Midazolam (injection)
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]; Associate Editor(s)-in-Chief: Chetan Lokhande, M.B.B.S [2]
WikiDoc MAKES NO GUARANTEE OF VALIDITY. WikiDoc is not a professional health care provider, nor is it a suitable replacement for a licensed healthcare provider. WikiDoc is intended to be an educational tool, not a tool for any form of healthcare delivery. The educational content on WikiDoc drug pages is based upon the FDA package insert, National Library of Medicine content and practice guidelines / consensus statements. WikiDoc does not promote the administration of any medication or device that is not consistent with its labeling. Please read our full disclaimer here.
Black Box Warning
|
Boxed warning
See full prescribing information for complete Boxed Warning.
Adults and Pediatric
Neonates
|
Overview
Midazolam (injection) is a general anesthetic that is FDA approved for the {{{indicationType}}} of anxiety - induction of amnesia - preoperative sedation, induction of general anesthesia, procedural sedation, sedation for a mechanically ventilated patient.. There is a Black Box Warning for this drug as shown here. Common adverse reactions include neurologic: excessive somnolence] (1.6%), [[]headache]] (1.3% to 1.5%), somnolence(1.2%), respiratory: hiccoughs (3.6% ).
Adult Indications and Dosage
FDA-Labeled Indications and Dosage (Adult)
- Anxiety good-risk patients less than 60 years of age, 0.07 to 0.08 mg/kg IM (approximately 5 mg) up to 1 hour before surgery.
- Anxiety - Induction of amnesia - Preoperative sedation: patients 60 years or older, patients with COPD, other higher-risk surgical patients, and patients who have received concomitant narcotics or other CNS depressants, 0.02 to 0.05 mg/kg IM (approximately 2 to 3 mg) 1 hour before surgery.
- Anxiety - Induction of amnesia - older patients when the anticipated intensity and duration of sedation is less critical, 1 mg IM 1 hour before surgery.
- Anxiety - Induction of amnesia - Preoperative sedation: healthy adults less than 60 years of age, initial, 1 to 2.5 mg IV over at least 2 minutes, may titrate using small increments to desired level of sedation (allowing 2 additional minutes to evaluate full effect after each increment); reduce dose by 30% with narcotic premedication or other CNS depressants (MAX total dose: 5 mg).
- Anxiety - Induction of amnesia - Preoperative sedation: patients 60 years or older, and debilitated or chronically ill patients, initial, 1 to 1.5 mg IV over at least 2 minutes, may titrate by no more than 1-mg increments (allowing 2 additional minutes to evaluate full effect after each increment); reduce dose by 50% with narcotic premedication or other CNS depressants (max total dose: 3.5 mg).
- Anxiety - Induction of amnesia - Preoperative sedation: maintenance, additional doses of 25% of total initial dose required to achieved desired sedation may be administered IV if additional sedation is required.
- Induction of general anesthesia: unpremedicated patients less than 55 years of age, initial, 0.3 to 0.35 mg/kg IV over 20 to 30 seconds; may increase by 25% of initial dose to desired effect (allowing 2 minutes for effect) to max total dose of 0.6 mg/kg.
- Induction of general anesthesia: unpremedicated patients greater than 55 years of age, initial, 0.3 mg/kg IV over 20 to 30 seconds.
- Induction of general anesthesia: unpremedicated patients with severe systemic disease or other debilitation, initial, 0.15 to 0.25 mg/kg IV over 20 to 30 seconds.
- Induction of general anesthesia: premedicated patients less than 55 years of age, initial, 0.25 mg/kg IV over 20 to 30 seconds.
- Induction of general anesthesia: premedicated good-risk patients greater than 55 years of age, initial, 0.2 mg/kg IV over 20 to 30 seconds.
- Induction of general anesthesia: premedicated patients with severe systemic disease or debilitation, initial, 0.15 mg/kg IV over 20 to 30 seconds.
- Induction of general anesthesia: maintenance dose, incremental injections of approximately 25% of the induction dose should be given in response to signs of lightening of anesthesia .and repeated as necessary.
- Procedural sedation: healthy adults below 60 years of age, initial, 1 to 2.5 mg IV over at least 2 minutes, may titrate using small increments to desired level of sedation (allowing 2 additional minutes to evaluate full effect after each increment); reduce dose by 30% with narcotic premedication or other CNS depressants (max total dose: 5 mg).
- Procedural sedation: patients 60 years or older, and debilitated or chronically ill patients, initial, 1 to 1.5 mg IV over at least 2 minutes, may titrate by no more than 1-mg increments (allowing 2 additional minutes to evaluate full effect after each increment); reduce dose by 50% with narcotic premedication or other CNS depressants (max total dose: 3.5 mg).
- Procedural sedation: maintenance, additional doses of 25% of total initial dose required to achieved desired sedation may be administered IV if additional sedation is required.
- Sedation for a mechanically ventilated patient: loading dose, 0.01 to 0.05 mg/kg IV administered over several minutes; may repeat at 10- to 15-minute intervals until adequate sedation is achieved.
- Sedation for a mechanically ventilated patient: maintenance, continuous IV infusion initiated at a rate of 0.02 to 0.1 mg/kg/hr; may adjust rate up or down by 25% to 50% of initial infusion rate to achieve adequate sedation.
Off-Label Use and Dosage (Adult)
Guideline-Supported Use
Non–Guideline-Supported Use
There is limited information about Off-Label Non–Guideline-Supported Use of Midazolam in adult patients.
