Glycogen storage disease type V
For the main page on glycogen storage disease, please click here
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]; Associate Editor(s)-in-Chief: Sargun Singh Walia M.B.B.S.[2], Anmol Pitliya, M.B.B.S. M.D.[3]
Overview
Glycogen storage disease type V also known as McArdle's disease is caused by the deficiency of myophosphorylase. In 1951, Brian McArdle first described glycogen storage disease type V. It is an autosomal recessive disorder caused by mutation of the gene PYGM which is located on chromosome 11q13. Myophosphorylase is the isozyme of phosphorylase specific to muscles. Its deficiency leads to blockage of carbohydrate metabolic pathways causing accumulation of excess glycogen in the muscles. Muscles are unable to produce energy from glycogen and may lead to rhabdomyolysis. Glycogen storage disease type V commonly affects individuals in the 2nd-3rd decade of their life. There is insufficient evidence to recommend routine screening for glycogen storage disease type V but carrier screening of at-risk relatives may be done. Different individuals present uniquely depending on the severity of enzyme deficiency. Some adults can develop progressive proximal muscle weakness or fixed motor weakness. A "Second-wind phenomenon" can be seen in patients suffering from glycogen storage disease type V. Burgundy-colored urine can be seen after an intense session of exercise due to rhabdomyolysis. On physical examination, persistent weakness and muscle wasting may be seen as the patient ages. Laboratory findings may show elevated levels of creatine kinase levels in almost 90% of patients suffering from glycogen storage disease type V. Forearm exercise test and electromyography are used to diagnose GSD type V. Treatment includes dietary modification and mild to moderate aerobic exercises like walking or bicycling.
Historical Perspective
- Glycogen storage disease type V was first described by Brian McArdle, a Scottish physician, in 1951.[1]
- In 1993, the first PYGM pathogenic mutations were described.[2]
Classification
There is no established system for the classification of glycogen storage disease type V.
Pathophysiology
Pathogenesis
- Glycogen storage disease type V is caused by the deficiency of liver myophosphorylase.
- Normal function of phophorylase is:[3]
- Maintains the glucose homeostasis.
- Leads to the degradation of glycogen into glucose-1-phosphate.
- Alpha-1,4-glucoside residues are removed from the outer branches of the glycogen molecule.
- Degradation is done until glycogen is broken down to branches of approximately 4 glucosyl units.
- 3 Isozymes of phosphorylase are found in the muscles, liver, and brain.
- Myophosphorylase is the isozyme specific to muscles.
- Carbohydrate metabolic pathways are blocked which lead to accumulation of excess glycogen in muscles.
- Muscles are unable to produce energy from glycogen due to deficiency of this enzyme.
- There is an increase in the cellular mechanical stress due to:
- Accumulation of glycogen.
- Downregulation of sodium-potassium pumps.
- Elevation of sarcoplasmic calcium.
- This processes may lead to rhabdomyolysis.[4]
- Muscle damage is done due to oxidative stress and purine nucleotide metabolism.
- Hypoglycemia is not seen as liver phosphorylase is not involved.
Metabolic Pathway
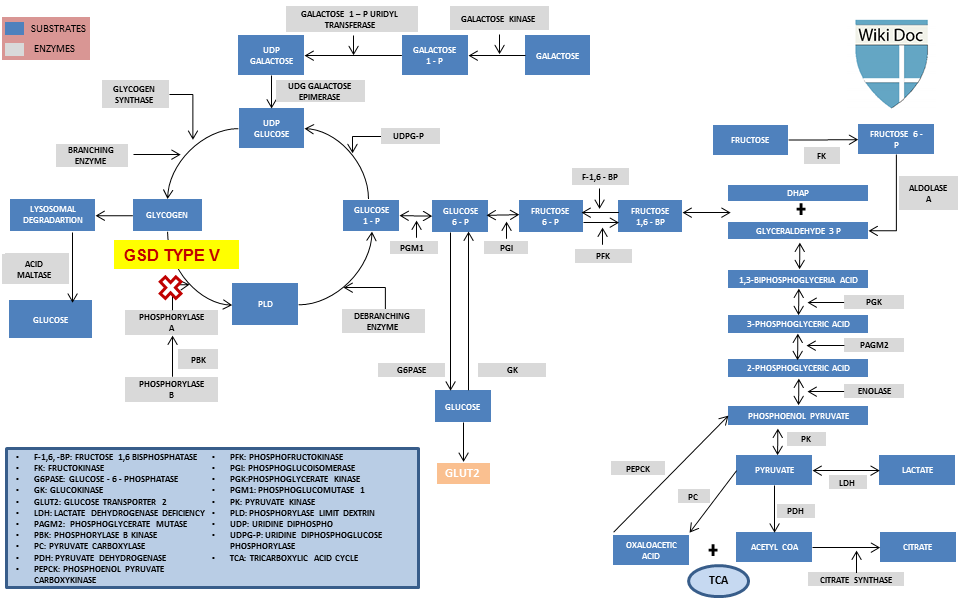
Causes
- Glycogen storage disease type V is an autosomal recessive disorder.
