Amikacin sulfate
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]; Associate Editor(s)-in-Chief: Alberto Plate [2]
WikiDoc MAKES NO GUARANTEE OF VALIDITY. WikiDoc is not a professional health care provider, nor is it a suitable replacement for a licensed healthcare provider. WikiDoc is intended to be an educational tool, not a tool for any form of healthcare delivery. The educational content on WikiDoc drug pages is based upon the FDA package insert, National Library of Medicine content and practice guidelines / consensus statements. WikiDoc does not promote the administration of any medication or device that is not consistent with its labeling. Please read our full disclaimer here.
Black Box Warning
|
Warning
See full prescribing information for complete Boxed Warning.
Condition Name: Patients treated with parenteral aminoglycosides should be under close clinical observation because of the potential ototoxicity and nephrotoxicity associated with their use. Safety for treatment periods which are longer than 14 days has not been established.
Neurotoxicity, manifested as vestibular and permanent bilateral auditory ototoxicity, can occur in patients with preexisting renal damage and in patients with normal renal function treated at higher doses and/or for periods longer than those recommended. The risk of aminoglycoside-induced ototoxicity is greater in patients with renal damage. High frequency deafness usually occurs first and can be detected only by audiometric testing. Vertigo may occur and may be evidence of vestibular injury. Other manifestations of neurotoxicity may include numbness, skin tingling, muscle twitching and convulsions. The risk of hearing loss due to aminoglycosides increases with the degree of exposure to either high peak or high trough serum concentrations. Patients developing cochlear damage may not have symptoms during therapy to warn them of developing eighth-nerve toxicity, and total or partial irreversible bilateral deafness may occur after the drug has been discontinued. Aminoglycoside-induced ototoxicity is usually irreversible. Aminoglycosides are potentially nephrotoxic. The risk of nephrotoxicity is greater in patients with impaired renal function and in those who receive high doses or prolonged therapy. Neuromuscular blockade and respiratory paralysis have been reported following parenteral injection, topical instillation (as in orthopedic and abdominal irrigation or in local treatment of empyema), and following oral use of aminoglycosides. The possibility of these phenomena should be considered if aminoglycosides are administered by any route, especially in patients receiving anesthetics; neuromuscular blocking agents such as tubocurarine, succinylcholine, decamethonium; or in patients receiving massive transfusions of citrate-anticoagulated blood. If blockage occurs, calcium salts may reverse these phenomena, but mechanical respiratory assistance may be necessary. Renal and eighth-nerve function should be closely monitored especially in patients with known or suspected renal impairment at the onset of therapy and also in those whose renal function is initially normal but who develop signs of renal dysfunction during therapy. Serum concentrations of amikacin should be monitored when feasible to assure adequate levels and to avoid potentially toxic levels and prolonged peak concentrations above 35 micrograms per mL. Urine should be examined for decreased specific gravity, increased excretion of proteins and the presence of cells or casts. Blood urea nitrogen, serum creatinine or creatinine clearance should be measured periodically. Serial audiograms should be obtained where feasible in patients old enough to be tested, particularly high risk patients. Evidence of ototoxicity (dizziness, vertigo, tinnitus, roaring in the ears and hearing loss) or nephrotoxicity requires discontinuation of the drug or dosage adjustment. Concurrent and/or sequential systemic, oral or topical use of other neurotoxic or nephrotoxic products, particularly bacitracin, cisplatin, amphotericin B, cephaloridine, paromomycin, viomycin, polymyxin B, colistin, vancomycin or other aminoglycosides should be avoided. Other factors that may increase risk of toxicity are advanced age and dehydration. The concurrent use of amikacin with potent diuretics (ethacrynic acid or furosemide) should be avoided since diuretics by themselves may cause ototoxicity. In addition, when administered intravenously, diuretics may enhance aminoglycoside toxicity by altering antibiotic concentrations in serum and tissue.) |
Overview
Amikacin sulfate is an antibiotic that is FDA approved for the treatment of infections due to susceptible strains of Gram-negative bacteria, including Pseudomonas species, Escherichia coli, species of indole-positive and indole-negative Proteus, Providencia species, Klebsiella-Enterobacter-Serratia species, Acinetobacter (Mima-Herellea) species, Staphylococcus and Gram-positive organisms, such as streptococcus. Clinical studies have proven efficacy in: bacterial septicemia (including neonatal sepsis), severe respiratory tract infections, bone infections, joint infections, central nervous system infections (including meningitis), skin and soft tissue infections, intra-abdominal infections (including peritonitis), burns, post-operative infections (including post-vascular surgery), serious complicated and recurrent urinary tract infections. There is a Black Box Warning for this drug as shown here. Common adverse reactions include neuromuscular blockade, ototoxicity, nephrotoxicity and respiratory paralysis..