Pediatric Indications and Dosage
FDA-Labeled Indications and Dosage (Pediatric)
- High amounts of benzyl alcohol have been associated with toxicity, particularly in neonates; consider the daily metabolic load of benzyl alcohol being given.
- Anxiety - Induction of amnesia - Preoperative sedation: (6 months or older) 0.25 to 0.5 mg/kg orally as a single dose; up to 1 mg/kg for younger (6 months to less than 6 years) or uncooperative patients (max dose: 20 mg).
- Anxiety - Induction of amnesia - Preoperative sedation: 0.1 to 0.15 mg/kg IM as a single dose; 0.5 mg/kg for more anxious patients (max total dose: 10 mg).
- Anxiety - Induction of amnesia - Preoperative sedation: (6 months to 5 years of age) initial, 0.05 to 0.1 mg/kg IV over 2 to 3 minutes; may titrate to desired level of sedation (allowing 2 to 3 additional minutes to evaluate full effect after each increment); a total dose up to 0.6 mg/kg may be necessary (max total dose: 6 mg).
- Anxiety - Induction of amnesia - Preoperative sedation: (6 to 12 years of age) initial, 0.025 to 0.05 mg/kg IV over 2 to 3 minutes, may titrate to desired level of sedation (allowing 2 to 3 additional minutes to evaluate full effect after each increment); a total dose up to 0.4 mg/kg may be necessary (MAX total dose: 10 mg).
- Anxiety - Induction of amnesia - Preoperative sedation: (12 years or older) initial, 1 to 2.5 mg IV over at least 2 minutes, may titrate using small increments to desired level of sedation (allowing 2 additional minutes to evaluate full effect after each increment) (max total dose: 10 mg).
- Procedural sedation: (6 months or older) 0.25 to 0.5 mg/kg orally as a single dose; up to 1 mg/kg for younger (6 months to less than 6 years) or uncooperative patients (max dose: 20 mg).
- Procedural sedation: 0.1 to 0.15 mg/kg IM as a single dose; 0.5 mg/kg for more anxious patients (MAX total dose: 10 mg).
- Procedural sedation: (6 months to 5 years of age) initial, 0.05 to 0.1 mg/kg IV over 2 to 3 minutes; may titrate to desired level of sedation (allowing 2 to 3 additional minutes to evaluate full effect after each increment); a total dose up to 0.6 mg/kg may be necessary (MAX total dose: 6 mg).
- Procedural sedation: (6 to 12 years of age) initial, 0.025 to 0.05 mg/kg IV over 2 to 3 minutes, may titrate to desired level of sedation (allowing 2 to 3 additional minutes to evaluate full effect after each increment); a total dose up to 0.4 mg/kg may be necessary (MAX total dose 10 mg).
- Procedural sedation: (12 years or older) initial, 1 to 2.5 mg IV over at least 2 minutes, may titrate using small increments to desired level of sedation (allowing 2 additional minutes to evaluate full effect after each increment) (max total dose: 10 mg).
- Sedation for a mechanically ventilated patient: (non-neonatal) loading dose, 0.05 to 0.2 mg/kg IV over at least 2 to 3 minutes.
- Sedation for a mechanically ventilated patient: (non-neonatal), maintenance, continuous IV infusion initiated at a rate of 0.06 to 0.12 mg/kg/hr (1 to 2 mcg/kg/min), may adjust rate up or down by 25% of initial or subsequent rate to achieve adequate sedation.
- Sedation for a mechanically ventilated patient: (neonatal younger than 32 weeks) continuous IV infusion initiated at a rate of 0.03 mg/kg/hr (0.5 mcg/kg/min).
- Sedation for a mechanically ventilated patient: (neonatal older than 32 weeks) continuous IV infusion initiated at a rate of 0.06 mg/kg/hr (1 mcg/kg/min).
Off-Label Use and Dosage (Pediatric)
Guideline-Supported Use
There is limited information about Off-Label Guideline-Supported Use of Midazolam in pediatric patients.
Non–Guideline-Supported Use
There is limited information about Off-Label Non–Guideline-Supported Use of Midazolam in pediatric patients.
Contraindications
- Injectable midazolam hydrochloride is contraindicated in patients with a known hypersensitivity to the drug.
- Benzodiazepines are contraindicated in patients with acute narrow-angle glaucoma. Benzodiazepines may be used in patients with open-angle glaucoma only if they are receiving appropriate therapy. Measurements of intraocular pressure in patients without eye disease show a moderate lowering following induction with midazolam hydrochloride; patients with glaucoma have not been studied.
Warnings
|
Boxed warning
See full prescribing information for complete Boxed Warning.
Adults and Pediatric
Neonates
|
- Midazolam hydrochloride must never be used without individualization of dosage particularly when used with other medications capable of producing central nervous system depression. Prior to the intravenous administration of midazolam hydrochloride in any dose, the immediate availability of oxygen, resuscitative drugs, age- and size-appropriate equipment for bag/valve/mask ventilation and intubation, and skilled personnel for the maintenance of a patent airway and support of ventilation should be ensured. Patients should be continuously monitored with some means of detection for early signs of hypoventilation, airway obstruction, or apnea, i.e., pulse oximetry. Hypoventilation, airway obstruction, and apnea can lead to hypoxia and/or cardiac arrest unless effective countermeasures are taken immediately. The immediate availability of specific reversal agents (flumazenil) is highly recommended. Vital signs should continue to be monitored during the recovery period. Because intravenous midazolam depresses respiration (see Clinical pharmacology) and because opioid agonists and other sedatives can add to this depression, midazolam should be administered as an induction agent only by a person trained in general anesthesia and should be used for sedation/anxiolysis/amnesia only in the presence of personnel skilled in early detection of hypoventilation, maintaining a patent airway and supporting ventilation. When used for sedation/anxiolysis/amnesia, midazolam should always be titrated slowly in adult or pediatric patients. Adverse hemodynamic events have been reported in pediatric patients with cardiovascular instability; rapid intravenous administration should also be avoided in this population. See Dosage and administration for complete information.