- Glycogen storage disease type V is caused by the deficiency of the myophosphorylase enzyme. PYGM gene mutation responsible for the myophosphorylase enzyme deficiency is located on chromosome 11q13.[5]
- Disease severity can be correlated with the genotype at the angiotensin-converting enzyme (ACE) locus.
Differentiating Glycogen storage disease type V from Other Diseases
| Differentiating Glycogen Storage Diseases | ||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Glycogen storage disease | Enzyme deficiency | Genetics | History and symptoms | Physical examination | Laboratory findings | Imaging | Other features | |||||||
| Gene mutation | Inheritance | Chromosome | Hypoglycemia | Muscle weakness | Hypotonia | Hepatomegaly | Elevated CK | Cardiomegaly | ||||||
| Glycogen storage disease type I[6][7][8][9][10][11] | Von Gierke's disease | GSD type Ia | Glucose-6-phosphatase | G6PC gene mutation | Autosomal recessive | 17q21 | + | + | + | + | - | - | ||
| GSD type Ib | Microsomal glucose-6-phosphate transporter | SLC37A4 gene mutation | Autosomal recessive | 11q23 | ||||||||||
| Glycogen storage disease type II[12][13][14][15][16][17][18][19][20] | Pompe disease | Infantile onset | Acid alpha-glucosidase | GAA gene | Autosomal recessive | 17q25 | - | + | + | + | + | + | ||
| Late onset | Autosomal recessive | - | + | + | + | + | +/- | |||||||
| Glycogen storage disease type III[21][22][23][24][25][26] | Cori disease | GSD type IIIa | Debranching enzyme (deficiency in muscle and liver) | AGL gene mutation | Autosomal recessive | 1p21 | + | + | + | + | + | + |
| |
| GSD type IIIb | Debranching enzyme (deficiency in liver only) | Autosomal recessive | ||||||||||||
| Glycogen storage disease type IV[27][28][29][30][31] | Andersen's disease | Branching enzyme | GBE1 gene mutation | Autosomal recessive | 3p12 | +/- | + | + | + | + | + | - | ||
| Glycogen storage disease type V[1][32][33][34][35][36][37] | McArdle disease | Muscle glycogen phosphorylase | PYGM gene mutation | Autosomal recessive | 11q13 | - | + | - | - | + | - |
| ||
| Glycogen storage disease type VI[38][39][40][41][42] | Hers' disease | Autosomal | Liver glycogen phosphorylase | PYGL gene mutation | Autosomal recessive | 14q22 | +/- | + | +/- | + | - | - |
| |
| X-linked | PYGL gene mutation | X-linked recessive | X | |||||||||||
| Glycogen storage disease type VII[43][44][45][46][47][48] | Tarui's disease | Muscle phosphofructokinase | PFKM gene mutation | Autosomal recessive | 12q13 | + | + | - | - | + | + | |||
| Glycogen storage disease type IX[49][39][50] | GSD type IXa[51][52][53][54][55] | Phosphorylase b kinase (deficiency in liver only) | PHKA2 gene mutation | X-linked recessive | Xp22 | + | - | - | + | - | - |
| ||
| GSD type IXb[56][57][58] | Phosphorylase b kinase (deficiency in liver and muscle) | PHKB gene mutation | Autosomal recessive | 16q12 | + | - | - | + | - | - |
| |||
| Glycogen storage disease type X[59][60][61][62] | Phosphoglycerate mutase | PGAM2 gene mutation | Autosomal recessive | 7p13 | - | - | - | - | + | - |
| |||
| Glycogen storage disease type XI[63][64][65][66] | Lactate dehydrogenase A deficiency | Lactate dehydrogenase A | LDHA gene mutation | Autosomal recessive | 11p15 | - | - | - | - | + | - |
| ||
| Glycogen storage disease type XII[67][68][69][70] | Aldolase A deficiency | Aldolase A | ALDOA gene mutation | Autosomal recessive | 16p11 | - | + | - | + | - | - | |||
| Glycogen storage disease type XIII[71] | Beta-enolase | ENO3 gene mutation | Autosomal recessive | 17p13 | - | + | - | - | + | - | - | |||
| Glycogen storage disease type XIV[72][73] | Phosphoglucomutase type 2 | PGM1 gene mutation | Autosomal recessive | 1p31 | +/- | + | - | - | + | - |
| |||
| Glycogen storage disease type 0[74][75][76][77] | Lewis' disease | Hepatic glycogen synthase | GYS2 gene mutation (liver) | Autosomal recessive | 12p12 | + | - | - | - | - | - |
| ||
Epidemiology and Demographics
- The prevalence of glycogen storage disease type V is approximately 0.6 in 100,000 individuals in spanish community.[78]
- The prevalence of glycogen storage disease type V is approximately 1 in 100,000 individuals in Texas, USA.[79]
- Glycogen storage disease type V commonly affects individuals in the 2nd-3rd decade of life.
Risk Factors
- The most potent risk factor in the development of glycogen storage disease type V is a sibling with glycogen storage disease type V.
Screening
- There is insufficient evidence to recommend routine screening for glycogen storage disease type V.
