Behçet's disease pathophysiology
|
Behçet's disease Microchapters |
|
Diagnosis |
|---|
|
Treatment |
|
Case Studies |
|
Behçet's disease pathophysiology On the Web |
|
American Roentgen Ray Society Images of Behçet's disease pathophysiology |
|
Risk calculators and risk factors for Behçet's disease pathophysiology |
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]; Associate Editor(s)-in-Chief: Hamid Qazi, MD, BSc [2], Mahda Alihashemi M.D. [3] Dheeraj Makkar, M.D.[4]
Overview
It is understood that Behçet disease is the result of vasculitis. It involves all sizes of blood vessels ( small, medium, and large). Arteries and veins are both involved in Behçet disease. Major mechanisms in pathogenesis of Behçet disease include environmental factors such as bacteria, viruses, and heat shock proteins (present in some bacteria and some of the bacterial HSPs share similaritis with human HSPs). Streptococcus sanguinis, streptococcus pyogenes, and mycobacterium tuberculosis produce HSPs that trigger anti HSP60 and anti HSP65 antibodies and then they target human HSPs and immune response such as uveitis in parenchymal neuro-Behçet disease, CD4+ T cells activation, secretion of cytokines and inflammation. Genes involved in the pathogenesis of Behçet disease include human leukocyte antigens, particularly HLA-B51.
Pathophysiology
Pathophysiology of Behçet’s Syndrome
- Genetic Predisposition
Behçet’s syndrome develops in genetically predisposed hosts after exposure to environmental triggers.
The first genetic association was with HLA-B*51, particularly HLA-B*51:01, which increases disease risk nearly sixfold.
Genome-wide association studies (GWAS) have since revealed additional key genes:
ERAP1 (antigen processing, interacts with HLA-B*51).
IL23R–IL12RB2, STAT4, IL10 (T-cell polarization toward Th1 and Th17 responses).
KLRC4 (NK cell regulation).
CCR1–CCR3 (cell chemotaxis).
Other implicated genes include TNFAIP3 (A20 haploinsufficiency), MEFV (familial Mediterranean fever), and TLR4/NOD2/FUT2, linking Behçet’s to autoinflammatory syndromes.
Epigenetic changes, such as aberrant DNA methylation and histone modifications in immune cells, further amplify susceptibility .
- Environmental and Microbial Triggers
Proposed triggers include:
Microorganisms (e.g., Streptococcus species, HSV-1).
Dietary factors (histamine-releasing foods such as citrus, nuts, and cheese).
Poor oral hygiene and stress.
Studies show gut and salivary microbiome dysbiosis, leading to abnormal antigen repertoires that may drive immune responses .
- Immune Dysregulation
Adaptive immunity:
CD4+ T-helper lymphocytes differentiate into Th1 (producing TNF-α, IFN-γ) and Th17 (producing IL-17, IL-23) subsets.
There is diminished regulatory T-cell activity, further tipping the balance toward inflammation.
CD8+ T cells and natural killer (NK) cells also contribute to cytotoxicity.
Innate immunity:
Neutrophils are the main infiltrating cell type in lesions.
They generate excessive reactive oxygen species (ROS) and release neutrophil extracellular traps (NETs), promoting vascular inflammation and thrombosis.
Key inflammatory pathway:
The NF-κB pathway is upregulated in antigen-presenting cells, neutrophils, and T cells, amplifying proinflammatory cytokine production .
- Histopathology
Biopsies often show:
Leukocytoclastic vasculitis.
Neutrophilic or lymphocytic perivascular infiltrates.
Microvascular thrombi.
Neutrophilic dermal infiltrates.