- Serious cardiorespiratory adverse events have occurred after administration of midazolam. These have included respiratory depression, airway obstruction, oxygen desaturation, apnea, respiratory arrest and/or cardiac arrest, sometimes resulting in death or permanent neurologic injury. There have also been rare reports of hypotensive episodes requiring treatment during or after diagnostic or surgical manipulations particularly in adult or pediatric patients with hemodynamic instability. Hypotension occurred more frequently in the sedation studies in patients premedicated with a narcotic.
- Reactions such as agitation, involuntary movements (including tonic/clonic movements and muscle tremor), hyperactivity and combativeness have been reported in both adult and pediatric patients. These reactions may be due to inadequate or excessive dosing or improper administration of midazolam hydrochloride; however, consideration should be given to the possibility of cerebral hypoxia or true paradoxical reactions. Should such reactions occur, the response to each dose of midazolam hydrochloride and all other drugs, including local anesthetics, should be evaluated before proceeding. Reversal of such responses with flumazenil has been reported in pediatric patients.
- Concomitant use of barbiturates, alcohol or other central nervous system depressants may increase the risk of hypoventilation, airway obstruction, desaturation, or apnea and may contribute to profound and/or prolonged drug effect. Narcotic premedication also depresses the ventilatory response to carbon dioxide stimulation.
- Higher risk adult and pediatric surgical patients, elderly patients and debilitated adult and pediatric patients require lower dosages, whether or not concomitant sedating medications have been administered. Adult or pediatric patients with COPD are unusually sensitive to the respiratory depressant effect of midazolam hydrochloride. Pediatric and adult patients undergoing procedures involving the upper airway such as upper endoscopy or dental care, are particularly vulnerable to episodes of desaturation and hypoventilation due to partial airway obstruction. Adult and pediatric patients with chronic renal failure and patients with congestive heart failure eliminate midazolam more slowly (see Clinical pharmacology). Because elderly patients frequently have inefficient function of one or more organ systems and because dosage requirements have been shown to decrease with age, reduced initial dosage of midazolam hydrochloride is recommended and the possibility of profound and/or prolonged effect should be considered.
- Injectable midazolam should not be administered to adult or pediatric patients in shock or coma, or in acute alcohol intoxication with depression of vital signs. Particular care should be exercised in the use of intravenous midazolam in adult or pediatric patients with uncompensated acute illnesses, such as severe fluid or electrolyte disturbances.
- There have been limited reports of intra-arterial injection of midazolam hydrochloride. Adverse events have included local reactions, as well as isolated reports of seizure activity in which no clear causal relationship was established. Precautions against unintended intra-arterial injection should be taken. Extravasation should also be avoided.
- The safety and efficacy of midazolam following nonintravenous and nonintramuscular routes of administration have not been established. Midazolam hydrochloride should only be administered intramuscularly or intravenously.
- The decision as to when patients who have received injectable midazolam, particularly on an outpatient basis, may again engage in activities requiring complete mental alertness, operate hazardous machinery or drive a motor vehicle must be individualized. Gross tests of recovery from the effects of midazolam (see Clinical pharmacology) cannot be relied upon to predict reaction time under stress. It is recommended that no patient operate hazardous machinery or a motor vehicle until the effects of the drug, such as drowsiness, have subsided or until one full day after anesthesia and surgery, whichever is longer. For pediatric patients, particular care should be taken to assure safe ambulation.
Usage in Pregnancy
- An increased risk of congenital malformations associated with the use of benzodiazepine drugs (diazepam and chlordiazepoxide) has been suggested in several studies. If this drug is used during pregnancy, the patient should be apprised of the potential hazard to the fetus.
- Withdrawal symptoms of the barbiturate type have occurred after the discontinuation of benzodiazepines (see Drug abuse and dependence section).
Usage In Preterm Infants And Neonates
- Rapid injection should be avoided in the neonatal population. Midazolam hydrochloride administered rapidly as an intravenous injection (less than 2 minutes) has been associated with severe hypotension in neonates, particularly when the patient has also received fentanyl. Likewise, severe hypotension has been observed in neonates receiving a continuous infusion of midazolam who then receive a rapid intravenous injection of fentanyl. Seizures have been reported in several neonates following rapid intravenous administration.
- The neonate also has reduced and/or immature organ function and is also vulnerable to profound and/or prolonged respiratory effects of midazolam.
Adverse Reactions
Clinical Trials Experience
- See Warnings concerning serious cardiorespiratory events and possible paradoxical reactions. Fluctuations in vital signs were the most frequently seen findings following parenteral administration of midazolam in adults and included decreased tidal volume and/or respiratory rate decrease (23.3% of patients following IV and 10.8% of patients following IM administration) and apnea (15.4% of patients following IV administration), as well as variations in blood pressure and pulse rate. The majority of serious adverse effects, particularly those associated with oxygenation and ventilation, have been reported when midazolam hydrochloride is administered with other medications capable of depressing the central nervous system. The incidence of such events is higher in patients undergoing procedures involving the airway without the protective effect of an endotracheal tube (e.g., upper endoscopy and procedures).