Adult Indications and Dosage
FDA-Labeled Indications and Dosage (Adult)
Infections due to susceptible strains of Gram-negative bacteria, including Pseudomonas species, Escherichia coli, species of indole-positive and indole-negative Proteus, Providencia species, Klebsiella-Enterobacter-Serratia species, Acinetobacter (Mima-Herellea) species, Staphylococcus and Gram-positive organisms, such as streptococcus. Clinical studies have proven efficacy in:
- Bacterial Septicemia (including neonatal sepsis)
- Severe respiratory tract infections
- Bone infections
- Joint infections
- Central nervous system infections (including meningitis)
- Dosage:
- IV: 15 mg/kg/day IV divided in doses q8h.
- Intraventricular: 5mg-50 mg/day.
- Dosage:
- Skin and soft tissue infections
- Intra-abdominal infections (including peritonitis).
- Burns
- Post-operative infections (including post-vascular surgery)
- Serious complicated and recurrent urinary tract infections
Dosage
Intramuscular Administration for Patients with Normal Renal Function
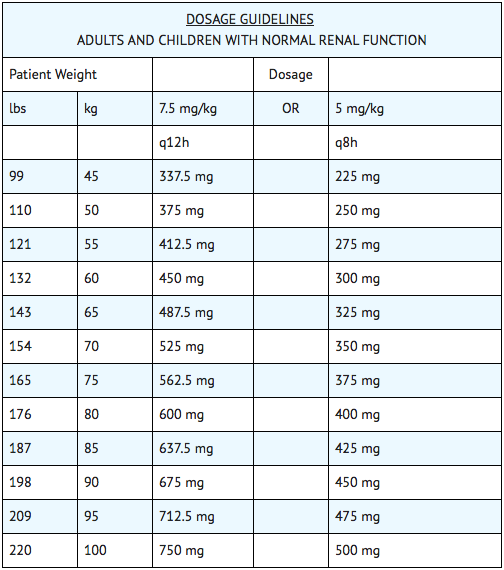
- The recommended dosage for adults, children and older infants with normal renal function is 15 mg/kg/day divided into 2 or 3 equal doses administered at equally divided intervals, i.e., 7.5 mg/kg q12h or 5 mg/kg q8h. Treatment of patients in the heavier weight classes should not exceed 1.5 g/day. When amikacin is indicated in newborns, it is recommended that a loading dose of 10 mg/kg be administered initially to be followed with 7.5 mg/kg every 12 hours.
- The usual duration of treatment is 7 to 10 days. It is desirable to limit the duration of treatment to short-term whenever feasible. The total daily dose by all routes of administration should not exceed 15 mg/kg/day. In difficult and complicated infections where treatment beyond 10 days is considered, the use of amikacin should be reevaluated. If continued, amikacin serum levels and renal, auditory and vestibular functions should be monitored. At the recommended dosage level, uncomplicated infections due to amikacin-sensitive organisms should respond in 24 to 48 hours. If definite clinical response does not occur within 3 to 5 days, therapy should be stopped and the antibiotic susceptibility pattern of the invading organism should be rechecked. Failure of the infection to respond may be due to resistance of the organism or to the presence of septic foci requiring surgical drainage. When amikacin is indicated in uncomplicated urinary tract infections, a dose of 250 mg twice daily may be used.
Intramuscular Administration for Patients with Impaired Renal Function
- Whenever possible, serum amikacin concentrations should be monitored by appropriate assay procedures. Doses may be adjusted in patients with impaired renal function either by administering normal doses at prolonged intervals or by administering reduced doses at a fixed interval.
- Both methods are based on the patient's creatinine clearance or serum creatinine values since these have been found to correlate with aminoglycoside half-lives in patients with diminished renal function. These dosage schedules must be used in conjunction with careful clinical and laboratory observations of the patient and should be modified as necessary. Neither method should be used when dialysis is being performed.
- Normal Dosage at Prolonged Intervals: If the creatinine clearance rate is not available and the patient's condition is stable, a dosage interval in hours for the normal dose can be calculated by multiplying the patient's serum creatinine by 9, e.g., if the serum creatinine concentration is 2 mg/100 mL, the recommended single dose (7.5 mg/kg) should be administered every 18 hours.
