Bevacizumab
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]; Associate Editor(s)-in-Chief: Sophia Saad, Associate Editor - WikiDoc [2]; Sree Teja Yelamanchili, MBBS [3]
WikiDoc MAKES NO GUARANTEE OF VALIDITY. WikiDoc is not a professional health care provider, nor is it a suitable replacement for a licensed healthcare provider. WikiDoc is intended to be an educational tool, not a tool for any form of healthcare delivery. The educational content on WikiDoc drug pages is based upon the FDA package insert, National Library of Medicine content and practice guidelines / consensus statements. WikiDoc does not promote the administration of any medication or device that is not consistent with its labeling. Please read our full disclaimer here.
Black Box Warning
|
WARNING: GASTROINTESTINAL PERFORATIONS, SURGERY AND WOUND HEALING COMPLICATIONS, and HEMORRHAGE
See full prescribing information for complete Boxed Warning.
Gastrointestinal Perforation: Occurs in up to 2.4% of Bevacizumab-treated patients. Discontinue Bevacizumab for gastrointestinal perforation. (5.1)
Surgery and Wound Healing Complications: Discontinue in patients with wound dehiscence. Discontinue at least 28 days prior to elective surgery. Do not initiate Bevacizumab for at least 28 days after surgery and until the surgical wound is fully healed. (5.2) Hemorrhage: Severe or fatal hemorrhage, hemoptysis, gastrointestinal bleeding, CNS hemorrhage, and vaginal bleeding are increased in Bevacizumab- treated patients. Do not administer Bevacizumab to patients with serious hemorrhage or recent hemoptysis. (5.3) |
Overview
Bevacizumab is an antineoplastic agent that is FDA approved for the treatment of metastatic colorectal cancer (mcrc),non-squamous non–small cell lung cancer (nsclc),glioblastoma, metastatic renal cell carcinoma (mrcc). There is a Black Box Warning for this drug as shown here. Common adverse reactions include hypertension, any grade, alopecia, hand-foot syndrome due to cytotoxic therapy abdominal pain, any grade, constipation, any grade, diarrhea, loss of appetite, stomatitis, taste sense altered, hemorrhage, any grade, asthenia, dizziness, headache, any grade proteinuria, any grade, dyspnea, epistaxis, upper respiratory infection.
Adult Indications and Dosage
FDA-Labeled Indications and Dosage (Adult)
Metastatic Colorectal Cancer (mCRC)
- Dosing information
- Patients should continue treatment until disease progression or unacceptable toxicity.
- 5 mg/kg or 10 mg/kg every 2 weeks when used in combination with intravenous 5-FU-based chemotherapy.
- Administer 5 mg/kg when used in combination with bolus-IFL.
- Administer 10 mg/kg when used in combination with FOLFOX4.
- Administer 5 mg/kg every 2 weeks or 7.5 mg/kg every 3 weeks when used in combination with a fluoropyrimidine-irinotecan or fluoropyrimidine-oxaliplatin based chemotherapy regimen in patients who have progressed on a first-line Bevacizumab-containing regimen.
Non-Squamous Non-Small Cell Lung Cancer (NSCLC)
- Dosing information
- 15 mg/kg every 3 weeks in combination with carboplatin and paclitaxel.
Glioblastoma
- Dosing information
- Recommended dosage: 10 mg/kg every 2 weeks.
Metastatic Renal Cell Carcinoma (mRCC)
- Dosing information
- Recommended dosage: 10 mg/kg every 2 weeks in combination with interferon alfa.
Off-Label Use and Dosage (Adult)
Guideline-Supported Use
There is limited information regarding Off-Label Guideline-Supported Use of Bevacizumab in adult patients.