Adults
- The following additional adverse reactions were reported after intramuscular administration:

- Administration of IM midazolam hydrochloride to elderly and/or higher risk surgical patients has been associated with rare reports of death under circumstances compatible with cardiorespiratory depression. In most of these cases, the patients also received other central nervous system depressants capable of depressing respiration, especially narcotics (see Dosage and administration).
- The following additional adverse reactions were reported subsequent to intravenous administration as a single sedative/anxiolytic/amnestic agent in adult patients:
Pediatric Patients
- The following adverse events related to the use of IV midazolam hydrochloride in pediatric patients were reported in the medical literature:desaturation 4.6%, apnea 2.8%, hypotension 2.7%, paradoxical reactions 2.0%, hiccough 1.2%, seizure-like activity 1.1% and nystagmus 1.1%. The majority of airway-related events occurred in patients receiving other CNS depressing medications and in patients where midazolam was not used as a single sedating agent.
Neonates
- For information concerning hypotensive episodes and seizures following the administration of midazolam hydrochloride to neonates, see Box Warning, Contraindications, Warnings and Precautions.
- Other adverse experiences, observed mainly following IV injection as a single sedative/anxiolytic/amnesia agent and occurring at an incidence of <1.0% in adult and pediatric patients, are as follows:
Respiratory
- Laryngospasm, bronchospasm, dyspnea, hyperventilation, wheezing, shallow respirations, airway obstruction, tachypnea
Cardiovascular
- Bigeminy, premature ventricular contractions, vasovagal episode, bradycardia, tachycardia, nodal rhythm
Gastrointestinal
- Acid taste, excessive salivation, retching
CNS/Neuromuscular
- Retrograde amnesia, euphoria, hallucination, confusion, argumentativeness, nervousness, anxiety, grogginess, restlessness, emergence delirium or agitation, prolonged emergence from anesthesia, dreaming during emergence, sleep disturbance, insomnia, nightmares, athetoid movements, seizure-like activity, ataxia, dizziness, dysphonia, slurred speech, dysphoria, paresthesia
Special senses
- Blurred vision, diplopia, nystagmus, pinpoint pupils, cyclic movements of eyelids, visual disturbance, difficulty focusing eyes, ears blocked, loss of balance, light-headedness
Integumentary
- Hive-like elevation at injection site, swelling or feeling of burning, warmth or coldness at injection site
Hypersensitivity
- Allergic reactions including anaphylactoid reactions, hives, rash, pruritus
Miscellaneous

Postmarketing Experience
There is limited information regarding Midazolam (injection) Postmarketing Experience in the drug label.
Drug Interactions
- The sedative effect of intravenous midazolam is accentuated by any concomitantly administered medication, which depresses the central nervous system, particularly narcotics (e.g., morphine, meperidine and fentanyl) and also secobarbital and droperidol. Consequently, the dosage of midazolam should be adjusted according to the type and amount of concomitant medications administered and the desired clinical response (see Dosage and administration).
- Caution is advised when midazolam is administered concomitantly with drugs that are known to inhibit the P450 3A4 enzyme system such as cimetidine (not ranitidine), erythromycin, diltiazem, verapamil, ketoconazole and itraconazole. These drug interactions may result in prolonged sedation due to a decrease in plasma clearance of midazolam.
- The effect of single oral doses of 800 mg cimetidine and 300 mg ranitidine on steady-state concentrations of midazolam was examined in a randomized crossover study (n=8). Cimetidine increased the mean midazolam steady-state concentration from 57 to 71 ng/mL. Ranitidine increased the mean steady-state concentration to 62 ng/mL. No change in choice reaction time or sedation index was detected after dosing with the H2 receptor antagonists.
- In a placebo-controlled study, erythromycin administered as a 500 mg dose, tid, for 1 week (n=6), reduced the clearance of midazolam following a single 0.5 mg/kg IV dose. The half-life was approximately doubled.
- Caution is advised when midazolam is administered to patients receiving erythromycin since this may result in a decrease in the plasma clearance of midazolam.
- The effects of diltiazem (60 mg tid) and verapamil (80 mg tid) on the pharmacokinetics and pharmacodynamics of midazolam were investigated in a three-way cross-over study (n=9). The half-life of midazolam increased from 5 to 7 hours when midazolam was taken in conjunction with verapamil or diltiazem. No interaction was observed in healthy subjects between midazolam and nifedipine.
- In a placebo-controlled study, saquinavir administered as a 1200 mg dose, tid, for 5 days (n=12), a 56% reduction in the clearance of midazolam following a single 0.05 mg/kg IV dose was observed. The half-life was approximately doubled.
- A moderate reduction in induction dosage requirements of thiopental (about 15%) has been noted following use of intramuscular midazolam hydrochloride for premedication in adults.