- Reduced Dosage at Fixed Time Intervals: When renal function is impaired and it is desirable to administer amikacin at a fixed time interval, dosage must be reduced. In these patients, serum amikacin concentrations should be measured to assure accurate administration of amikacin and to avoid concentrations above 35 mcg/mL. If serum assay determinations are not available and the patient's condition is stable, serum creatinine and creatinine clearance values are the most readily available indicators of the degree of renal impairment to use as a guide for dosage. First, initiate therapy by administering a normal dose, 7.5 mg/kg, as a loading dose. This loading dose is the same as the normally recommended dose which would be calculated for a patient with a normal renal function as described above. To determine the size of maintenance doses administered every 12 hours, the loading dose should be reduced in proportion to the reduction in the patient's creatinine clearance rate:

Intravenous Administrations
- The individual dose, the total daily dose, and the total cumulative dose of amikacin sulfate are identical to the dose recommended for intramuscular administration. The solution for intravenous use is prepared by adding the contents of a 500 mg vial to 100 or 200 mL of sterile diluent such as 0.9% sodium chloride injection or 5% dextrose injection or any other compatible solutions listed below. The solution is administered to adults over a 30 to 60 minute period. The total daily dose should not exceed 15 mg/kg/day and may be divided into either 2 or 3 equally-divided doses at equally-divided intervals.
- In pediatric patients, the amount of fluid used will depend on the amount of amikacin sulfate ordered for the patient. It should be a sufficient amount to infuse the amikacin over a 30 to 60 minute period. Infants should receive a 1 to 2 hour infusion.
- Amikacin should not be physically premixed with other drugs but should be administered separately according to the recommended dose and route.
Off-Label Use and Dosage (Adult)
Guideline-Supported Use
Febrile Neutropenia
- 2010 Guideline - Treatment of febrile neutropenia in cancer patients [1]
Tuberculosis
Used as a second-line drug
- Dosage: 15 mg/kg per day (1.0 g/day), intramuscular or intravenous, usually given as a single daily dose (5--7 days/week) [2]
Nosocomial Pneumonia
- Dosage: 20 mg/kg per day
Non–Guideline-Supported Use
Bacterial Endocarditis
- Dosage: Amikacin[3] 7.5 mg/kg/12h + Cloxacillin 2 g/4h (Duration of therapy: 2 weeks).
Eye Infection
- Dosage: 25 mg subconjunctival
Mycobacteriosis
- Dosage: 7.5 mg/kg IV administered for 4 weeks in a multidrug regimen including ethambutol, rifampin, and ciprofloxacin[4].
Nocardiosis
- Dosage: 15 mg/kg/day IM dividen in 2 doses every 21 days [5]
Pediatric Indications and Dosage
FDA-Labeled Indications and Dosage (Pediatric)
Infections due to susceptible strains of Gram-negative bacteria, including Pseudomonas species, Escherichia coli, species of indole-positive and indole-negative Proteus, Providencia species, Klebsiella-Enterobacter-Serratia species, Acinetobacter (Mima-Herellea) species, Staphylococcus and Gram-positive organisms, such as streptococcus. Clinical studies have proven efficacy in:
- Bacterial Septicemia (including neonatal sepsis)
- Severe respiratory tract infections
- Bone infections
- Joint infections
- Central nervous system infections (including meningitis)
- Dosage:
- IV: 15 mg/kg/day IV divided in doses q8h.
- Intraventricular: 5mg-50 mg/day.
- Dosage:
- Skin and soft tissue infections
- Intra-abdominal infections (including peritonitis).
- Burns
- Post-operative infections (including post-vascular surgery)
- Serious complicated and recurrent urinary tract infections
Off-Label Use and Dosage (Pediatric)
Guideline-Supported Use
Tuberculosis
Used as a second-line drug
- Dosage: 15-30 mg/kg per day (1 g/day) intramuscular or intravenous as a single daily dose.) [6]
Nosocomial Pneumonia
- Dosage: 20 mg/kg per day
Non–Guideline-Supported Use
There is limited information regarding Off-Label Non–Guideline-Supported Use of Amikacin sulfate in pediatric patients.
Contraindications
A history of hypersensitivity to amikacin is a contraindication for its use. A history of hypersensitivity or serious toxic reactions to aminoglycosides may contraindicate the use of any other aminoglycoside because of the known cross-sensitivities of patients to drugs in this class.
