
https://mq.b2i.sg/snow-owl/#!terminology/snomed/10743008
Image

Image to the right
 |
Image and text to the right
<figure-inline><figure-inline><figure-inline><figure-inline><figure-inline><figure-inline><figure-inline><figure-inline><figure-inline><figure-inline><figure-inline><figure-inline><figure-inline><figure-inline><figure-inline><figure-inline><figure-inline><figure-inline><figure-inline><figure-inline><figure-inline> </figure-inline></figure-inline></figure-inline></figure-inline></figure-inline></figure-inline></figure-inline></figure-inline></figure-inline></figure-inline></figure-inline></figure-inline></figure-inline></figure-inline></figure-inline></figure-inline></figure-inline></figure-inline></figure-inline></figure-inline></figure-inline> Recent out break of leptospirosis is reported in Bronx, New York and found 3 cases in the months January and February, 2017.
</figure-inline></figure-inline></figure-inline></figure-inline></figure-inline></figure-inline></figure-inline></figure-inline></figure-inline></figure-inline></figure-inline></figure-inline></figure-inline></figure-inline></figure-inline></figure-inline></figure-inline></figure-inline></figure-inline></figure-inline></figure-inline> Recent out break of leptospirosis is reported in Bronx, New York and found 3 cases in the months January and February, 2017.
Gallery
-
Histopathology of a pancreatic endocrine tumor (insulinoma). Source:https://librepathology.org/wiki/Neuroendocrine_tumour_of_the_pancreas[18]
-
Histopathology of a pancreatic endocrine tumor (insulinoma). Chromogranin A immunostain. Source:https://librepathology.org/wiki/Neuroendocrine_tumour_of_the_pancreas[18]
-
Histopathology of a pancreatic endocrine tumor (insulinoma). Insulin immunostain. Source:https://librepathology.org/wiki/Neuroendocrine_tumour_of_the_pancreas[18]
References
- ↑ 1.0 1.1 Chaudhuri MR (1973). "Primary pulmonary cavitating carcinomas". Thorax. 28 (3): 354–66. PMC 470041. PMID 4353362.
- ↑ Mouroux J, Padovani B, Elkaïm D, Richelme H (1996). "Should cavitated bronchopulmonary cancers be considered a separate entity?". Ann. Thorac. Surg. 61 (2): 530–2. doi:10.1016/0003-4975(95)00973-6. PMID 8572761.
- ↑ Onn A, Choe DH, Herbst RS, Correa AM, Munden RF, Truong MT, Vaporciyan AA, Isobe T, Gilcrease MZ, Marom EM (2005). "Tumor cavitation in stage I non-small cell lung cancer: epidermal growth factor receptor expression and prediction of poor outcome". Radiology. 237 (1): 342–7. doi:10.1148/radiol.2371041650. PMID 16183941.
- ↑ 4.0 4.1 Langford CA, Hoffman GS (1999). "Rare diseases.3: Wegener's granulomatosis". Thorax. 54 (7): 629–37. PMC 1745525. PMID 10377211.
- ↑ Lee KS, Kim TS, Fujimoto K, Moriya H, Watanabe H, Tateishi U, Ashizawa K, Johkoh T, Kim EA, Kwon OJ (2003). "Thoracic manifestation of Wegener's granulomatosis: CT findings in 30 patients". Eur Radiol. 13 (1): 43–51. doi:10.1007/s00330-002-1422-2. PMID 12541109.
- ↑ Baughman RP, Teirstein AS, Judson MA, Rossman MD, Yeager H, Bresnitz EA, DePalo L, Hunninghake G, Iannuzzi MC, Johns CJ, McLennan G, Moller DR, Newman LS, Rabin DL, Rose C, Rybicki B, Weinberger SE, Terrin ML, Knatterud GL, Cherniak R (2001). "Clinical characteristics of patients in a case control study of sarcoidosis". Am. J. Respir. Crit. Care Med. 164 (10 Pt 1): 1885–9. doi:10.1164/ajrccm.164.10.2104046. PMID 11734441.