- The intravenous administration of midazolam hydrochloride decreases the minimum alveolar concentration (MAC) of halothane required for general anesthesia. This decrease correlates with the dose of midazolam hydrochloride administered; no similar studies have been carried out in pediatric patients but there is no scientific reason to expect that pediatric patients would respond differently than adults. Although the possibility of minor interactive effects has not been fully studied, midazolam and pancuronium have been used together in patients without noting clinically significant changes in dosage, onset or duration in adults. Midazolam hydrochloride does not protect against the characteristic circulatory changes noted after administration of succinylcholine or pancuronium and does not protect against the increased intracranial pressure noted following administration of succinylcholine. Midazolam does not cause a clinically significant change in dosage, onset or duration of a single intubating dose of succinylcholine; no similar studies have been carried out in pediatric patients but there is no scientific reason to expect that pediatric patients would respond differently than adults.
- No significant adverse interactions with commonly used premedications or drugs used during anesthesia and surgery (including atropine, scopolamine, glycopyrrolate, diazepam, hydroxyzine, d-tubocurarine, succinylcholine and other nondepolarizing muscle relaxants) or topical local anesthetics (including lidocaine, dyclonine HCl and Cetacaine) have been observed in adults or pediatric patients. In neonates, however, severe hypotension has been reported with concomitant administration of fentanyl. This effect has been observed in neonates on an infusion of midazolam who received a rapid injection of fentanyl and in patients on an infusion of fentanyl who have received a rapid injection of midazolam.
Use in Specific Populations
Pregnancy
- Segment II teratology studies, performed with midazolam maleate injectable in rabbits and rats at 5 and 10 times the human dose of 0.35 mg/kg, did not show evidence of teratogenicity.
- Nonteratogenic Effects: Studies in rats showed no adverse effects on reproductive parameters during gestation and lactation. Dosages tested were approximately 10 times the human dose of 0.35 mg/kg.
Pregnancy Category (AUS):
There is no Australian Drug Evaluation Committee (ADEC) guidance on usage of Midazolam (injection) in women who are pregnant.
Labor and Delivery
- In humans, measurable levels of midazolam were found in maternal venous serum, umbilical venous and arterial serum and amniotic fluid, indicating placental transfer of the drug. Following intramuscular administration of 0.05 mg/kg of midazolam, both the venous and the umbilical arterial serum concentrations were lower than maternal concentrations.
- The use of injectable midazolam in obstetrics has not been evaluated in clinical studies. Because midazolam is transferred transplacentally and because other benzodiazepines given in the last weeks of pregnancy have resulted in neonatal CNS depression, midazolam is not recommended for obstetrical use.
Nursing Mothers
- Midazolam is excreted in human milk. Caution should be exercised when midazolam hydrochloride is administered to a nursing woman.
Pediatric Use
- The safety and efficacy of midazolam for sedation/anxiolysis/amnesia following single dose intramuscular administration, intravenously by intermittent injections and continuous infusion have been established in pediatric and neonatal patients. For specific safety monitoring and dosage guidelines see Box warning, clinical pharmacology, indications and usage, warnings, precautions, adverse reactions, overdosage and dosage and administration sections. Unlike adult patients, pediatric patients generally receive increments of midazolam on a mg/kg basis. As a group, pediatric patients generally require higher dosages of midazolam (mg/kg) than do adults. Younger (less than six years) pediatric patients may require higher dosages (mg/kg) than older pediatric patients, and may require closer monitoring. In obese Pediatric Patients, the dose should be calculated based on ideal body weight. When midazolam is given in conjunction with opioids or other sedatives, the potential for respiratory depression, airway obstruction, or hypoventilation is increased. The health care practitioner who uses this medication in pediatric patients should be aware of and follow accepted professional guidelines for pediatric sedation appropriate to their situation.
- Midazolam hydrochloride should not be administered by rapid injection in the neonatal population. Severe hypotension and seizures have been reported following rapid IV administration, particularly with concomitant use of fentanyl.
Geriatic Use
- Because geriatric patients may have altered drug distribution and diminished hepatic and/or renal function, reduced doses of midazolam are recommended. Intravenous and intramuscular doses of midazolam should be decreased for elderly and for debilitated patients (see Warnings and dosage and administration) and subjects over 70 years of age may be particularly sensitive. These patients will also probably take longer to recover completely after midazolam administration for the induction of anesthesia. Administration of IM and IV midazolam to elderly and/or high risk surgical patients has been associated with rare reports of death under circumstances compatible with cardiorespiratory depression. In most of these cases, the patients also received other central nervous system depressants capable of depressing respiration, especially narcotics (see Dosage and administration).
- Specific dosing and monitoring guidelines for geriatric patients are provided in the Dosage and administration section for premedicated patients for sedation/anxiolysis/amnesia following IV and IM administration for induction of anesthesia following IV administration and for continuous infusion.
Gender
There is no FDA guidance on the use of Midazolam (injection) with respect to specific gender populations.
Race
There is no FDA guidance on the use of Midazolam (injection) with respect to specific racial populations.
Renal Impairment
There is no FDA guidance on the use of Midazolam (injection) in patients with renal impairment.
Hepatic Impairment
There is no FDA guidance on the use of Midazolam (injection) in patients with hepatic impairment.
Females of Reproductive Potential and Males
There is no FDA guidance on the use of Midazolam (injection) in women of reproductive potentials and males.
Immunocompromised Patients
There is no FDA guidance one the use of Midazolam (injection) in patients who are immunocompromised.
Administration and Monitoring
Administration
There is limited information regarding Midazolam (injection) Administration in the drug label.
Monitoring
There is limited information regarding Midazolam (injection) Monitoring in the drug label.
IV Compatibility
There is limited information regarding the compatibility of Midazolam (injection) and IV administrations.