Warnings
|
Warning
See full prescribing information for complete Boxed Warning.
Condition Name: Patients treated with parenteral aminoglycosides should be under close clinical observation because of the potential ototoxicity and nephrotoxicity associated with their use. Safety for treatment periods which are longer than 14 days has not been established.
Neurotoxicity, manifested as vestibular and permanent bilateral auditory ototoxicity, can occur in patients with preexisting renal damage and in patients with normal renal function treated at higher doses and/or for periods longer than those recommended. The risk of aminoglycoside-induced ototoxicity is greater in patients with renal damage. High frequency deafness usually occurs first and can be detected only by audiometric testing. Vertigo may occur and may be evidence of vestibular injury. Other manifestations of neurotoxicity may include numbness, skin tingling, muscle twitching and convulsions. The risk of hearing loss due to aminoglycosides increases with the degree of exposure to either high peak or high trough serum concentrations. Patients developing cochlear damage may not have symptoms during therapy to warn them of developing eighth-nerve toxicity, and total or partial irreversible bilateral deafness may occur after the drug has been discontinued. Aminoglycoside-induced ototoxicity is usually irreversible. Aminoglycosides are potentially nephrotoxic. The risk of nephrotoxicity is greater in patients with impaired renal function and in those who receive high doses or prolonged therapy. Neuromuscular blockade and respiratory paralysis have been reported following parenteral injection, topical instillation (as in orthopedic and abdominal irrigation or in local treatment of empyema), and following oral use of aminoglycosides. The possibility of these phenomena should be considered if aminoglycosides are administered by any route, especially in patients receiving anesthetics; neuromuscular blocking agents such as tubocurarine, succinylcholine, decamethonium; or in patients receiving massive transfusions of citrate-anticoagulated blood. If blockage occurs, calcium salts may reverse these phenomena, but mechanical respiratory assistance may be necessary. Renal and eighth-nerve function should be closely monitored especially in patients with known or suspected renal impairment at the onset of therapy and also in those whose renal function is initially normal but who develop signs of renal dysfunction during therapy. Serum concentrations of amikacin should be monitored when feasible to assure adequate levels and to avoid potentially toxic levels and prolonged peak concentrations above 35 micrograms per mL. Urine should be examined for decreased specific gravity, increased excretion of proteins and the presence of cells or casts. Blood urea nitrogen, serum creatinine or creatinine clearance should be measured periodically. Serial audiograms should be obtained where feasible in patients old enough to be tested, particularly high risk patients. Evidence of ototoxicity (dizziness, vertigo, tinnitus, roaring in the ears and hearing loss) or nephrotoxicity requires discontinuation of the drug or dosage adjustment. Concurrent and/or sequential systemic, oral or topical use of other neurotoxic or nephrotoxic products, particularly bacitracin, cisplatin, amphotericin B, cephaloridine, paromomycin, viomycin, polymyxin B, colistin, vancomycin or other aminoglycosides should be avoided. Other factors that may increase risk of toxicity are advanced age and dehydration. The concurrent use of amikacin with potent diuretics (ethacrynic acid or furosemide) should be avoided since diuretics by themselves may cause ototoxicity. In addition, when administered intravenously, diuretics may enhance aminoglycoside toxicity by altering antibiotic concentrations in serum and tissue.) |
Fetal Toxicity
- Aminoglycosides can cause fetal harm when administered to a pregnant woman. Aminoglycosides cross the placenta and there have been several reports of total irreversible, bilateral congenital deafness in children whose mothers received streptomycin during pregnancy. Although serious side effects to the fetus or newborns have not been reported in the treatment of pregnant women with other aminoglycosides, the potential for harm exists. Reproduction studies of amikacin have been performed in rats and mice and revealed no evidence of impaired fertility or harm to the fetus due to amikacin. There are no well-controlled studies in pregnant women, but investigational experience does not include any positive evidence of adverse effects to the fetus. If this drug is used during pregnancy, or if the patient becomes pregnant while taking this drug, the patient should be apprised of the potential hazard to the fetus.
Hypersensitivity
- Contains sodium metabisulfite, a sulfite that may cause allergic-type reactions including anaphylactic symptoms and life-threatening or less severe asthmatic episodes in certain susceptible people. The overall prevalence of sulfite sensitivity in the general population is unknown and probably low. Sulfite sensitivity is seen more frequently in asthmatic than non-asthmatic people.
