Uveitis pathophysiology
|
Uveitis Microchapters |
|
Diagnosis |
|---|
|
Treatment |
|
Case Studies |
|
Uveitis pathophysiology On the Web |
|
American Roentgen Ray Society Images of Uveitis pathophysiology |
|
Risk calculators and risk factors for Uveitis pathophysiology |
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]; Associate Editor(s)-in-Chief: Tarek Nafee, M.D. [2]
Overview
The healthy eye possesses immune privilege, which suppresses immune responses to both endogenous retinal antigens (such as S-antigen) and exogenous antigens. This immune privilege is maintained by the blood–retina barrier, cellular mechanisms including regulatory T cells, and cytokine-mediated mechanisms involving transforming growth factor beta and interleukin-10.[1][2]
Uveitis is mediated by a genetic predisposition and an acquired cross immunity to infectious agents, or cross immunity to self-antigens due to underlying autoimmune diseases. It is estimated that 35% to 50% of cases of uveitis are idiopathic.[3][4] Development of uveitis is hypothesized to be the result of a genetic predisposition for acquiring the disease; contributions of several human leukocyte antigen (HLA) alleles are most commonly implicated.[4][5]This includes HLA-B27 in seronegative spondyloarthropathies, HLA-A29 in birdshot chorioretinopathy, HLA-B51 in Behcet syndrome and multiple sclerosis, HLA-B8, HLA-DR15, and HLA-DR2.[4][5]
Pathogenesis
Initiation of Uveitis
Noninfectious uveitis is hypothesized to result from reduced immune tolerance to retinal proteins, leading to inappropriate immune activation within the eye. In infectious uveitis, pathogenic organisms breach the blood–retina barrier and initiate inflammation; pathogen-derived antigens may resemble retinal proteins, a process known as antigenic mimicry, which can exacerbate the inflammatory response.[1][2] Infectious uveitis is hypothesized to be secondary to a T-cell mediated hypersensitivity reaction resulting in immunoactivating cytokine production by proliferating Tн17 and Tн1 cells.[5][6] A similar mechanism is hypothesized for autoimmune etiologies of uveitis with the exception of an unknown source of the triggering antigen.[7][8][9][10]
Antigen Presentation and T-Cell Activation
The prevailing mechanism in both infectious and noninfectious uveitis involves antigen presentation via major histocompatibility complex class II molecules, leading to activation of naïve CD4⁺ T cells. In infectious uveitis, antigen presentation is initiated by pathogen-derived antigens, whereas in noninfectious uveitis it is initiated by ocular autoantigens.[1][2]
Effector T-Cell Response
Upon activation, naïve CD4⁺ T cells differentiate predominantly into Th1 and Th17 subsets, which migrate to the retina and uveal tissues. These effector T cells play a central role in sustaining intraocular inflammation.[1][2]
Cytokine Cascade and Innate Immune Recruitment
Activated Th1 and Th17 cells release proinflammatory cytokines, including interferon-gamma, interleukin-2, and interleukin-17, triggering a cytokine cascade that recruits innate immune cells such as macrophages and neutrophils into ocular tissues.[2]
Inflammatory Tissue Injury
The accumulation of inflammatory cells and sustained cytokine signaling result in tissue damage, manifesting as chorioretinitis, retinal vasculitis, and edema, which contribute directly to visual dysfunction in uveitis.[2]

The retina’s immune privilege relies on the blood-retina barrier, which shields
ocular tissue proteins from the systemic immune system. This protective
mechanism can be compromised, leading to autoimmune reactions. Within the
retina, Tregs marked by CD4+, CD25+, and FoxP3+ identifiers contribute to
immune tolerance by emitting neuropeptides and anti-inflammatory cytokines.
These Tregs can suppress other T cells that have escaped elimination in the
thymus during development and have the potential to react against
self-antigens, producing cytokines, TGFβ, and IL-35 to reduce inflammation.
Furthermore, retinal pigment epithelium and retinal cells express certain
proteins on their surfaces that deactivate lymphocytes, thereby regulating
ocular inflammation. Disease is typically associated with major
histocompatibility complex class II molecule–mediated presentation of
autoantigens or cross-reactive foreign peptides to naive T cells. Activated CD4+
T cells differentiate into CD4+ TH1 and TH17 cells that migrate to the affected
tissue, recruiting inflammatory cells and producing tissue damage. T cells
differentiate into CD4+ TH1 and TH17 cells, producing IFNγ and IL-17,
respectively. These facilitate the recruitment and activation of downstream
cytokine release and innate inflammatory response, such as IL-6, TNF, and
granzyme B (a protease involved in programmed cell death), which in turn can
lead to vasculitis and edema.21,22
IFNγ indicates interferon gamma; TGFβ, transforming growth factor beta.