Overdosage
- The manifestations of midazolam overdosage reported are similar to those observed with other benzodiazepines, including sedation, somnolence, confusion, impaired coordination, diminished reflexes, coma and untoward effects on vital signs. No evidence of specific organ toxicity from midazolam hydrochloride overdosage has been reported.
Treatment of Overdosage
- Treatment of injectable midazolam overdosage is the same as that followed for overdosage with other benzodiazepines. Respiration, pulse rate and blood pressure should be monitored and general supportive measures should be employed. Attention should be given to the maintenance of a patent airway and support of ventilation, including administration of oxygen. An intravenous infusion should be started. Should hypotension develop, treatment may include intravenous fluid therapy, repositioning, judicious use of vasopressors appropriate to the clinical situation, if indicated, and other appropriate countermeasures. There is no information as to whether peritoneal dialysis, [[forced diuresis
]] or hemodialysis are of any value in the treatment of midazolam overdose.
- Flumazenil, a specific benzodiazepine-receptor antagonist, is indicated for the complete or partial reversal of the sedative effects of benzodiazepines and may be used in situations when an overdose with a benzodiazepine is known or suspected. There are anecdotal reports of reversal of adverse hemodynamic responses associated with midazolam hydrochloride following administration of flumazenil to pediatric patients. Prior to the administration of flumazenil, necessary measures should be instituted to secure the airway, assure adequate ventilation, and establish adequate intravenous access. Flumazenil is intended as an adjunct to, not as a substitute for, proper management of benzodiazepine overdose. Patients treated with flumazenil should be monitored for resedation, respiratory depression and other residual benzodiazepine effects for an appropriate period after treatment. Flumazenil will only reverse benzodiazepines-induced effects but will not reverse the effects of other concomitant medications. The reversal of benzodiazepine effects may be associated with the onset of seizures in certain high-risk patients. The prescriber should be aware of a risk of seizure in association with flumazenil treatment, particularly in long-term benzodiazepine users and in cyclic antidepressant overdose. The complete flumazenil package insert, including Contraindications, warnings and precautions, should be consulted prior to use.
Pharmacology
| |
| |
Midazolam (injection)
| |
| Systematic (IUPAC) name | |
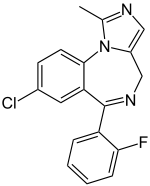
| 8-chloro-6-(2-fluorophenyl)-1-methyl-4H-imidazo[1,5-a][1,4]benzodiazepine | |
| Identifiers | |
| CAS number | |
| ATC code | N05 |
| PubChem | |
| DrugBank | |
| Chemical data | |
| Formula | Template:OrganicBox atomTemplate:OrganicBox atomTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBox atomTemplate:OrganicBoxTemplate:OrganicBox atomTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBox atomTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBox |
| Mol. mass | 325.78 |
| SMILES | & |
| Pharmacokinetic data | |
| Bioavailability | Oral ~36% I.M. 90%+ |
| Protein binding | 97% |
| Metabolism | Hepatic 3A3, 3A4, 3A5 |
| Half life | 1.8-6.4 hours |
| Excretion | Renal |
| Therapeutic considerations | |
| Pregnancy cat. | |
| Legal status | |
| Routes | Oral, I.M., I.V., parenteral |
Mechanism of Action
There is limited information regarding Midazolam (injection) Mechanism of Action in the drug label.
Structure
- Midazolam hydrochloride is a water-soluble benzodiazepine available as a sterile, nonpyrogenic parenteral dosage form for intravenous or intramuscular injection use only. Each mL contains midazolam hydrochloride equivalent to 1 mg midazolam compounded with 0.8% sodium chloride and 0.01% edetate disodium, and sodium hydroxide and/or hydrochloric acid for pH adjustment (pH 2.9 to 3.5).
- Midazolam is a white or yellowish crystalline powder, insoluble in water. The hydrochloride salt of midazolam, which is formed in situ, is soluble in aqueous solutions. Chemically, midazolam HCl is 8-chloro-6-(2-fluorophenyl)-1-methyl-4H-imidazo[1,5-a][1,4] benzodiazepine hydrochloride. Midazolam hydrochloride has the molecular formula C18H13ClFN3•HCl, a calculated molecular weight of 362.25 and the following structural formula:

- Under the acidic conditions required to solubilize midazolam in the product, midazolam is present as an equilibrium mixture (shown below) of the closed-ring form and an open-ring structure formed by the acid-catalyzed ring opening of the 4,5-double bond of the diazepine ring. The amount of open-ring form is dependent upon the pH of the solution. At the specified pH of the product, the solution may contain up to about 25% of the open-ring compound. At the physiologic conditions under which the product is absorbed (pH of 5 to 8) into the systemic circulation, any open-ring form present reverts to the physiologically active, lipophilic, closed-ring form (midazolam) and is absorbed as such.

- The following chart plots the percentage of midazolam present as the open-ring form as a function of pH in aqueous solutions. As indicated in the graph, the amount of open-ring compound present in solution is sensitive to changes in pH over the pH range specified for the product: 3.0 to 4.0 for the 1 mg/mL concentration. Above pH 5, at least 99% of the mixture is present in the closed-ring form.