Clostridium Difficile Associated Diarrhea
- Clostridium difficile associated diarrhea (CDAD) has been reported with use of nearly all antibacterial agents, including amikacin, and may range in severity from mild diarrhea to fatal colitis. Treatment with antibacterial agents alters the normal flora of the colon leading to overgrowth of C. difficile.
- C. difficile produces toxins A and B which contribute to the development of CDAD. Hypertoxin-producing strains of C. difficile cause increased morbidity and mortality, as these infections can be refractory to antimicrobial therapy and may require colectomy. CDAD must be considered in all patients who present with diarrhea following antibacterial use. Careful medical history is necessary since CDAD has been reported to occur over 2 months after the administration of antibacterial agents.
- If CDAD is suspected or confirmed, ongoing antibiotic use not directed against C. difficile may need to be discontinued. Appropriate fluid and electrolyte management, protein supplementation, antibiotic treatment of C. difficile, and surgical evaluation should be instituted as clinically indicated
Adverse Reactions
Clinical Trials Experience
Cardiovascular Effects
Dermatologic Effects
Gastrointestinal Effects
Hematological Effects
Musculoeskeletal Effects
Neurologic Effects
- Headache
- Neuromuscular blockade: Acute muscular paralysis and apnea can occur following treatment with aminoglycoside drugs.
- Paresthesia
- Tremor
Ophthalmic Effects
Otic Effects
- Ototoxicity: Toxic effects on the eighth cranial nerve can result in hearing loss, loss of balance, or both. Amikacin primarily affects auditory function. Cochlear damage includes high frequency deafness and usually occurs before clinical hearing loss can be detected.
Renal Effects
- Nephrotoxicity: Elevation of serum creatinine, albuminuria, presence of red and white cells, casts, azotemia, and oliguria have been reported. Renal function changes are usually reversible when the drug is discontinued. As would be expected with any aminoglycoside, reports of toxic nephropathy and acute renal failure have been received during postmarketing surveillance.
Respiratory Effects
Other
Postmarketing Experience
There is limited information regarding Amikacin sulfate Postmarketing Experience in the drug label.
Drug Interactions
There is limited information regarding Amikacin sulfate Drug Interactions in the drug label.
Use in Specific Populations
Pregnancy
Pregnancy Category (FDA): D
There is no FDA guidance on usage of Amikacin sulfate in women who are pregnant.
Pregnancy Category (AUS):
There is no Australian Drug Evaluation Committee (ADEC) guidance on usage of Amikacin sulfate in women who are pregnant.
Labor and Delivery
There is no FDA guidance on use of Amikacin sulfate during labor and delivery.
Nursing Mothers
- It is not known whether this drug is excreted in human milk. Because many drugs are excreted in human milk and because of the potential for serious adverse reactions in nursing infants from amikacin, a decision should be made whether to discontinue nursing or to discontinue the drug, taking into account the importance of the drug to the mother.
Pediatric Use
- Aminoglycosides should be used with caution in premature and neonatal infants because of the renal immaturity of these patients and the resulting prolongation of serum half-life of these drugs.
Geriatic Use
There is no FDA guidance on the use of Amikacin sulfate in geriatric settings.
Gender
There is no FDA guidance on the use of Amikacin sulfate with respect to specific gender populations.
Race
There is no FDA guidance on the use of Amikacin sulfate with respect to specific racial populations.
Renal Impairment
Intramuscular Administration for Patients with Impaired Renal Function
- Whenever possible, serum amikacin concentrations should be monitored by appropriate assay procedures. Doses may be adjusted in patients with impaired renal function either by administering normal doses at prolonged intervals or by administering reduced doses at a fixed interval.
- Both methods are based on the patient's creatinine clearance or serum creatinine values since these have been found to correlate with aminoglycoside half-lives in patients with diminished renal function. These dosage schedules must be used in conjunction with careful clinical and laboratory observations of the patient and should be modified as necessary. Neither method should be used when dialysis is being performed.
- Normal Dosage at Prolonged Intervals: If the creatinine clearance rate is not available and the patient's condition is stable, a dosage interval in hours for the normal dose can be calculated by multiplying the patient's serum creatinine by 9, e.g., if the serum creatinine concentration is 2 mg/100 mL, the recommended single dose (7.5 mg/kg) should be administered every 18 hours.