Non–Guideline-Supported Use
Age related macular degeneration, Secondary to choroidal neovascularization
- Dosing information
- Administration of intravitreal bevacizumab has been associated with improved visual acuity, decreased retinal thickness
Angioid streaks of choroid
- Dosing information
- Dosing information
- Not applicable [3]
- Dosing information
- 1.25 mg/0.05 mL via intravitreal injection[4]
Cervical cancer, Recurrent, persistent, or metastatic
- Dosing information
- 1.25 mg/0.05 mL [5]
- Dosing information
- Not applicable [6]
- Dosing information
- Not applicable [7]
Epistaxis - Osler hemorrhagic telangiectasia syndrome
- Dosing information
- 100 mg nasal spray via a metered dose atomizer [8]
- Dosing information
- 7.5 mg/kg [9]
- Dosing information
- Dosing information
- Dosing information
- 1.25 mg18463512
- 1.25 mg every 4 weeks for 3 doses [12]
- Dosing information
- Dosing information
- Dosing information
Pediatric Indications and Dosage
FDA-Labeled Indications and Dosage (Pediatric)
The safety, effectiveness and pharmacokinetic profile of Bevacizumab in pediatric patients have not been established.
Off-Label Use and Dosage (Pediatric)
Guideline-Supported Use
There is limited information regarding Off-Label Guideline-Supported Use of Bevacizumab in pediatric patients.
Non–Guideline-Supported Use
There is limited information regarding Off-Label Non–Guideline-Supported Use of Bevacizumab in pediatric patients.
Contraindications
None.
Warnings
|
WARNING: GASTROINTESTINAL PERFORATIONS, SURGERY AND WOUND HEALING COMPLICATIONS, and HEMORRHAGE
See full prescribing information for complete Boxed Warning.
Gastrointestinal Perforation: Occurs in up to 2.4% of Bevacizumab-treated patients. Discontinue Bevacizumab for gastrointestinal perforation. (5.1)
Surgery and Wound Healing Complications: Discontinue in patients with wound dehiscence. Discontinue at least 28 days prior to elective surgery. Do not initiate Bevacizumab for at least 28 days after surgery and until the surgical wound is fully healed. (5.2) Hemorrhage: Severe or fatal hemorrhage, hemoptysis, gastrointestinal bleeding, CNS hemorrhage, and vaginal bleeding are increased in Bevacizumab- treated patients. Do not administer Bevacizumab to patients with serious hemorrhage or recent hemoptysis. (5.3) |
Gastrointestinal perforations
Serious and sometimes fatal gastrointestinal perforation occurs at a higher incidence in Bevacizumab treated patients compared to controls. The incidence of gastrointestinal perforation ranged from 0.3 to 2.4% across clinical studies. The typical presentation may include abdominal pain, nausea, emesis, constipation, and fever. Perforation can be complicated by intra-abdominal abscess and fistula formation. The majority of cases occurred within the first 50 days of initiation of Bevacizumab. Discontinue Bevacizumab in patients with gastrointestinal perforation.
Surgery and Wound Healing Complications
Bevacizumab impairs wound healing in animal models. In clinical trials, administration of Bevacizumab was not allowed until at least 28 days after surgery. In a controlled clinical trial, the incidence of wound healing complications, including serious and fatal complications, in patients with mCRC who underwent surgery during the course of Bevacizumab treatment was 15% and in patients who did not receive Bevacizumab, was 4%. Bevacizumab should not be initiated for at least 28 days following surgery and until the surgical wound is fully healed. Discontinue Bevacizumab in patients with wound healing complications requiring medical intervention. The appropriate interval between the last dose of Bevacizumab and elective surgery is unknown; however, the half-life of Bevacizumab is estimated to be 20 days. Suspend Bevacizumab for at least 28 days prior to elective surgery. Do not administer Bevacizumab until the wound is fully healed. Necrotizing fasciitis including fatal cases, has been reported in patients treated with Bevacizumab; usually secondary to wound healing complications, gastrointestinal perforation or fistula formation. Discontinue Bevacizumab therapy in patients who develop necrotizing fasciitis.