Pharmacodynamics
- Midazolam’s activity is primarily due to the parent drug. Elimination of the parent drug takes place via hepatic metabolism of midazolam to hydroxylated metabolites that are conjugated and excreted in the urine. Six single-dose pharmacokinetic studies involving healthy adults yield pharmacokinetic parameters for midazolam in the following ranges: volume of distribution (Vd), 1.0 to 3.1 L/kg; elimination half-life, 1.8 to 6.4 hours (mean approximately 3 hours); total clearance (Cl), 0.25 to 0.54 L/hr/kg. In a parallel group study, there was no difference in the clearance, in subjects administered 0.15 mg/kg (n=4) and 0.30 mg/kg (n=4) IV doses indicating linear kinetics. The clearance was successively reduced by approximately 30% at doses of 0.45 mg/kg (n=4) and 0.6 mg/kg (n=5) indicating non-linear kinetics in this dose range.
Absorption
- The absolute bioavailability of the intramuscular route was greater than 90% in a cross-over study in which healthy subjects (n=17) were administered a 7.5 mg IV or IM dose. The mean peak concentration (Cmax) and time to peak (Tmax) following the IM dose was 90 ng/mL (20% CV) and 0.5 hr (50% CV). Cmax for the 1-hydroxy metabolite following the IM dose was 8 ng/mL (Tmax =1.0 hr).
- Following IM administration, Cmax for midazolam and its 1-hydroxy metabolite were approximately one-half of those achieved after intravenous injection.
Distribution
- The volume of distribution (Vd) determined from six single-dose pharmacokinetic studies involving healthy adults ranged from 1.0 to 3.1 L/kg. Female gender, old age, and obesity are associated with increased values of midazolam Vd. In humans, midazolam has been shown to cross the placenta and enter into fetal circulation and has been detected in human milk and CSF (see Special Populations).
- In adults and children patients older than 1 year, midazolam is approximately 97% bound to plasma protein, principally albumin.
Metabolism
- In vitro studies with human liver microsomes indicate that the biotransformation of midazolam is mediated by cytochrome P450 3A4. This cytochrome also appears to be present in gastrointestinal tract mucosa as well as liver. Sixty to seventy percent of the biotransformation products is 1-hydroxymidazolam (also termed alpha-hydroxymidazolam) while 4-hydroxy- midazolam constitutes 5% or less. Small amounts of a dihydroxy derivative have also been detected but not quantified. The principal urinary excretion products are glucuronide conjugates of the hydroxylated derivatives.
- Drugs that inhibit the activity of the cytochrome P450 3A4 may inhibit midazolam clearance and elevate steady-state midazolam concentrations.
- Studies of the intravenous administration of 1-hydroxy-midazolam in humans suggest that 1-hydroxy-midazolam is at least as potent as the parent compound and may contribute to the net pharmacologic activity of midazolam. In vitro studies have demonstrated that the affinities of 1- and 4-hydroxy-midazolam for the benzodiazepine receptor are approximately 20% and 7% respectively, relative to midazolam.
Excretion
- Clearance of midazolam is reduced in association with old age, congestive heart failure, liver disease (cirrhosis) or conditions which diminish cardiac output and hepatic blood flow.
- The principal urinary excretion product is 1-hydroxy-midazolam in the form of a glucuronide conjugate; smaller amounts of the glucuronide conjugates of 4-hydroxy- and dihydroxy- midazolam are detected as well. The amount of midazolam excreted unchanged in the urine after a single IV dose is less than 0.5% (n=5). Following a single IV infusion in 5 healthy volunteers, 45% to 57% of the dose was excreted in the urine as 1-hydroxymethyl midazolam conjugate.
Pharmacokinetics-Continuous Infusion
- The pharmacokinetic profile of midazolam following continuous infusion, based on 282 adult subjects, has been shown to be similar to that following single-dose administration for subjects of comparable age, gender, body habitus and health status. However, midazolam can accumulate in peripheral tissues with continuous infusion. The effects of accumulation are greater after long-term infusions than after short-term infusions.The effects of accumulation can be reduced by maintaining the lowest midazolam infusion rate that produces satisfactory sedation.
- Infrequent hypotensive episodes have occurred during continuous infusion; however, neither the time to onset nor the duration of the episode appeared to be related to plasma concentrations of midazolam or alpha-hydroxy-midazolam. Further, there does not appear to be an increased chance of occurrence of a hypotensive episode with increased loading doses.
- Patients with renal impairment may have longer elimination half-lives for midazolam (see Special Populations :Renal Failure).
Special Populations
- Changes in the pharmacokinetic profile of midazolam due to drug interactions, physiological variables, etc., may result in changes in the plasma concentration time profile and pharmacological response to midazolam in these patients. For example, patients with acute renal failure appear to have a longer elimination half-life for midazolam and may experience delayed recovery (see Special Populations:Renal Failure). In other groups, the relationship between prolonged half-life and duration of effect has not been established.
Pediatrics and Neonates
- In pediatric patients aged 1 year and older, the pharmacokinetic properties following a single dose of midazolam reported in 10 separate studies of midazolam are similar to those in adults. Weight-normalized clearance is similar or higher (0.19 to 0.80 L/hr/kg) than in adults and the terminal elimination half-life (0.78 to 3.3 hours) is similar to or shorter than in adults. The pharmacokinetic properties during and following continuous intravenous infusion in pediatric patients in the operating room as an adjunct to general anesthesia and in the intensive care environment are similar to those in adults.
- In seriously ill neonates, however, the terminal elimination half-life of midazolam is substantially prolonged (6.5 to 12.0 hours) and the clearance reduced (0.07 to 0.12 L/hr/kg) compared to healthy adults or other groups of pediatric patients. It cannot be determined if these differences are due to age, immature organ function or metabolic pathways, underlying illness or debility.