TNF, tumor necrosis factor; Treg, regulatory T cell.[11]
Associated Conditions
The following conditions are associated with uveitis:[3][4][12]
- Seronegative spondyloarthropathy
- Sarcoidosis
- Multiple sclerosis
- Inflammatory bowel disease
- Systemic lupus erythematosus (SLE)
- Behçet's syndrome
- Birdshot retinochoroidopathy
- Juvenile idiopathic arthritis (JIA)
- Vogt-Koyanagi-Harada syndrome
- Fuch's heterochromic iridocyclitis
- Lymphoma
- Lyme Disease
- Acquired immunodeficiency syndrome (AIDS)
- Herpes simplex
- Varicella Zoster
- Cytomegalovirus
- Syphilis
- Toxoplasmosis
- Toxocariasis
- Tuberculosis
- Rubella
- Cat scratch disease (Bartonella spp.)
Gross Pathology
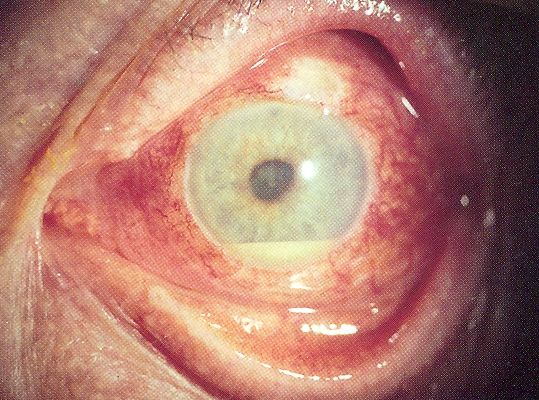
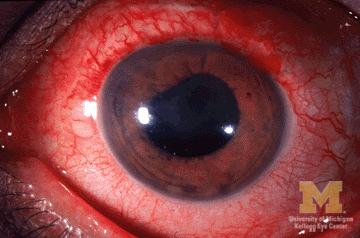
On gross pathology, redness of the eye and dilated cilliary vessels are common yet non-specific findings in uveitis. The following are images of the gross pathology of uveitis:[13][14]
-
Uveitis with pus in the anterior chamber
-
Anterior Uveitis
Microscopic Pathology
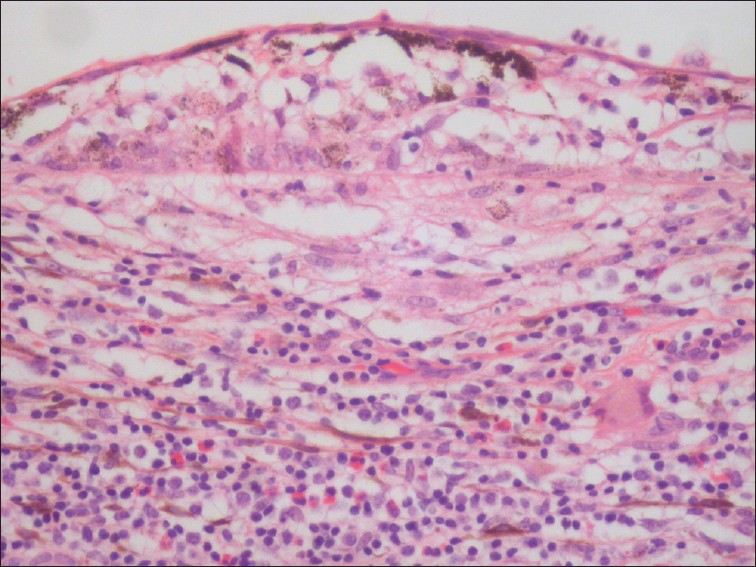
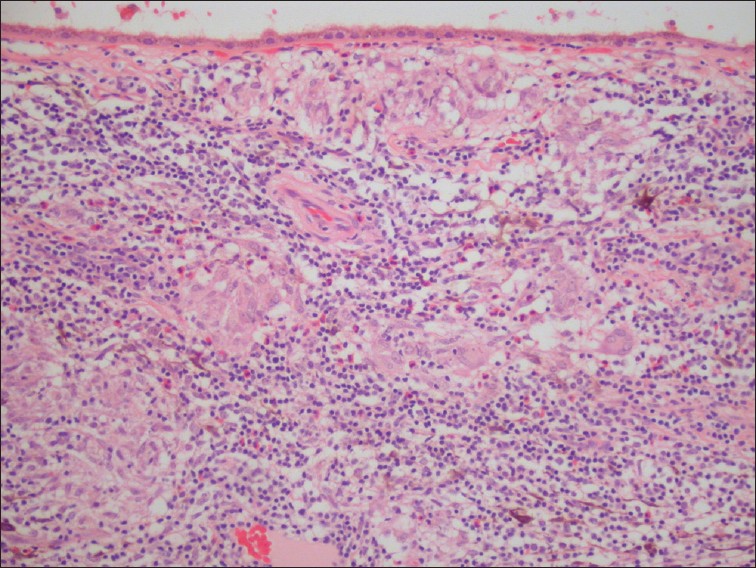
On microscopic histopathological analysis, lymphocytic infiltration, monocytic infiltration, epitheloid cells, and granulomatous inflammation of the uveal structures are characteristic findings of uveitis. The following are images of the microscopic pathology of uveitis:[15]
-
Uveitis with infiltration of lymphocytes
-
Uveitis with granulomatous inflammation and infiltration of the choroid by lymphocytes and macrophages
References
- ↑ 1.0 1.1 1.2 1.3 Lee, R. W. J., & Dick, A. D. (2012). Current concepts and future directions in the pathogenesis and treatment of non-infectious intraocular inflammation. Eye (London, England), 26(1), 17–28. https://doi.org/10.1038/eye.2011.255
- ↑ 2.0 2.1 2.2 2.3 2.4 2.5 Egwuagu, C. E., Alhakeem, S. A., & Mbanefo, E. C. (2021). Uveitis: Molecular pathogenesis and emerging therapies. Frontiers in Immunology, 12, 623725. https://doi.org/10.3389/fimmu.2021.623725
- ↑ 3.0 3.1 Rodriguez A, Calonge M, Pedroza-Seres M, Akova YA, Messmer EM, D'Amico DJ; et al. (1996). "Referral patterns of uveitis in a tertiary eye care center". Arch Ophthalmol. 114 (5): 593–9. PMID 8619771.