Natural History, Complications, and Prognosis
Complications
- Glycogen storage disease type V may have potential anesthetic and perioperative risks.[80]
- Statin should be used with caution as they can lead to myopathy in individuals suffering from glycogen storage disease type V.
Prognosis
- Glycogen storage disease type V is a chronic disorder.
Diagnosis
Diagnostic Study of Choice
Forearm exercise test
- Forearm exercise test is of 2 types:
- Normal chemical reactions and products of muscle activity are analysed.
- One-second handgrips are performed every other second for one minute (approximately 30 contractions)
- Blood samples are taken to check for lactate, ammonia, and creatine kinase before starting the test and at 1, 2, 4, 6, 10, 20, and 30 minutes after the end of the exercise.
- A flat venous lactate curve with normal increase in ammonia is diagnostic of myophosphorylase deficiency.
- Earlier blood pressure cuff was used instead of handgrips, but it was stopped because it lead to more muscle pain and acute compartment syndrome.
Electromyography
- Electromyography of resting muscle are normal.
- No electrical activity during contracture is diagnostic of myophosphorylase deficiency.
History and Symptoms
- Different individuals present uniquely depending on the severity on enzyme deficiency.
- Some adults can develop:
- Progressive proximal muscle weakness.
- Fixed motor weakness.
- Second-wind phenomenon:[83]
- When a patient nearing fatigue slows down the intensity of exercise to a certain level, it can be increased again without recurrence of symptoms.[84]
- This phenomenon may be due to recruitment of: [85]
- Increased number of motor units
- Increased cardiac output
- Use of free fatty acids for muscle metabolism
- Burgundy-colored urine can be seen after an intense session of exercise.
- This is believed to be due to rhabdomyolysis.
Physical Examination
- Normal muscle strength and reflexes may be seen.
- Persistent weakness and muscle wasting may be seen as the patient ages.
Laboratory Findings
- Creatine kinase levels are checked in all cases with suspicion of glycogen storage disease.
- 90% patients suffering from glycogen storage disease type V have elevated levels.
- Fasting glucose testing is done to rule out hypoglycemia.
- Myoglobinuria
- Urine analysis is done to check for it.
- It is seen in 50% cases after exercise.
- Biochemical assay
- Phosphorylase reaction is absent.
Other Diagnostic Studies
Muscle Biopsy
- Muscle biopsy reveals:[86]
- Fiber size variability
- Positive subsarcolemmal blebs with periodic acid-Schiff stain
- Intermyofibril vacuoles
- Selective atrophy of type 1 muscle fibers may be seen.
Treatment
Medical Therapy
- Dietary modification
- Higher-carbohydrate diet
- High-protein intake
- Simple carbohydrates (eg, sucrose) consumption before exercise can help to improve exercise tolerance.
- Dietary supplementation with creatine can help to some extent.[87]
- Exercise
Surgery
- Surgical intervention is not recommended for the management of glycogen storage disease type V.
Primary Prevention
- Effective measures for primary prevention of glycogen storage disease type 5 include genetic counseling, prenatal diagnosis, and screening.
External links
References
- ↑ 1.0 1.1 McARDLE B (1951). "Myopathy due to a defect in muscle glycogen breakdown". Clin Sci. 10 (1): 13–35. PMID 24540673.
- ↑ Bartram C, Edwards RH, Clague J, Beynon RJ (1993). "McArdle's disease: a nonsense mutation in exon 1 of the muscle glycogen phosphorylase gene explains some but not all cases". Hum. Mol. Genet. 2 (8): 1291–3. PMID 8401511.
- ↑ Nogales-Gadea G, Brull A, Santalla A, Andreu AL, Arenas J, Martín MA, Lucia A, de Luna N, Pinós T (2015). "McArdle Disease: Update of Reported Mutations and Polymorphisms in the PYGM Gene". Hum. Mutat. 36 (7): 669–78. doi:10.1002/humu.22806. PMID 25914343.
- ↑ Nogales-Gadea G, Santalla A, Brull A, de Luna N, Lucia A, Pinós T (2015). "The pathogenomics of McArdle disease--genes, enzymes, models, and therapeutic implications". J. Inherit. Metab. Dis. 38 (2): 221–30. doi:10.1007/s10545-014-9743-2. PMID 25053163.
- ↑ Nogales-Gadea G, Brull A, Santalla A, Andreu AL, Arenas J, Martín MA, Lucia A, de Luna N, Pinós T (2015). "McArdle Disease: Update of Reported Mutations and Polymorphisms in the PYGM Gene". Hum. Mutat. 36 (7): 669–78. doi:10.1002/humu.22806. PMID 25914343.
- ↑ Mansfield BC (1999). "Molecular Genetics of Type 1 Glycogen Storage Diseases". Trends Endocrinol Metab. 10 (3): 104–113. PMID 10322403.
- ↑ Ozen H (2007). "Glycogen storage diseases: new perspectives". World J Gastroenterol. 13 (18): 2541–53. PMC 4146814. PMID 17552001.