- Reduced Dosage at Fixed Time Intervals: When renal function is impaired and it is desirable to administer amikacin at a fixed time interval, dosage must be reduced. In these patients, serum amikacin concentrations should be measured to assure accurate administration of amikacin and to avoid concentrations above 35 mcg/mL. If serum assay determinations are not available and the patient's condition is stable, serum creatinine and creatinine clearance values are the most readily available indicators of the degree of renal impairment to use as a guide for dosage. First, initiate therapy by administering a normal dose, 7.5 mg/kg, as a loading dose. This loading dose is the same as the normally recommended dose which would be calculated for a patient with a normal renal function as described above. To determine the size of maintenance doses administered every 12 hours, the loading dose should be reduced in proportion to the reduction in the patient's creatinine clearance rate:

Hepatic Impairment
There is no FDA guidance on the use of Amikacin sulfate in patients with hepatic impairment.
Females of Reproductive Potential and Males
There is no FDA guidance on the use of Amikacin sulfate in women of reproductive potentials and males.
Immunocompromised Patients
There is no FDA guidance one the use of Amikacin sulfate in patients who are immunocompromised.
Administration and Monitoring
Administration
- Intramuscular
- IV
- Subconjuntival
- Topic
Monitoring
Elderly patients may have reduced renal function which may not be evident in routine screening tests such as BUN or serum creatinine. A creatinine clearance determination may be more useful. Monitoring of renal function during treatment with aminoglycosides is particularly important.
IV Compatibility
There is limited information regarding the compatibility of Amikacin sulfate and IV administrations.
Overdosage
- In the event of overdosage or toxic reaction, peritoneal dialysis or hemodialysis will aid in the removal of amikacin from the blood. In the newborn infant, exchange transfusion may also be considered.
Pharmacology
Mechanism of Action
There is limited information regarding Amikacin sulfate Mechanism of Action in the drug label.
Structure
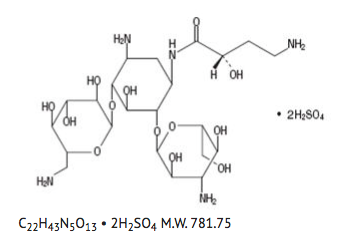
- Amikacin Sulfate Injection USP is a semi-synthetic aminoglycoside antibiotic derived from kanamycin. D-Streptamine,O-3-amino-3-deoxy-α-D-glucopyranosyl-(1→6)-O-[6-amino-6-deoxy-α-D-glucopyranosyl-(1→4)]-N1-(4-amino-2-hydroxy-1-oxobutyl)-2-deoxy-, (S)-, sulfate (1:2) (salt).
Pharmacodynamics
There is limited information regarding Amikacin sulfate Pharmacodynamics in the drug label.
Pharmacokinetics
General
- Pharmacokinetic studies in normal adult subjects reveal the mean serum half-life to be slightly over 2 hours with a mean total apparent volume of distribution of 24 liters (28% of the body weight). By the ultrafiltration technique, reports of serum protein binding range from 0 to 11%. The mean serum clearance rate is about 100 mL/min and the renal clearance rate is 94 mL/min in subjects with normal renal function.
- Amikacin is excreted primarily by glomerular filtration. Patients with impaired renal function or diminished glomerular filtration pressure excrete the drug much more slowly (effectively prolonging the serum half-life). Therefore, renal function should be monitored carefully and dosage adjusted accordingly.
- Following administration at the recommended dose, therapeutic levels are found in bone, heart, gallbladder, and lung tissue in addition to significant concentrations in urine, bile, sputum, bronchial secretions, interstitial, pleural, and synovial fluids.
- Spinal fluid levels in normal infants are approximately 10 to 20% of the serum concentrations and may reach 50% when the meninges are inflamed. Amikacin has been demonstrated to cross the placental barrier and yield significant concentrations in amniotic fluid. The peak fetal serum concentration is about 16% of the peak maternal serum concentration and maternal and fetal serum half-life values are about 2 and 3.7 hours, respectively.
Intramuscular Administration
- Amikacin is rapidly absorbed after intramuscular administration. In normal adult volunteers, average peak serum concentrations of about 12, 16 and 21 mcg/mL are obtained 1 hour after intramuscular administration of 250 mg (3.7 mg/kg), 375 mg (5 mg/kg), 500 mg (7.5 mg/kg), single doses, respectively. At 10 hours, serum levels are about 0.3 mcg/mL, 1.2 mcg/mL and 2.1 mcg/mL, respectively.