Hemorrhage
Bevacizumab can result in two distinct patterns of bleeding: minor hemorrhage, most commonly Grade 1 epistaxis; and serious, and in some cases fatal, hemorrhagic events. Severe or fatal hemorrhage, including hemoptysis, gastrointestinal bleeding, hematemesis, CNS hemorrhage, epistaxis, and vaginal bleeding occurred up to five-fold more frequently in patients receiving Bevacizumab compared to patients receiving only chemotherapy. Across indications, the incidence of Grade ≥ 3 hemorrhagic events among patients receiving Bevacizumab ranged from 1.2 to 4.6%. Serious or fatal pulmonary hemorrhage occurred in four of 13 (31%) patients with squamous cell histology and two of 53 (4%) patients with non-squamous non-small cell lung cancer receiving Bevacizumab and chemotherapy compared to none of the 32 (0%) patients receiving chemotherapy alone. In clinical studies in non–small cell lung cancer where patients with CNS metastases who completed radiation and surgery more than 4 weeks prior to the start of Bevacizumab were evaluated with serial CNS imaging, symptomatic Grade 2 CNS hemorrhage was documented in one of 83 Bevacizumab-treated patients (rate 1.2%, 95% CI 0.06%–5.93%). Intracranial hemorrhage occurred in 8 of 163 patients with previously treated glioblastoma; two patients had Grade 3–4 hemorrhage. Do not administer Bevacizumab to patients with recent history of hemoptysis of ≥ 1/2 teaspoon of red blood. Discontinue Bevacizumab in patients with hemorrhage. [See Boxed Warning, Dosage and Administration (2.4).]
Non-Gastrointestinal Fistula Formation
Serious and sometimes fatal non-gastrointestinal fistula formation involving tracheo-esophageal, bronchopleural, biliary, vaginal, renal and bladder sites occurs at a higher incidence in Bevacizumab-treated patients compared to controls. The incidence of non-gastrointestinal perforation was ≤ 0.3% in clinical studies. Most events occurred within the first 6 months of Bevacizumab therapy. Discontinue Bevacizumab in patients with fistula formation involving an internal organ.
Arterial Thromboembolic Events
Serious, sometimes fatal, arterial thromboembolic events (ATE)including cerebral infarction, transient ischemic attacks, myocardial infarction, angina, and a variety of other ATE occurred at a higher incidence in patients receiving Bevacizumab compared to those in the control arm. Across indications, the incidence of Grade ≥ 3 ATE in the Bevacizumab containing arms was 2.6% compared to 0.8% in the control arms. Among patients receiving Bevacizumab in combination with chemotherapy, the risk of developing ATE during therapy was increased in patients with a history of arterial thromboembolism, diabetes, or age greater than 65 years. The safety of resumption of Bevacizumab therapy after resolution of an ATE has not been studied. Discontinue Bevacizumab in patients who experience a severe ATE.
Hypertension
The incidence of severe hypertension is increased in patients receiving Bevacizumab as compared to controls. Across clinical studies the incidence of Grade 3 or 4 hypertension ranged from 5-18%. Monitor blood pressure every two to three weeks during treatment with Bevacizumab. Treat with appropriate anti-hypertensive therapy and monitor blood pressure regularly. Continue to monitor blood pressure at regular intervals in patients with Bevacizumab-induced or -exacerbated hypertension after discontinuation of Bevacizumab. Temporarily suspend Bevacizumab in patients with severe hypertension that is not controlled with medical management. Discontinue Bevacizumab in patients with hypertensive crisis or hypertensive encephalopathy.