- ↑ Froissart R, Piraud M, Boudjemline AM, Vianey-Saban C, Petit F, Hubert-Buron A; et al. (2011). "Glucose-6-phosphatase deficiency". Orphanet J Rare Dis. 6: 27. doi:10.1186/1750-1172-6-27. PMC 3118311. PMID 21599942.
- ↑ Kishnani, Priya S.; Austin, Stephanie L.; Abdenur, Jose E.; Arn, Pamela; Bali, Deeksha S.; Boney, Anne; Chung, Wendy K.; Dagli, Aditi I.; Dale, David; Koeberl, Dwight; Somers, Michael J.; Burns Wechsler, Stephanie; Weinstein, David A.; Wolfsdorf, Joseph I.; Watson, Michael S. (2014). "Diagnosis and management of glycogen storage disease type I: a practice guideline of the American College of Medical Genetics and Genomics". Genetics in Medicine. doi:10.1038/gim.2014.128. ISSN 1098-3600.
- ↑ Rake JP, Visser G, Labrune P, Leonard JV, Ullrich K, Smit GP (2002). "Glycogen storage disease type I: diagnosis, management, clinical course and outcome. Results of the European Study on Glycogen Storage Disease Type I (ESGSD I)". Eur. J. Pediatr. 161 Suppl 1: S20–34. doi:10.1007/s00431-002-0999-4. PMID 12373567.
- ↑ Bali DS, Chen YT, Austin S, et al. Glycogen Storage Disease Type I. 2006 Apr 19 [Updated 2016 Aug 25]. In: Adam MP, Ardinger HH, Pagon RA, et al., editors. GeneReviews® [Internet]. Seattle (WA): University of Washington, Seattle; 1993-2017. Available from: https://www.ncbi.nlm.nih.gov/books/NBK1312/
- ↑ Leslie N, Bailey L. Pompe Disease. 2007 Aug 31 [Updated 2017 May 11]. In: Adam MP, Ardinger HH, Pagon RA, et al., editors. GeneReviews® [Internet]. Seattle (WA): University of Washington, Seattle; 1993-2018. Available from: https://www.ncbi.nlm.nih.gov/books/NBK1261/
- ↑ Di Rocco M, Buzzi D, Tarò M (2007). "Glycogen storage disease type II: clinical overview". Acta Myol. 26 (1): 42–4. PMC 2949314. PMID 17915568.
- ↑ Kishnani PS, Hwu WL, Mandel H, Nicolino M, Yong F, Corzo D; et al. (2006). "A retrospective, multinational, multicenter study on the natural history of infantile-onset Pompe disease". J Pediatr. 148 (5): 671–676. doi:10.1016/j.jpeds.2005.11.033. PMID 16737883.
- ↑ van den Hout HM, Hop W, van Diggelen OP, Smeitink JA, Smit GP, Poll-The BT; et al. (2003). "The natural course of infantile Pompe's disease: 20 original cases compared with 133 cases from the literature". Pediatrics. 112 (2): 332–40. PMID 12897283.
- ↑ Slonim AE, Bulone L, Ritz S, Goldberg T, Chen A, Martiniuk F (2000). "Identification of two subtypes of infantile acid maltase deficiency". J Pediatr. 137 (2): 283–5. doi:10.1067/mpd.2000.107112. PMID 10931430.
- ↑ Martiniuk F, Mehler M, Tzall S, Meredith G, Hirschhorn R (1990). "Sequence of the cDNA and 5'-flanking region for human acid alpha-glucosidase, detection of an intron in the 5' untranslated leader sequence, definition of 18-bp polymorphisms, and differences with previous cDNA and amino acid sequences". DNA Cell Biol. 9 (2): 85–94. doi:10.1089/dna.1990.9.85. PMID 2111708.
- ↑ Hoefsloot LH, Hoogeveen-Westerveld M, Kroos MA, van Beeumen J, Reuser AJ, Oostra BA (1988). "Primary structure and processing of lysosomal alpha-glucosidase; homology with the intestinal sucrase-isomaltase complex". EMBO J. 7 (6): 1697–704. PMC 457155. PMID 3049072.
- ↑ Hoefsloot LH, Hoogeveen-Westerveld M, Reuser AJ, Oostra BA (1990). "Characterization of the human lysosomal alpha-glucosidase gene". Biochem J. 272 (2): 493–7. PMC 1149727. PMID 2268276.
- ↑ Kuo WL, Hirschhorn R, Huie ML, Hirschhorn K (1996). "Localization and ordering of acid alpha-glucosidase (GAA) and thymidine kinase (TK1) by fluorescence in situ hybridization". Hum Genet. 97 (3): 404–6. PMID 8786092.
- ↑ Shen J, Bao Y, Liu HM, Lee P, Leonard JV, Chen YT (1996). "Mutations in exon 3 of the glycogen debranching enzyme gene are associated with glycogen storage disease type III that is differentially expressed in liver and muscle". J Clin Invest. 98 (2): 352–7. doi:10.1172/JCI118799. PMC 507437. PMID 8755644.
- ↑ Ding JH, de Barsy T, Brown BI, Coleman RA, Chen YT (1990). "Immunoblot analyses of glycogen debranching enzyme in different subtypes of glycogen storage disease type III". J Pediatr. 116 (1): 95–100. PMID 2295969.