Pathogenesis
- It is understood that Behçet disease is the result of vasculitis. It involves all sizes of blood vessels ( small, medium, and large). Arteries and veins are both involved in Behçet disease. Major mechanisms in pathogenesis of Behçet disease include:[1]
- Polygenic
- Environmental factors such as bacteria, viruses, and heat shock proteins (present in some bacteria and some of the bacterial HSPs share similaritis with human HSPs).[2]
- Streptococcus sanguinis, streptococcus pyogenes, and mycobacterium tuberculosis produce HSPs that trigger anti HSP60 and anti HSP65 antibodies and then they target human HSPs and immune response such as uveitis in parenchymal neuro-Behçet disease.[3]
- CD4+ T cells activation, secretion of cytokines and inflammation.
Genetics
- The development of Behçet disease is the result of multiple genetic mutations.
- Genes involved in the pathogenesis of Behçet disease include human leukocyte antigens, particularly HLA-B51.[4]
- Familial cases of Behçet disease have higher rates of HLA-B51 in compare to sporadic cases.[5]
- Genetic anticipation: earlier age of onset of disease in children of patients with Behçet disease.[6]
- Some studies shows that major histocompatibility complex class I chain related gene A (MICA) A6 allele is related to Behçet disease. [7]
- Non-HLA genes also have roles in Behçet diesease. They include: [8]
- Intercellular adhesion molecule (ICAM)-1 gene
- Endothelial nitric oxide synthase gene[9]
- Tumor necrosis factor (TNF) genes[10]
- Vascular endothelial growth factor (VEGF) gene[11]
- Manganese superoxide dismutase gene[12]
- Cytochrome P450 gene[13]
- Interleukin (IL)-10 gene[14]
- IL-23 receptor gene[15]
- Missense mutations of the familial Mediterranean fever (MEFV) gene that encodes protein pyrin on the surface of neutrophils[16]
Gross Pathology
- On gross pathology oral ulcers, genital ulcers, uveitis, and skin lesions are characteristic findings of Behçet disease.
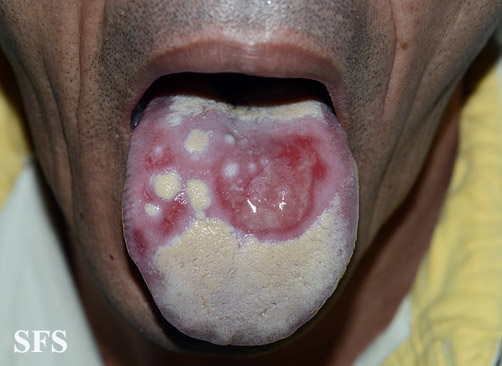
Microscopic Pathology
- On microscopic histopathological analysis, neutrophilic and CD4+ T lymphocytes infiltrate and vasculitis are characteristic findings of Behçet disease.
References
- ↑ Zeidan MJ, Saadoun D, Garrido M, Klatzmann D, Six A, Cacoub P (December 2016). "Behçet's disease physiopathology: a contemporary review". Auto Immun Highlights. 7 (1): 4. doi:10.1007/s13317-016-0074-1. PMC 4751097. PMID 26868128.
- ↑ Direskeneli H (2013). "Innate and Adaptive Responses to Heat Shock Proteins in Behcet's Disease". Genet Res Int. 2013: 249157. doi:10.1155/2013/249157. PMC 3893747. PMID 24490075.
- ↑ Tanaka T, Yamakawa N, Koike N, Suzuki J, Mizuno F, Usui M (June 1999). "Behçet's disease and antibody titers to various heat-shock protein 60s". Ocul. Immunol. Inflamm. 7 (2): 69–74. PMID 10420201.
- ↑ de Menthon M, Lavalley MP, Maldini C, Guillevin L, Mahr A (October 2009). "HLA-B51/B5 and the risk of Behçet's disease: a systematic review and meta-analysis of case-control genetic association studies". Arthritis Rheum. 61 (10): 1287–96. doi:10.1002/art.24642. PMC 3867978. PMID 19790126.
- ↑ Akpolat T, Koç Y, Yeniay I, Akpek G, Güllü I, Kansu E, Kiraz S, Ersoy F, Batman F, Kansu T (November 1992). "Familial Behçet's disease". Eur J Med. 1 (7): 391–5. PMID 1341477.