Reversible Posterior Leukoencephalopathy Syndrome (RPLS)
RPLS has been reported with an incidence of < 0.1% in clinical studies. The onset of symptoms occurred from 16 hours to 1 year after initiation of Bevacizumab. RPLS is a neurological disorder which can present with headache, seizure, lethargy, confusion, blindness and other visual and neurologic disturbances. Mild to severe hypertension may be present. Magnetic resonance imaging (MRI) is necessary to confirm the diagnosis of RPLS. Discontinue Bevacizumab in patients developing RPLS. Symptoms usually resolve or improve within days, although some patients have experienced ongoing neurologic sequelae. The safety of reinitiating Bevacizumab therapy in patients previously experiencing RPLS is not known.
Proteinuria
The incidence and severity of proteinuria is increased in patients receiving Bevacizumab as compared to controls. Nephrotic syndrome occurred in < 1% of patients receiving Bevacizumab in clinical trials, in some instances with fatal outcome. [See Adverse Reactions (6.1).] In a published case series, kidney biopsy of six patients with proteinuria showed findings consistent with thrombotic microangiopathy. Monitor proteinuria by dipstick urine analysis for the development or worsening of proteinuria with serial urinalyses during Bevacizumab therapy. Patients with a 2 + or greater urine dipstick reading should undergo further assessment with a 24-hour urine collection. Suspend Bevacizumab administration for ≥ 2 grams of proteinuria/24 hours and resume when proteinuria is < 2 gm/24 hours. Discontinue Bevacizumab in patients with nephrotic syndrome. Data from a postmarketing safety study showed poor correlation between UPCR (Urine Protein/Creatinine Ratio) and 24 hour urine protein (Pearson Correlation 0.39 (95% CI 0.17, 0.57).
Infusion Reactions
Infusion reactions reported in the clinical trials and post-marketing experience include hypertension, hypertensive crises associated with neurologic signs and symptoms, wheezing, oxygen desaturation, Grade 3 hypersensitivity, chest pain, headaches, rigors, and diaphoresis. In clinical studies, infusion reactions with the first dose of Bevacizumab were uncommon ( < 3%) and severe reactions occurred in 0.2% of patients. Stop infusion if a severe infusion reaction occurs and administer appropriate medical therapy.
Ovarian Failure
The incidence of ovarian failure was higher (34% vs. 2%) in premenopausal women receiving Bevacizumab in combination with mFOLFOX chemotherapy as compared to those receiving mFOLFOX chemotherapy alone for adjuvant treatment for colorectal cancer, a use for which Bevacizumab is not approved. Inform females of reproductive potential of the risk of ovarian failure prior to starting treatment with Bevacizumab.
Adverse Reactions
Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice. The data below reflect exposure to Bevacizumab in 4599 patients with CRC, non-squamous NSCLC, glioblastoma, or mRCC trials including controlled (Studies 1, 2, 4, 5 and 8) or uncontrolled, single arm (Study 6) treated at the recommended dose and schedule for a median of 8 to 23 doses of Bevacizumab. [See Clinical Studies (14).] The population was aged 18-89 years (median 60 years), 45.4% male and 85.8% (3729/4345) White. The population included 2184 first- and second-line mCRC patients who received a median of 10 doses of Bevacizumab, 480 first-line metastatic NSCLC patients who received a median of 8 doses of Bevacizumab, 163 glioblastoma patients who received a median of 9 doses of Bevacizumab, and 337 mRCC patients who received a median of 16 doses of Bevacizumab. These data also reflect exposure to Bevacizumab in 363 patients with metastatic breast cancer (MBC) who received a median of 9.5 doses of Bevacizumab, 669 female adjuvant CRC patients who received a median of 23 doses of Bevacizumab and exposure to Bevacizumab in 403 previously untreated patients with diffuse large B-cell lymphoma (DLBCL) who received a median of 8 doses of Bevacizumab. Bevacizumab is not approved for use in MBC, adjuvant CRC, or DLBCL.