- ↑ Aoyama Y, Ozer I, Demirkol M, Ebara T, Murase T, Podskarbi T; et al. (2009). "Molecular features of 23 patients with glycogen storage disease type III in Turkey: a novel mutation p.R1147G associated with isolated glucosidase deficiency, along with 9 AGL mutations". J Hum Genet. 54 (11): 681–6. doi:10.1038/jhg.2009.100. PMID 19834502.
- ↑ Kishnani, Priya S; Austin, Stephanie L; Arn, Pamela; Bali, Deeksha S; Boney, Anne; Case, Laura E; Chung, Wendy K; Desai, Dev M; El-Gharbawy, Areeg; Haller, Ronald; Smit, G Peter A; Smith, Alastair D; Hobson-Webb, Lisa D; Wechsler, Stephanie Burns; Weinstein, David A; Watson, Michael S (2010). "Glycogen Storage Disease Type III diagnosis and management guidelines". Genetics in Medicine. 12 (7): 446–463. doi:10.1097/GIM.0b013e3181e655b6. ISSN 1098-3600.
- ↑ Dagli A, Sentner CP, Weinstein DA. Glycogen Storage Disease Type III. 2010 Mar 9 [Updated 2016 Dec 29]. In: Adam MP, Ardinger HH, Pagon RA, et al., editors. GeneReviews® [Internet]. Seattle (WA): University of Washington, Seattle; 1993-2017. Available from: https://www.ncbi.nlm.nih.gov/books/NBK26372/
- ↑ Wolfsdorf JI, Weinstein DA (2003). "Glycogen storage diseases". Rev Endocr Metab Disord. 4 (1): 95–102. PMID 12618563.
- ↑ Bruno C, van Diggelen OP, Cassandrini D, Gimpelev M, Giuffrè B, Donati MA; et al. (2004). "Clinical and genetic heterogeneity of branching enzyme deficiency (glycogenosis type IV)". Neurology. 63 (6): 1053–8. PMID 15452297.
- ↑ Bruno C, Cassandrini D, Assereto S, Akman HO, Minetti C, Di Mauro S (2007). "Neuromuscular forms of glycogen branching enzyme deficiency". Acta Myol. 26 (1): 75–8. PMC 2949312. PMID 17915577.
- ↑ Brown BI, Brown DH (1966). "Lack of an alpha-1,4-glucan: alpha-1,4-glucan 6-glycosyl transferase in a case of type IV glycogenosis". Proc Natl Acad Sci U S A. 56 (2): 725–9. PMC 224432. PMID 5229990.
- ↑ McConkie-Rosell A, Wilson C, Piccoli DA, Boyle J, DeClue T, Kishnani P; et al. (1996). "Clinical and laboratory findings in four patients with the non-progressive hepatic form of type IV glycogen storage disease". J Inherit Metab Dis. 19 (1): 51–8. PMID 8830177.
- ↑ Magoulas PL, El-Hattab AW. Glycogen Storage Disease Type IV. 2013 Jan 3. In: Adam MP, Ardinger HH, Pagon RA, et al., editors. GeneReviews® [Internet]. Seattle (WA): University of Washington, Seattle; 1993-2018. Available from: https://www.ncbi.nlm.nih.gov/books/NBK115333/
- ↑ SCHMID R, MAHLER R (1959). "Chronic progressive myopathy with myoglobinuria: demonstration of a glycogenolytic defect in the muscle". J Clin Invest. 38: 2044–58. doi:10.1172/JCI103983. PMC 441792. PMID 14442994.
- ↑ Mommaerts WF, Illingworth B, Pearson CM, Guillory RJ, Seraydarian K (1959). "A FUNCTIONAL DISORDER OF MUSCLE ASSOCIATED WITH THE ABSENCE OF PHOSPHORYLASE". Proc Natl Acad Sci U S A. 45 (6): 791–7. PMC 222638. PMID 16590445.
- ↑ PEARSON CM, RIMER DG, MOMMAERTS WF (1961). "A metabolic myopathy due to absence of muscle phosphorylase". Am J Med. 30: 502–17. PMID 13733779.
- ↑ Grünfeld JP, Ganeval D, Chanard J, Fardeau M, Dreyfus JC (1972). "Acute renal failure in McArdle's disease. Report of two cases". N Engl J Med. 286 (23): 1237–41. doi:10.1056/NEJM197206082862304. PMID 4502558.
- ↑ Schmidt B, Servidei S, Gabbai AA, Silva AC, de Sousa Bulle de Oliveira A, DiMauro S (1987). "McArdle's disease in two generations: autosomal recessive transmission with manifesting heterozygote". Neurology. 37 (9): 1558–61. PMID 3476861.
- ↑ Martín MA, Lucía A, Arenas J, et al. Glycogen Storage Disease Type V. 2006 Apr 19 [Updated 2014 Jun 26]. In: Adam MP, Ardinger HH, Pagon RA, et al., editors. GeneReviews® [Internet]. Seattle (WA): University of Washington, Seattle; 1993-2018. Available from: https://www.ncbi.nlm.nih.gov/books/NBK1344/
- ↑ Wallis PG, Sidbury JB, Harris RC (1966). "Hepatic phosphorylase defect. Studies on peripheral blood". Am J Dis Child. 111 (3): 278–82. PMID 5904467.
