Clemastine
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]; Associate Editor(s)-in-Chief: Rabin Bista, M.B.B.S. [2]
WikiDoc MAKES NO GUARANTEE OF VALIDITY. WikiDoc is not a professional health care provider, nor is it a suitable replacement for a licensed healthcare provider. WikiDoc is intended to be an educational tool, not a tool for any form of healthcare delivery. The educational content on WikiDoc drug pages is based upon the FDA package insert, National Library of Medicine content and practice guidelines / consensus statements. WikiDoc does not promote the administration of any medication or device that is not consistent with its labeling. Please read our full disclaimer here.
Overview
Clemastine is a antihistamine that is FDA approved for the treatment of allergic rhinitis, urticaria and angioedema. Common adverse reactions include drowsiness, urinary retention.
Adult Indications and Dosage
FDA-Labeled Indications and Dosage (Adult)
Indications
- Clemastine fumarate tablets are indicated for the relief of symptoms associated with allergic rhinitis such as sneezing, rhinorrhea, pruritus, and lacrimation.
- Clemastine fumarate tablets are also indicated for the relief of mild, uncomplicated allergic skin manifestations of urticaria and angioedema.
- It should be noted that clemastine is indicated for the dermatologic indications at the 2.68 mg dosage level only.
Dosage
- DOSAGE SHOULD BE INDIVIDUALIZED ACCORDING TO THE NEEDS AND RESPONSE OF THE PATIENT.
- The recommended starting dose is 1.34 mg (1/2 tablet) twice daily. Dosage may be increased as required. Clemastine fumarate tablets are recommended for the dermatologic indications at the 2.68 mg dosage level only.
- The maximum recommended dosage is 2.68 mg three times daily. Many patients respond favorably to a single dose which may be repeated as required, but not to exceed three tablets daily.
Off-Label Use and Dosage (Adult)
Guideline-Supported Use
There is limited information regarding Off-Label Guideline-Supported Use of Clemastine in adult patients.
Non–Guideline-Supported Use
There is limited information regarding Off-Label Non–Guideline-Supported Use of Clemastine in adult patients.
Pediatric Indications and Dosage
FDA-Labeled Indications and Dosage (Pediatric)
- Safety and efficacy of clemastine fumarate have not been established in children under the age of 12 years.
Off-Label Use and Dosage (Pediatric)
Guideline-Supported Use
There is limited information regarding Off-Label Guideline-Supported Use of Clemastine in pediatric patients.
Non–Guideline-Supported Use
There is limited information regarding Off-Label Non–Guideline-Supported Use of Clemastine in pediatric patients.
Contraindications
Use in Nursing Mothers
- Because of the higher risk of antihistamines for infants generally and for newborns and prematures in particular, antihistamine therapy is contraindicated in nursing mothers.
Use in Lower Respiratory Disease
- Antihistamines should not be used to treat lower respiratory tract symptoms including asthma.
- Antihistamines are also contraindicated in the following conditions:
- Hypersensitivity to clemastine fumarate or other antihistamines of similar chemical structure.
- Monoamine oxidase inhibitor therapy
Warnings
- Antihistamines should be used with considerable caution in patients with: narrow angle glaucoma, stenosing peptic ulcer, pyloroduodenal obstruction, symptomatic prostatic hypertrophy, and bladder neck obstruction.
Use with CNS Depressants
- Clemastine has additive effects with alcohol and other CNS depressants (hypnotics, sedatives, tranquilizers, etc.)
Use in Activities Requiring Mental Alertness
- Patients should be warned about engaging in activities requiring mental alertness such as driving a car or operating appliances, machinery, etc.
Precautions
General
- Clemastine fumarate should be used with caution in patients with: history of bronchial asthma, increased intraocular pressure, hyperthyroidism, cardiovascular disease, and hypertension.
Adverse Reactions
Clinical Trials Experience
- Transient drowsiness, the most common adverse reaction associated with clemastine fumarate, occurs relatively frequently and may require discontinuation of therapy in some instances.