- Tolerance studies in normal volunteers reveal that amikacin is well tolerated locally following repeated intramuscular dosing, and when given at maximally recommended doses, no ototoxicity or nephrotoxicity has been reported. There is no evidence of drug accumulation with repeated dosing for 10 days when administered according to recommended doses.
- With normal renal function, about 91.9% of an intramuscular dose is excreted unchanged in the urine in the first 8 hours and 98.2% within 24 hours. Mean urine concentrations for 6 hours are 563 mcg/mL following a 250 mg dose, 697 mcg/mL following a 375 mg dose and 832 mcg/mL following a 500 mg dose.
- Preliminary intramuscular studies in newborns of different weights (less than 1.5 kg, 1.5 to 2 kg, over 2 kg) at a dose of 7.5 mg/kg revealed that, like other aminoglycosides, serum half-life values were correlated inversely with post-natal age and renal clearances of amikacin. The volume of distribution indicates that amikacin, like other aminoglycosides, remains primarily in the extra-cellular fluid space of neonates. Repeated dosing every 12 hours in all the above groups did not demonstrate accumulation after 5 days.
Intravenous Administration
- Single doses of 500 mg (7.5 mg/kg) administered to normal adults as an infusion over a period of 30 minutes produced a mean peak serum concentration of 38 mcg/mL at the end of the infusion and levels of 24 mcg/mL, 18 mcg/mL and 0.75 mcg/mL at 30 minutes, 1 hour and 10 hours post-infusion, respectively. Eighty-four percent of the administered dose was excreted in the urine in 9 hours and about 94% within 24 hours. Repeat infusions of 7.5 mg/kg every 12 hours in normal adults were well tolerated and caused no drug accumulation.
Nonclinical Toxicology
There is limited information regarding Amikacin sulfate Nonclinical Toxicology in the drug label.
Clinical Studies
- Clinical studies have shown amikacin sulfate injection USP to be effective in bacterial septicemia (including neonatal sepsis); in serious infections of the respiratory tract, bones and joints, central nervous system (including meningitis) and skin and soft tissue; intra-abdominal infections (including peritonitis); and in burns and postoperative infections (including post-vascular surgery). Clinical studies have shown amikacin also to be effective in serious complicated and recurrent urinary tract infections due to these organisms. Aminoglycosides, including amikacin sulfate injection USP, are not indicated in uncomplicated initial episodes of urinary tract infections unless the causative organisms are not susceptible to antibiotics having less potential toxicity.
How Supplied
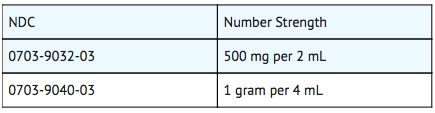
- Amikacin Sulfate Injection, USP is supplied as a colorless solution which requires no refrigeration. At times the solution may become a very pale yellow; this does not indicate a decrease in potency.