- ↑ Fresko I, Soy M, Hamuryudan V, Yurdakul S, Yavuz S, Tümer Z, Yazici H (January 1998). "Genetic anticipation in Behçet's syndrome". Ann. Rheum. Dis. 57 (1): 45–8. PMC 1752455. PMID 9536823.
- ↑ Wei F, Zhang YU, Li W (June 2016). "A meta-analysis of the association between Behçet's disease and MICA-A6". Biomed Rep. 4 (6): 741–745. doi:10.3892/br.2016.644. PMC 4887777. PMID 27284416.
- ↑ Sakane T, Takeno M (September 2000). "Novel approaches to Behçet's disease". Expert Opin Investig Drugs. 9 (9): 1993–2005. doi:10.1517/13543784.9.9.1993. PMID 11060788.
- ↑ Karasneh JA, Hajeer AH, Silman A, Worthington J, Ollier WE, Gul A (May 2005). "Polymorphisms in the endothelial nitric oxide synthase gene are associated with Behçet's disease". Rheumatology (Oxford). 44 (5): 614–7. doi:10.1093/rheumatology/keh561. PMID 15705632.
- ↑ Ahmad T, Wallace GR, James T, Neville M, Bunce M, Mulcahy-Hawes K, Armuzzi A, Crawshaw J, Fortune F, Walton R, Stanford MR, Welsh KI, Marshall SE, Jewell DP (March 2003). "Mapping the HLA association in Behçet's disease: a role for tumor necrosis factor polymorphisms?". Arthritis Rheum. 48 (3): 807–13. doi:10.1002/art.10815. PMID 12632436.
- ↑ Salvarani C, Boiardi L, Casali B, Olivieri I, Cantini F, Salvi F, Malatesta R, La Corte R, Triolo G, Ferrante A, Filippini D, Paolazzi G, Sarzi-Puttini P, Nicoli D, Farnetti E, Chen Q, Pulsatelli L (September 2004). "Vascular endothelial growth factor gene polymorphisms in Behçet's disease". J. Rheumatol. 31 (9): 1785–9. PMID 15338501.
- ↑ Nakao K, Isashiki Y, Sonoda S, Uchino E, Shimonagano Y, Sakamoto T (February 2007). "Nitric oxide synthase and superoxide dismutase gene polymorphisms in Behçet disease". Arch. Ophthalmol. 125 (2): 246–51. doi:10.1001/archopht.125.2.246. PMID 17296902.
- ↑ Tursen U, Tamer L, Api H, Yildirim H, Baz K, Ikizoglu G, Atik U (February 2007). "Cytochrome P450 polymorphisms in patients with Behcet's disease". Int. J. Dermatol. 46 (2): 153–6. doi:10.1111/j.1365-4632.2007.02957.x. PMID 17269966.
- ↑ Sousa I, Shahram F, Francisco D, Davatchi F, Abdollahi BS, Ghaderibarmi F, Nadji A, Mojarad Shafiee N, Xavier JM, Oliveira SA (October 2015). "Brief report: association of CCR1, KLRC4, IL12A-AS1, STAT4, and ERAP1 With Behçet's disease in Iranians". Arthritis Rheumatol. 67 (10): 2742–8. doi:10.1002/art.39240. PMID 26097239.
- ↑ Yalçin B, Atakan N, Dogan S (December 2014). "Association of interleukin-23 receptor gene polymorphism with Behçet disease". Clin. Exp. Dermatol. 39 (8): 881–7. doi:10.1111/ced.12400. PMID 25156021.
- ↑ Livneh A, Aksentijevich I, Langevitz P, Torosyan Y, G-Shoham N, Shinar Y, Pras E, Zaks N, Padeh S, Kastner DL, Pras M (March 2001). "A single mutated MEFV allele in Israeli patients suffering from familial Mediterranean fever and Behçet's disease (FMF-BD)". Eur. J. Hum. Genet. 9 (3): 191–6. doi:10.1038/sj.ejhg.5200608. PMID 11313758.