Surgery and Wound Healing Complications
The incidence of post-operative wound healing and/or bleeding complications was increased in patients with mCRC receiving Bevacizumab as compared to patients receiving only chemotherapy. Among patients requiring surgery on or within 60 days of receiving study treatment, wound healing and/or bleeding complications occurred in 15% (6/39) of patients receiving bolus-IFL plus Bevacizumab as compared to 4% (1/25) of patients who received bolus-IFL alone. In Study 6, events of post-operative wound healing complications (craniotomy site wound dehiscence and cerebrospinal fluid leak) occurred in patients with previously treated glioblastoma: 3/84 patients in the Bevacizumab alone arm and 1/79 patients in the Bevacizumab plus irinotecan arm.
Hemorrhage
The incidence of epistaxis was higher (35% vs. 10%) in patients with mCRC receiving bolus-IFL plus Bevacizumab compared with patients receiving bolus-IFL plus placebo. All but one of these events were Grade 1 in severity and resolved without medical intervention. Grade 1 or 2 hemorrhagic events were more frequent in patients receiving bolus-IFL plus Bevacizumab when compared to those receiving bolus-IFL plus placebo and included gastrointestinal hemorrhage (24% vs. 6%), minor gum bleeding (2% vs. 0), and vaginal hemorrhage (4% vs. 2%).
Venous Thromboembolic Events
The overall incidence of venous thromboembolic events in Study 1 was 15.1% in patients receiving bolus-IFL plus Bevacizumab and 13.6% in patients receiving bolus-IFL plus placebo. In Study 1, more patients in the Bevacizumab containing arm experienced deep venous thrombosis (34 vs. 19 patients ) and intra-abdominal venous thrombosis (10 vs. 5 patients). The risk of developing a second thromboembolic event while on Bevacizumab and oral anticoagulants was evaluated in two randomized studies. In Study 1, 53 patients (14%) on the bolus-IFL plus Bevacizumab arm and 30 patients (8%) on the bolus-IFL plus placebo arm received full dose warfarin following a venous thromboembolic event (VTE). Among these patients, an additional thromboembolic event occurred in 21% (11/53) of patients receiving bolus-IFL plus Bevacizumab and 3% (1/30) of patients receiving bolus-IFL alone. In a second, randomized, 4-arm study in 1401 patients with mCRC, prospectively evaluating the incidence of VTE (all grades), the overall incidence of first VTE was higher in the Bevacizumab containing arms (13.5%) than the chemotherapy alone arms (9.6%). Among the 116 patients treated with anticoagulants following an initial VTE event (73 in the Bevacizumab plus chemotherapy arms and 43 in the chemotherapy alone arms), the overall incidence of subsequent VTEs was also higher among the Bevacizumab treated patients (31.5% vs. 25.6%). In this subgroup of patients treated with anticoagulants, the overall incidence of bleeding, the majority of which were Grade 1, was higher in the Bevacizumab treated arms than the chemotherapy arms (27.4% vs. 20.9%).
Neutropenia and Infection
The incidences of neutropenia and febrile neutropenia are increased in patients receiving Bevacizumab plus chemotherapy compared to chemotherapy alone. In Study 1, the incidence of Grade 3 or 4 neutropenia was increased in mCRC patients receiving IFL plus Bevacizumab (21%) compared to patients receiving IFL alone (14%). In Study 5, the incidence of Grade 4 neutropenia was increased in NSCLC patients receiving paclitaxel/carboplatin (PC) plus Bevacizumab (26.2%) compared with patients receiving PC alone (17.2%). Febrile neutropenia was also increased (5.4% for PC plus Bevacizumab vs. 1.8% for PC alone). There were 19 (4.5%) infections with Grade 3 or 4 neutropenia in the PC plus Bevacizumab arm of which 3 were fatal compared to 9 (2%) neutropenic infections in patients receiving PC alone, of which none were fatal. During the first 6 cycles of treatment, the incidence of serious infections including pneumonia, febrile neutropenia, catheter infections and wound infections was increased in the PC plus Bevacizumab arm [58 patients (13.6%)] compared to the PC alone arm [29 patients (6.6%)]. In Study 6, one fatal event of neutropenic infection occurred in a patient with previously treated glioblastoma receiving Bevacizumab alone. The incidence of any grade of infection in patients receiving Bevacizumab alone was 55% and the incidence of Grade 3–5 infection was 10%.