- ↑ 39.0 39.1 Roscher A, Patel J, Hewson S, Nagy L, Feigenbaum A, Kronick J; et al. (2014). "The natural history of glycogen storage disease types VI and IX: Long-term outcome from the largest metabolic center in Canada". Mol Genet Metab. 113 (3): 171–6. doi:10.1016/j.ymgme.2014.09.005. PMID 25266922.
- ↑ Burwinkel B, Bakker HD, Herschkovitz E, Moses SW, Shin YS, Kilimann MW (1998). "Mutations in the liver glycogen phosphorylase gene (PYGL) underlying glycogenosis type VI". Am J Hum Genet. 62 (4): 785–91. PMC 1377030. PMID 9529348.
- ↑ Chang S, Rosenberg MJ, Morton H, Francomano CA, Biesecker LG (1998). "Identification of a mutation in liver glycogen phosphorylase in glycogen storage disease type VI". Hum Mol Genet. 7 (5): 865–70. PMID 9536091.
- ↑ Dagli AI, Weinstein DA. Glycogen Storage Disease Type VI. 2009 Apr 23 [Updated 2011 May 17]. In: Adam MP, Ardinger HH, Pagon RA, et al., editors. GeneReviews® [Internet]. Seattle (WA): University of Washington, Seattle; 1993-2018. Available from: https://www.ncbi.nlm.nih.gov/books/NBK5941/
- ↑ Raben N, Sherman JB (1995). "Mutations in muscle phosphofructokinase gene". Hum Mutat. 6 (1): 1–6. doi:10.1002/humu.1380060102. PMID 7550225.
- ↑ TARUI S, OKUNO G, IKURA Y, TANAKA T, SUDA M, NISHIKAWA M (1965). "PHOSPHOFRUCTOKINASE DEFICIENCY IN SKELETAL MUSCLE. A NEW TYPE OF GLYCOGENOSIS". Biochem Biophys Res Commun. 19: 517–23. PMID 14339001.
- ↑ Layzer RB, Rowland LP, Ranney HM (1967). "Muscle phosphofructokinase deficiency". Arch Neurol. 17 (5): 512–23. PMID 4228297.
- ↑ Satoyoshi E, Kowa H (1967). "A myopathy due to glycolytic abnormality". Arch Neurol. 17 (3): 248–56. PMID 4228753.
- ↑ Waterbury L, Frenkel EP (1972). "Hereditary nonspherocytic hemolysis with erythrocyte phosphofructokinase deficiency". Blood. 39 (3): 415–25. PMID 4258222.
- ↑ Vora S, Corash L, Engel WK, Durham S, Seaman C, Piomelli S (1980). "The molecular mechanism of the inherited phosphofructokinase deficiency associated with hemolysis and myopathy". Blood. 55 (4): 629–35. PMID 6444532.
- ↑ Beauchamp NJ, Dalton A, Ramaswami U, Niinikoski H, Mention K, Kenny P; et al. (2007). "Glycogen storage disease type IX: High variability in clinical phenotype". Mol Genet Metab. 92 (1–2): 88–99. doi:10.1016/j.ymgme.2007.06.007. PMID 17689125.
- ↑ Goldstein J, Austin S, Kishnani P, et al. Phosphorylase Kinase Deficiency. 2011 May 31. In: Adam MP, Ardinger HH, Pagon RA, et al., editors. GeneReviews® [Internet]. Seattle (WA): University of Washington, Seattle; 1993-2018. Available from: https://www.ncbi.nlm.nih.gov/books/NBK55061/
- ↑ Keating JP, Brown BI, White NH, DiMauro S (1985). "X-linked glycogen storage disease. A cause of hypotonia, hyperuricemia, and growth retardation". Am J Dis Child. 139 (6): 609–13. PMID 3859203.
- ↑ Hendrickx J, Coucke P, Hors-Cayla MC, Smit GP, Shin YS, Deutsch J; et al. (1994). "Localization of a new type of X-linked liver glycogenosis to the chromosomal region Xp22 containing the liver alpha-subunit of phosphorylase kinase (PHKA2)". Genomics. 21 (3): 620–5. PMID 7959740.
- ↑ Schimke RN, Zakheim RM, Corder RC, Hug G (1973). "Glycogen storage disease type IX: benign glycogenosis of liver and hepatic phosphorylase kinase deficiency". J Pediatr. 83 (6): 1031–4. PMID 4518931.
- ↑ Willems PJ, Gerver WJ, Berger R, Fernandes J (1990). "The natural history of liver glycogenosis due to phosphorylase kinase deficiency: a longitudinal study of 41 patients". Eur J Pediatr. 149 (4): 268–71. PMID 2303074.