Storage
- Store at 20° to 25°C (68° to 77°F) [see USP Controlled Room Temperature].
Images
Drug Images
{{#ask: Page Name::Amikacin sulfate |?Pill Name |?Drug Name |?Pill Ingred |?Pill Imprint |?Pill Dosage |?Pill Color |?Pill Shape |?Pill Size (mm) |?Pill Scoring |?NDC |?Drug Author |format=template |template=DrugPageImages |mainlabel=- |sort=Pill Name }}
Package and Label Display Panel
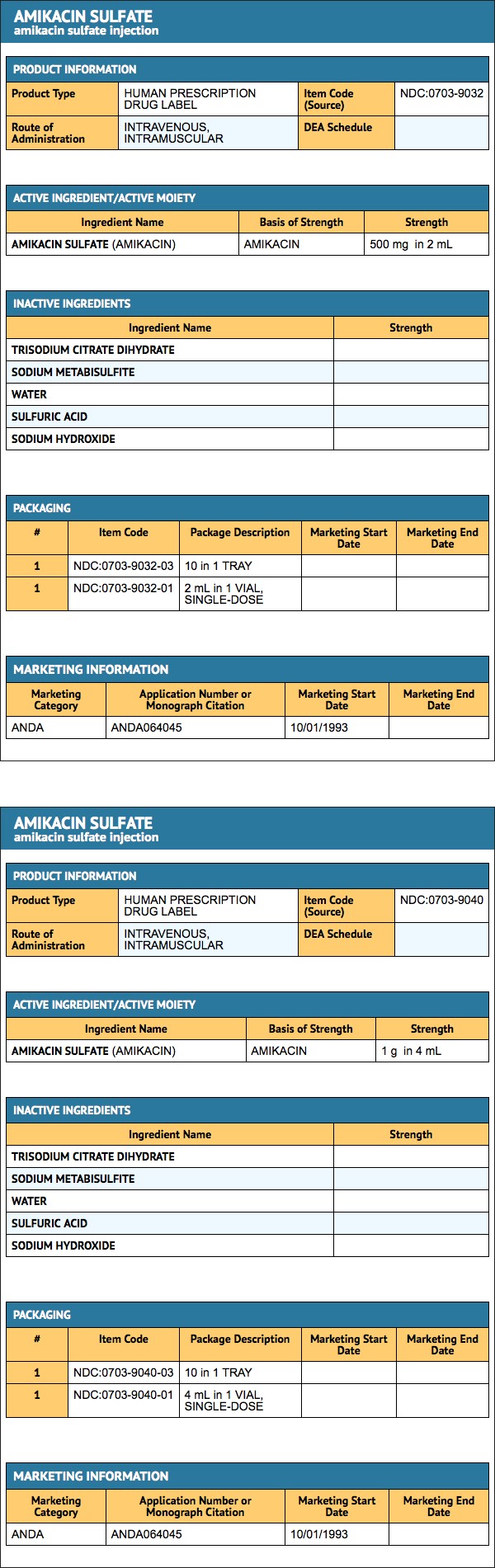
{{#ask: Label Page::Amikacin sulfate |?Label Name |format=template |template=DrugLabelImages |mainlabel=- |sort=Label Page }}
Patient Counseling Information
There is limited information regarding Amikacin sulfate Patient Counseling Information in the drug label.
Precautions with Alcohol
- Alcohol-Amikacin sulfate interaction has not been established. Talk to your doctor about the effects of taking alcohol with this medication.
Brand Names
Look-Alike Drug Names
There is limited information regarding Amikacin sulfate Look-Alike Drug Names in the drug label.
Price
References
The contents of this FDA label are provided by the National Library of Medicine.
- ↑ "Clinical practice guideline for the use of antimicrobial agents in neutropenic patients with cancer: 2010 update by the infectious diseases society of america" (PDF).
- ↑ "CDC Guideline for treatment of Tuberculosis".
- ↑ Torres-Tortosa M, de Cueto M, Vergara A, Sánchez-Porto A, Pérez-Guzmán E, González-Serrano M; et al. (1994). "Prospective evaluation of a two-week course of intravenous antibiotics in intravenous drug addicts with infective endocarditis. Grupo de Estudio de Enfermedades Infecciosas de la Provincia de Cádiz". Eur J Clin Microbiol Infect Dis. 13 (7): 559–64. PMID 7805683.
- ↑ Chiu J, Nussbaum J, Bozzette S, Tilles JG, Young LS, Leedom J; et al. (1990). "Treatment of disseminated Mycobacterium avium complex infection in AIDS with amikacin, ethambutol, rifampin, and ciprofloxacin. California Collaborative Treatment Group". Ann Intern Med. 113 (5): 358–61. PMID 2382918.
- ↑ Welsh O, Sauceda E, Gonzalez J, Ocampo J (1987). "Amikacin alone and in combination with trimethoprim-sulfamethoxazole in the treatment of actinomycotic mycetoma". J Am Acad Dermatol. 17 (3): 443–8. PMID 3308980.
- ↑ "CDC Guideline for treatment of Tuberculosis".
- ↑ Galloway G, Ramsay A, Jordan K, Vivian A (2002). "Macular infarction after intravitreal amikacin: mounting evidence against amikacin". Br J Ophthalmol. 86 (3): 359–60. PMC 1771055. PMID 11864905.
- ↑ Verma L, Arora R, Sachdev MS (1993). "Macular infarction after intravitreal injection of amikacin". Can J Ophthalmol. 28 (5): 241–3. PMID 8221375.
{{#subobject:
|Label Page=Amikacin sulfate |Label Name=500y2.png
}}
{{#subobject:
|Label Page=Amikacin sulfate |Label Name=500y2b.png
}}
{{#subobject:
|Label Page=Amikacin sulfate |Label Name=1y4.png
}}
{{#subobject:
|Label Page=Amikacin sulfate |Label Name=1y4b.png
}}