- ↑ Brauner MW, Grenier P, Mompoint D, Lenoir S, de Crémoux H (1989). "Pulmonary sarcoidosis: evaluation with high-resolution CT". Radiology. 172 (2): 467–71. doi:10.1148/radiology.172.2.2748828. PMID 2748828.
- ↑ Murphy J, Schnyder P, Herold C, Flower C (1998). "Bronchiolitis obliterans organising pneumonia simulating bronchial carcinoma". Eur Radiol. 8 (7): 1165–9. doi:10.1007/s003300050527. PMID 9724431.
- ↑ 9.0 9.1 Al-Ghanem S, Al-Jahdali H, Bamefleh H, Khan AN (2008). "Bronchiolitis obliterans organizing pneumonia: pathogenesis, clinical features, imaging and therapy review". Ann Thorac Med. 3 (2): 67–75. doi:10.4103/1817-1737.39641. PMC 2700454. PMID 19561910.
- ↑ Cordier JF, Loire R, Brune J (1989). "Idiopathic bronchiolitis obliterans organizing pneumonia. Definition of characteristic clinical profiles in a series of 16 patients". Chest. 96 (5): 999–1004. PMID 2805873.
- ↑ Lee KS, Kullnig P, Hartman TE, Müller NL (1994). "Cryptogenic organizing pneumonia: CT findings in 43 patients". AJR Am J Roentgenol. 162 (3): 543–6. doi:10.2214/ajr.162.3.8109493. PMID 8109493.
- ↑ Suri HS, Yi ES, Nowakowski GS, Vassallo R (2012). "Pulmonary langerhans cell histiocytosis". Orphanet J Rare Dis. 7: 16. doi:10.1186/1750-1172-7-16. PMC 3342091. PMID 22429393.
- ↑ Moore AD, Godwin JD, Müller NL, Naidich DP, Hammar SP, Buschman DL, Takasugi JE, de Carvalho CR (1989). "Pulmonary histiocytosis X: comparison of radiographic and CT findings". Radiology. 172 (1): 249–54. doi:10.1148/radiology.172.1.2787035. PMID 2787035.
- ↑ Blachar A, Federle MP, Brancatelli G (2001). "Primary biliary cirrhosis: clinical, pathologic, and helical CT findings in 53 patients". Radiology. 220 (2): 329–36. doi:10.1148/radiology.220.2.r01au36329. PMID 11477233.
- ↑ Blachar, Arye; Federle, Michael P.; Brancatelli, Giuseppe (2001). "Primary Biliary Cirrhosis: Clinical, Pathologic, and Helical CT Findings in 53 Patients". Radiology. 220 (2): 329–336. doi:10.1148/radiology.220.2.r01au36329. ISSN 0033-8419.
- ↑ "Terminology of nodular hepatocellular lesions". Hepatology. 22 (3): 983–93. 1995. PMID 7657307.
- ↑ "File:Jaundice08.jpg - Wikimedia Commons". External link in
|title=(help) - ↑ 18.0 18.1 18.2 Neuroendocrine tumor of the pancreas. Libre Pathology. http://librepathology.org/wiki/index.php/Neuroendocrine_tumour_of_the_pancreas
![Histopathology of a pancreatic endocrine tumor (insulinoma). Source:https://librepathology.org/wiki/Neuroendocrine_tumour_of_the_pancreas[18]](/images/2/2f/Pancreatic_insulinoma_histology_2.JPG)
![Histopathology of a pancreatic endocrine tumor (insulinoma). Chromogranin A immunostain. Source:https://librepathology.org/wiki/Neuroendocrine_tumour_of_the_pancreas[18]](/images/a/a3/Pancreatic_insulinoma_histopathology_3.JPG)
![Histopathology of a pancreatic endocrine tumor (insulinoma). Insulin immunostain. Source:https://librepathology.org/wiki/Neuroendocrine_tumour_of_the_pancreas[18]](/images/d/d5/Pancreatic_insulinoma_histology_4.JPG)