- ↑ 4.0 4.1 4.2 4.3 Guly CM, Forrester JV (2010). "Investigation and management of uveitis". BMJ. 341: c4976. doi:10.1136/bmj.c4976. PMID 20943722.
- ↑ 5.0 5.1 5.2 Przeździecka-Dołyk J, Węgrzyn A, Turno-Kręcicka A, Misiuk-Hojło M (2016). "Immunopathogenic Background of Pars Planitis". Arch Immunol Ther Exp (Warsz). 64 (2): 127–37. doi:10.1007/s00005-015-0361-y. PMC 4805694. PMID 26438050.
- ↑ Urban B, Bakunowicz-Łazarczyk A, Michalczuk M (2014). "Immune recovery uveitis: pathogenesis, clinical symptoms, and treatment". Mediators Inflamm. 2014: 971417. doi:10.1155/2014/971417. PMC 4096001. PMID 25089078.
- ↑ Sun D, Liang D, Kaplan HJ, Shao H (2015). "The role of Th17-associated cytokines in the pathogenesis of experimental autoimmune uveitis (EAU)". Cytokine. 74 (1): 76–80. doi:10.1016/j.cyto.2014.12.017. PMC 4457592. PMID 25742774.
- ↑ Bi HS, Liu ZF, Cui Y (2015). "Pathogenesis of innate immunity and adaptive immunity in the mouse model of experimental autoimmune uveitis". J Chin Med Assoc. 78 (5): 276–82. doi:10.1016/j.jcma.2015.01.002. PMID 25769932.
- ↑ Horai R, Caspi RR (2011). "Cytokines in autoimmune uveitis". J Interferon Cytokine Res. 31 (10): 733–44. doi:10.1089/jir.2011.0042. PMC 3189550. PMID 21787221.
- ↑ Ke Y, Liu K, Huang GQ, Cui Y, Kaplan HJ, Shao H; et al. (2009). "Anti-inflammatory role of IL-17 in experimental autoimmune uveitis". J Immunol. 182 (5): 3183–90. doi:10.4049/jimmunol.0802487. PMC 3275433. PMID 19234216.
- ↑ Maghsoudlou, P., Epps, S. J., Guly, C. M., & Dick, A. D. (2025). Uveitis in adults: A review: A review. The Journal of the American Medical Association, 334(5), 419–434. https://doi.org/10.1001/jama.2025.4358
- ↑ American Academy of Ophthalmology EyeWiki(2015)http://eyewiki.aao.org/Category:Uveitis
- ↑ Wikipedia Uveitis(2006)https://en.wikipedia.org/wiki/Uveitis#/media/File:Hypopyon.jpg
- ↑ University of Michigan Eyes Have It(2009)http://kellogg.umich.edu/theeyeshaveit/red-eye/anterior-uveitis.html
- ↑ Arevalo JF, Garcia RA, Al-Dhibi HA, Sanchez JG, Suarez-Tata L (2012). "Update on sympathetic ophthalmia". Middle East Afr J Ophthalmol. 19 (1): 13–21. doi:10.4103/0974-9233.92111. PMC 3277011. PMID 22346110.