- ↑ Hendrickx J, Bosshard NU, Willems P, Gitzelmann R (1998). "Clinical, biochemical and molecular findings in a patient with X-linked liver glycogenosis followed for 40 years". Eur J Pediatr. 157 (11): 919–23. PMID 9835437.
- ↑ Bashan N, Iancu TC, Lerner A, Fraser D, Potashnik R, Moses SW (1981). "Glycogenosis due to liver and muscle phosphorylase kinase deficiency". Pediatr Res. 15 (4 Pt 1): 299–303. doi:10.1203/00006450-198104000-00002. PMID 6938920.
- ↑ Gray RG, Kumar D, Whitfield AE (1983). "Glycogen phosphorylase b kinase deficiency in three siblings". J Inherit Metab Dis. 6 (3): 107. PMID 6422139.
- ↑ Burwinkel B, Maichele AJ, Aagenaes O, Bakker HD, Lerner A, Shin YS; et al. (1997). "Autosomal glycogenosis of liver and muscle due to phosphorylase kinase deficiency is caused by mutations in the phosphorylase kinase beta subunit (PHKB)". Hum Mol Genet. 6 (7): 1109–15. PMID 9215682.
- ↑ Hadjigeorgiou GM, Kawashima N, Bruno C, Andreu AL, Sue CM, Rigden DJ; et al. (1999). "Manifesting heterozygotes in a Japanese family with a novel mutation in the muscle-specific phosphoglycerate mutase (PGAM-M) gene". Neuromuscul Disord. 9 (6–7): 399–402. PMID 10545043.
- ↑ Tsujino S, Shanske S, Sakoda S, Fenichel G, DiMauro S (1993). "The molecular genetic basis of muscle phosphoglycerate mutase (PGAM) deficiency". Am J Hum Genet. 52 (3): 472–7. PMC 1682163. PMID 8447317.
- ↑ Kissel JT, Beam W, Bresolin N, Gibbons G, DiMauro S, Mendell JR (1985). "Physiologic assessment of phosphoglycerate mutase deficiency: incremental exercise test". Neurology. 35 (6): 828–33. PMID 2987758.
- ↑ DiMauro S, Miranda AF, Khan S, Gitlin K, Friedman R (1981). "Human muscle phosphoglycerate mutase deficiency: newly discovered metabolic myopathy". Science. 212 (4500): 1277–9. PMID 6262916.
- ↑ Yoshikuni K, Tagami H, Yamada M, Sudo K, Kanno T (1986). "Erythematosquamous skin lesions in hereditary lactate dehydrogenase M-subunit deficiency". Arch Dermatol. 122 (12): 1420–4. PMID 3789777.
- ↑ Kanno T, Sudo K, Maekawa M, Nishimura Y, Ukita M, Fukutake K (1988). "Lactate dehydrogenase M-subunit deficiency: a new type of hereditary exertional myopathy". Clin Chim Acta. 173 (1): 89–98. PMID 3383424.
- ↑ Maekawa M, Sudo K, Kanno T (1986). "Immunochemical studies on lactate dehydrogenase A subunit deficiencies". Am J Hum Genet. 39 (2): 232–8. PMC 1683931. PMID 3092644.
- ↑ Takayasu S, Fujiwara S, Waki T (1991). "Hereditary lactate dehydrogenase M-subunit deficiency: lactate dehydrogenase activity in skin lesions and in hair follicles". J Am Acad Dermatol. 24 (2 Pt 2): 339–42. PMID 1999544.
- ↑ Kishi H, Mukai T, Hirono A, Fujii H, Miwa S, Hori K (1987). "Human aldolase A deficiency associated with a hemolytic anemia: thermolabile aldolase due to a single base mutation". Proc Natl Acad Sci U S A. 84 (23): 8623–7. PMC 299598. PMID 2825199.
- ↑ Beutler E, Scott S, Bishop A, Margolis N, Matsumoto F, Kuhl W (1973). "Red cell aldolase deficiency and hemolytic anemia: a new syndrome". Trans Assoc Am Physicians. 86: 154–66. PMID 4788792.
- ↑ Kreuder J, Borkhardt A, Repp R, Pekrun A, Göttsche B, Gottschalk U; et al. (1996). "Brief report: inherited metabolic myopathy and hemolysis due to a mutation in aldolase A." N Engl J Med. 334 (17): 1100–4. doi:10.1056/NEJM199604253341705. PMID 8598869.
- ↑ Hurst JA, Baraitser M, Winter RM (1987). "A syndrome of mental retardation, short stature, hemolytic anemia, delayed puberty, and abnormal facial appearance: similarities to a report of aldolase A deficiency". Am J Med Genet. 28 (4): 965–70. doi:10.1002/ajmg.1320280423. PMID 3688035.
- ↑ Comi GP, Fortunato F, Lucchiari S, Bordoni A, Prelle A, Jann S; et al. (2001). "Beta-enolase deficiency, a new metabolic myopathy of distal glycolysis". Ann Neurol. 50 (2): 202–7. PMID 11506403.
- ↑ Tegtmeyer LC, Rust S, van Scherpenzeel M, Ng BG, Losfeld ME, Timal S; et al. (2014). "Multiple phenotypes in phosphoglucomutase 1 deficiency". N Engl J Med. 370 (6): 533–42. doi:10.1056/NEJMoa1206605. PMC 4373661. PMID 24499211.