Antihistaminic Compounds
- It should be noted that the following reactions have occurred with one or more antihistamines and, therefore, should be kept in mind when prescribing drugs belonging to this class, including clemastine fumarate.
- The most frequent adverse reactions are underlined.
- General: Urticaria, drug rash, anaphylactic shock, photosensitivity, excessive perspiration, chills, dryness of mouth, nose, and throat.
- Cardiovascular System: Hypotension, headache, palpitations, tachycardia, extrasystoles.
- Hematologic System: Hemolytic anemia, thrombocytopenia, agranulocytosis.
- Nervous System: Sedation, sleepiness, dizziness, disturbed coordination, fatigue, confusion, restlessness, excitation, nervousness, tremor, irritability, insomnia, euphoria, paresthesias, blurred vision, diplopia, vertigo, tinnitus, acute labyrinthitis, hysteria, neuritis, convulsions.
- GI System: Epigastric distress, anorexia, nausea, vomiting, diarrhea, constipation.
- GU System: Urinary frequency, difficult urination, urinary retention, early menses.
- Respiratory System: Thickening of bronchial secretions, tightness of chest and wheezing, nasal stuffiness.
Postmarketing Experience
There is limited information regarding Postmarketing Experience of Clemastine in the drug label.
<--Drug Interactions-->
Drug Interactions
- MAO inhibitors prolong and intensify the anticholinergic (drying) effects of antihistamines.
Use in Specific Populations
Pregnancy
- Experience with this drug in pregnant women is inadequate to determine whether there exists a potential for harm to the developing fetus.
- Australian Drug Evaluation Committee (ADEC) Pregnancy Category
There is no Australian Drug Evaluation Committee (ADEC) guidance on usage of Clemastine in women who are pregnant.
Labor and Delivery
There is no FDA guidance on use of Clemastine during labor and delivery.
Nursing Mothers
- Because of the higher risk of antihistamines for infants generally and for newborns and prematures in particular, antihistamine therapy is contraindicated in nursing mothers.
Pediatric Use
- Safety and efficacy of clemastine fumarate have not been established in children under the age of 12 years.
Geriatic Use
- Antihistamines are more likely to cause dizziness, sedation, and hypotension in elderly patients.
Gender
There is no FDA guidance on the use of Clemastine with respect to specific gender populations.
Race
There is no FDA guidance on the use of Clemastine with respect to specific racial populations.
Renal Impairment
There is no FDA guidance on the use of Clemastine in patients with renal impairment.
Hepatic Impairment
There is no FDA guidance on the use of Clemastine in patients with hepatic impairment.
Females of Reproductive Potential and Males
There is no FDA guidance on the use of Clemastine in women of reproductive potentials and males.
Immunocompromised Patients
There is no FDA guidance one the use of Clemastine in patients who are immunocompromised.
Administration and Monitoring
Administration
- Oral
Monitoring
There is limited information regarding Monitoring of Clemastine in the drug label.
IV Compatibility
There is limited information regarding IV Compatibility of Clemastine in the drug label.
Overdosage
- Antihistamine overdosage reactions may vary from central nervous system depression to stimulation. Stimulation is particularly likely in children. Atropine-like signs and symptoms: dry mouth; fixed, dilated pupils; flushing; and gastrointestinal symptoms may also occur.
- If vomiting has not occured spontaneously the conscious patient should be induced to vomit. This is best done by having him drink a glass of water or milk after which he should be made to gag. Precautions against aspiration must be taken, especially in infants and children.
- If vomiting is unsuccessful gastric lavage is indicated within 3 hours after ingestion and even later if large amounts of milk or cream were given beforehand. Isotonic and 1/2 isotonic saline is the lavage solution of choice.
- Saline cathartics, such as milk of magnesia, by osmosis draw water into the bowel and therefore, are valuable for their action in rapid dilution of bowel content.
- Stimulants should not be used.
- Vasopressors may be used to treat hypotension.