Obese
- In a study comparing normals (n=20) and obese patients (n=20) the mean half-life was greater in the obese group (5.9 vs 2.3 hours). This was due to an increase of approximately 50% in the Vd corrected for total body weight. The clearance was not significantly different between groups.
Geriatric
- In three parallel group studies, the pharmacokinetics of midazolam administered IV or IM were compared in young (mean age 29, n=52) and healthy elderly subjects (mean age 73, n=53). Plasma half-life was approximately two-fold higher in the elderly. The mean Vd based on total body weight increased consistently between 15% to 100% in the elderly. The mean Cl decreased approximately 25% in the elderly in two studies and was similar to that of the younger patients in the other.
Congestive Heart Failure
- In patients suffering from congestive heart failure, there appeared to be a two-fold increase in the elimination half-life, a 25% decrease in the plasma clearance and a 40% increase in the volume of distribution of midazolam.
Hepatic Insufficiency
- Midazolam pharmacokinetics were studied after an IV single dose (0.075 mg/kg) was administered to 7 patients with biopsy proven alcoholic cirrhosis and 8 control patients. The mean half-life of midazolam increased 2.5-fold in the alcoholic patients. Clearance was reduced by 50% and the Vd increased by 20%. In another study in 21 male patients with cirrhosis, without ascites and with normal kidney function as determined by creatinine clearance, no changes in the pharmacokinetics of midazolam or 1-hydroxy-midazolam were observed when compared to healthy individuals.
Renal Failure
- Patients with renal impairment may have longer elimination half-lives for midazolam and its metabolites which may result in slower recovery.
- Midazolam and 1-hydroxy-midazolam pharmacokinetics in 6 ICU patients who developed acute renal failure (ARF) were compared with a normal renal function control group. Midazolam was administered as an infusion (5 to 15 mg/hr). Midazolam clearance was reduced (1.9 vs 2.8 mL/min/kg) and the half-life was prolonged (7.6 vs 13 hours ) in the ARF patients. The renal clearance of the 1-hydroxy-midazolam glucuronide was prolonged in the ARF group (4 vs 136 mL/min) and the half-life was prolonged (12 hours vs >25 hours). Plasma levels accumulated in all ARF patients to about ten times that of the parent drug. The relationship between accumulating metabolite levels and prolonged sedation is unclear.
- In a study of chronic renal failure patients (n=15) receiving a single IV dose, there was a two-fold increase in the clearance and volume of distribution but the half-life remained unchanged. Metabolite levels were not studied.
Plasma Concentration-Effect Relationship
- Concentration-effect relationships (after an IV dose) have been demonstrated for a variety of pharmacodynamic measures (eg, reaction time, eye movement, sedation) and are associated with extensive intersubject variability. Logistic regression analysis of sedation scores and steady-state plasma concentration indicated that at plasma concentrations greater than 100 ng/mL there was at least a 50% probability that patients would be sedated, but respond to verbal commands (sedation score = 3). At 200 ng/mL there was at least a 50% probability that patients would be asleep, but respond to glabellar tap (sedation score = 4).
Drug Interactions
- For information concerning pharmacokinetic drug interactions with midazolam, see Precautions.
Pharmacokinetics
There is limited information regarding Midazolam (injection) Pharmacokinetics in the drug label.
Nonclinical Toxicology
There is limited information regarding Midazolam (injection) Nonclinical Toxicology in the drug label.
Clinical Studies
There is limited information regarding Midazolam (injection) Clinical Studies in the drug label.
How Supplied
- Midazolam Injection, USP 1 mg/mL is supplied as follows:
- NDC 57664-633-43 2 mL fill in 2 mL single-dose vials (Carton of 10)
- Store at 20°C to 25°C (68°F to 77°F). [See USP Controlled Room Temperature.]
- Discard unused portion.
- Manufactured by:
- Gland Pharma Ltd.
- D.P. Pally, Hyderabad - 43, India.
- M.L. No.: 103/AP/RR/97/F/R
- Distributed by:
- Caraco Pharmaceutical Laboratories Ltd.
- 1150 Elijah McCoy Drive,
- Detroit, MI 48202
- Revised: May 2013
- LEA-019429-01
Storage
There is limited information regarding Midazolam (injection) Storage in the drug label.
Images
Drug Images
{{#ask: Page Name::Midazolam (injection) |?Pill Name |?Drug Name |?Pill Ingred |?Pill Imprint |?Pill Dosage |?Pill Color |?Pill Shape |?Pill Size (mm) |?Pill Scoring |?NDC |?Drug Author |format=template |template=DrugPageImages |mainlabel=- |sort=Pill Name }}
Package and Label Display Panel
{{#ask: Label Page::Midazolam (injection) |?Label Name |format=template |template=DrugLabelImages |mainlabel=- |sort=Label Page }}
Patient Counseling Information
There is limited information regarding Midazolam (injection) Patient Counseling Information in the drug label.
Precautions with Alcohol
Alcohol-Midazolam interaction has not been established. Talk to your doctor about the effects of taking alcohol with this medication.
Brand Names
There is limited information regarding Midazolam (injection) Brand Names in the drug label.
Look-Alike Drug Names
There is limited information regarding Midazolam (injection) Look-Alike Drug Names in the drug label.
Price
References
The contents of this FDA label are provided by the National Library of Medicine.
{{#subobject:
|Label Page=Midazolam (injection) |Label Name=Midazolam label.png
}}