- ↑ Stojkovic T, Vissing J, Petit F, Piraud M, Orngreen MC, Andersen G; et al. (2009). "Muscle glycogenosis due to phosphoglucomutase 1 deficiency". N Engl J Med. 361 (4): 425–7. doi:10.1056/NEJMc0901158. PMID 19625727.
- ↑ Orho M, Bosshard NU, Buist NR, Gitzelmann R, Aynsley-Green A, Blümel P; et al. (1998). "Mutations in the liver glycogen synthase gene in children with hypoglycemia due to glycogen storage disease type 0". J Clin Invest. 102 (3): 507–15. doi:10.1172/JCI2890. PMC 508911. PMID 9691087.
- ↑ Laberge AM, Mitchell GA, van de Werve G, Lambert M (2003). "Long-term follow-up of a new case of liver glycogen synthase deficiency". Am J Med Genet A. 120A (1): 19–22. doi:10.1002/ajmg.a.20110. PMID 12794686.
- ↑ Gitzelmann R, Spycher MA, Feil G, Müller J, Seilnacht B, Stahl M; et al. (1996). "Liver glycogen synthase deficiency: a rarely diagnosed entity". Eur J Pediatr. 155 (7): 561–7. PMID 8831078.
- ↑ Rutledge SL, Atchison J, Bosshard NU, Steinmann B (2001). "Case report: liver glycogen synthase deficiency--a cause of ketotic hypoglycemia". Pediatrics. 108 (2): 495–7. PMID 11483824.
- ↑ Lucia A, Ruiz JR, Santalla A, Nogales-Gadea G, Rubio JC, García-Consuegra I, Cabello A, Pérez M, Teijeira S, Vieitez I, Navarro C, Arenas J, Martin MA, Andreu AL (2012). "Genotypic and phenotypic features of McArdle disease: insights from the Spanish national registry". J. Neurol. Neurosurg. Psychiatry. 83 (3): 322–8. doi:10.1136/jnnp-2011-301593. PMID 22250184.
- ↑ Haller RG (2000). "Treatment of McArdle disease". Arch. Neurol. 57 (7): 923–4. PMID 10891971.
- ↑ Bollig G (2013). "McArdle's disease (glycogen storage disease type V) and anesthesia--a case report and review of the literature". Paediatr Anaesth. 23 (9): 817–23. doi:10.1111/pan.12164. PMID 23565573.
- ↑ Kazemi-Esfarjani P, Skomorowska E, Jensen TD, Haller RG, Vissing J (2002). "A nonischemic forearm exercise test for McArdle disease". Ann. Neurol. 52 (2): 153–9. doi:10.1002/ana.10263. PMID 12210784.
- ↑ Andersen ST, Dunø M, Schwartz M, Vissing J (2006). "Do carriers of PYGM mutations have symptoms of McArdle disease?". Neurology. 67 (4): 716–8. doi:10.1212/01.wnl.0000230154.79933.d7. PMID 16924035.
- ↑ Ørngreen MC, Jeppesen TD, Andersen ST, Taivassalo T, Hauerslev S, Preisler N, Haller RG, van Hall G, Vissing J (2009). "Fat metabolism during exercise in patients with McArdle disease". Neurology. 72 (8): 718–24. doi:10.1212/01.wnl.0000343002.74480.e4. PMID 19237700.
- ↑ Kitaoka Y (2014). "McArdle Disease and Exercise Physiology". Biology (Basel). 3 (1): 157–66. doi:10.3390/biology3010157. PMC 4009758. PMID 24833339.
- ↑ Braakhekke JP, de Bruin MI, Stegeman DF, Wevers RA, Binkhorst RA, Joosten EM (1986). "The second wind phenomenon in McArdle's disease". Brain. 109 ( Pt 6): 1087–101. PMID 3466659.
- ↑ Felice KJ, Grunnet ML, Sima AA (1996). "Selective atrophy of type 1 muscle fibers in McArdle's disease". Neurology. 47 (2): 581–3. PMID 8757045.
- ↑ Vorgerd M, Grehl T, Jager M, Muller K, Freitag G, Patzold T, Bruns N, Fabian K, Tegenthoff M, Mortier W, Luttmann A, Zange J, Malin JP (2000). "Creatine therapy in myophosphorylase deficiency (McArdle disease): a placebo-controlled crossover trial". Arch. Neurol. 57 (7): 956–63. PMID 10891977.
- ↑ Haller RG, Wyrick P, Taivassalo T, Vissing J (2006). "Aerobic conditioning: an effective therapy in McArdle's disease". Ann. Neurol. 59 (6): 922–8. doi:10.1002/ana.20881. PMID 16718692.
- ↑ Quinlivan R, Vissing J, Hilton-Jones D, Buckley J (2011). "Physical training for McArdle disease". Cochrane Database Syst Rev (12): CD007931. doi:10.1002/14651858.CD007931.pub2. PMID 22161416.
Template:Endocrine, nutritional and metabolic pathology