Pharmacology
| |
| |
Clemastine
| |
| Systematic (IUPAC) name | |
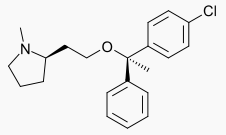
| (2R)-2-{2-[(1R)-1-(4-chlorophenyl)-1-phenylethoxy]ethyl}-1-methylpyrrolidine | |
| Identifiers | |
| CAS number | |
| ATC code | D04 R06AA04 (WHO) |
| PubChem | |
| DrugBank | |
| Chemical data | |
| Formula | Template:OrganicBox atomTemplate:OrganicBox atomTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBox atomTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBox atomTemplate:OrganicBoxTemplate:OrganicBox atomTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBox |
| Mol. mass | 343.9 |
| SMILES | & |
| Pharmacokinetic data | |
| Bioavailability | 39.2% |
| Metabolism | Hepatic |
| Half life | 21.3 Hours |
| Excretion | Renal |
| Therapeutic considerations | |
| Pregnancy cat. |
B (USA) |
| Legal status |
Unscheduled; |
| Routes | Oral |
Mechanism of Action
- Clemastine fumarate is an antihistamine with anticholinergic (drying) and sedative side effects. Antihistamines appear to compete with histamine for cell receptor sites on effector cells.
Structure
- Clemastine fumarate belongs to the benzhydryl ether group of antihistaminic compounds. The chemical name is (+)-2-[-2- [(p-chloro-α-methyl-α-phenylbenzyl) oxy] ethyl]-1-methylpyrrolidine hydrogen fumarate. It has the following structural formula:
Pharmacodynamics
There is limited information regarding Pharmacodynamics of Clemastine in the drug label.
Pharmacokinetics
- The inherently long duration of antihistaminic effects of clemastine has been demonstrated in wheal and flare studies. In normal human subjects who received histamine injections over a 24-hour period, the antihistaminic activity of clemastine reached a peak at 5 to 7 hours, persisted for 10 to 12 hours and, in some cases, for as long as 24 hours. Pharmacokinetic studies in man utilizing 3H and 14C labeled compound demonstrates that: clemastine is rapidly and nearly completely absorbed from the gastrointestinal tract, peak plasma concentrations are attained in 2 to 4 hours, and urinary excretion is the major mode of elimination.
Nonclinical Toxicology
There is limited information regarding Nonclinical Toxicology of Clemastine in the drug label.
Clinical Studies
There is limited information regarding Clinical Studies of Clemastine in the drug label.
How Supplied
- Clemastine fumarate tablets, USP are available as:
2.68 mg: Round, white tablets, scored, debossed GG 160 on one side and plain on the reverse side and supplied as:
NDC 0781-1359-01 bottles of 100
NDC 0781-1359-05 bottles of 500
NDC 0781-1359-10 bottles of 1000
Storage
- Store below 25°C. Dispense in a tight, light-resistant container.
Images
Drug Images
{{#ask: Page Name::Clemastine |?Pill Name |?Drug Name |?Pill Ingred |?Pill Imprint |?Pill Dosage |?Pill Color |?Pill Shape |?Pill Size (mm) |?Pill Scoring |?NDC |?Drug Author |format=template |template=DrugPageImages |mainlabel=- |sort=Pill Name }}
Package and Label Display Panel
2.68 MG LABEL
Clemastine
Fumarate
Tablets, USP
2.68 mg
Rx only
100 Tablets
SANDOZ
Ingredients and Appearance
{{#ask: Label Page::Clemastine |?Label Name |format=template |template=DrugLabelImages |mainlabel=- |sort=Label Page }}
Patient Counseling Information
There is limited information regarding Patient Counseling Information of Clemastine in the drug label.
Precautions with Alcohol
- Alcohol-Clemastine interaction has not been established. Talk to your doctor about the effects of taking alcohol with this medication.
Brand Names
- Tavist®[1]
Look-Alike Drug Names
There is limited information regarding Clemastine Look-Alike Drug Names in the drug label.
Price
References
The contents of this FDA label are provided by the National Library of Medicine.