Proteinuria
Grade 3–4 proteinuria ranged from 0.7 to 7.4% in Studies 1, 2, 4, 5 and 8. The overall incidence of proteinuria (all grades) was only adequately assessed in Study 8, in which the incidence was 20%. Median onset of proteinuria was 5.6 months (range 15 days to 37 months) after initiation of Bevacizumab. Median time to resolution was 6.1 months (95% CI 2.8 months, 11.3 months). Proteinuria did not resolve in 40% of patients after median follow up of 11.2 months and required permanent discontinuation of Bevacizumab in 30% of the patients who developed proteinuria (Study 8). In an exploratory, pooled analysis of 8,273 patients treated in 7 randomized clinical trials, 5.4% (271 of 5037) of patients receiving Bevacizumab in combination with chemotherapy experienced Grade ≥ 2 proteinuria. The Grade ≥ 2 proteinuria resolved in 74.2% (201 of 271) of patients. Bevacizumab was re-initiated in 41.7% (113 of 271) of patients. Of the 113 patients who re-initiated Bevacizumab, 47.8% (54 of 113) experienced a second episode of Grade ≥ 2 proteinuria. [See Warnings and Precautions (5.8).]
Congestive Heart Failure (CHF)
The incidence of Grade ≥ 3 left ventricular dysfunction was 1.0% in patients receiving Bevacizumab compared to 0.6% in the control arm across indications. In patients with metastatic breast cancer (MBC), an indication for which Bevacizumab is not approved, the incidence of Grade 3–4 CHF was increased in patients in the Bevacizumab plus paclitaxel arm (2.2%) as compared to the control arm (0.3%). Among patients receiving prior anthracyclines for MBC, the rate of CHF was 3.8% for patients receiving Bevacizumab as compared to 0.6% for patients receiving paclitaxel alone. The safety of continuation or resumption of Bevacizumab in patients with cardiac dysfunction has not been studied. In previously untreated patients with diffuse large B-cell lymphoma (DLBCL), an indication for which Bevacizumab is not approved, the incidence of CHF and decline in left-ventricular ejection fraction (LVEF) were signficantly increased in the Bevacizumab plus R-CHOP (rituximab, cyclophosphamide, doxorubicin, vincristine, and prednisone) arm (n=403) compared to the placebo plus R-CHOP arm (n=379); both regimens were given for 6 to 8 cycles. At the completion of R-CHOP therapy, the incidence of CHF was 10.9% in the Bevacizumab plus R-CHOP arm compared to 5.0% in the R-CHOP alone arm [relative risk (95% CI) of 2.2 (1.3, 3.7)]. The incidence of a LVEF event, defined as a decline from baseline of 20% or more in LVEF or a decline from baseline of 10% or more to a LVEF value of less than 50%, was also increased in the Bevacizumab plus R-CHOP arm (10.4%) compared to the R-CHOP alone arm (5.0%). Time to onset of left-ventricular dysfunction or CHF was 1-6 months after initiation of therapy in at least 85% of the patients and was resolved in 62% of the patients experiencing CHF in the Bevacizumab arm compared to 82% in the control arm.
Ovarian Failure
The incidence of new cases of ovarian failure (defined as amenorrhoea lasting 3 or more months, FSH level ≥ 30 mIU/mL and a negative serum β-HCG pregnancy test) was prospectively evaluated in a subset of 179 women receiving mFOLFOX chemotherapy alone (n = 84) or with Bevacizumab (n = 95). New cases of ovarian failure were identified in 34% (32/95) of women receiving Bevacizumab in combination with chemotherapy compared with 2% (2/84) of women receiving chemotherapy alone [relative risk of 14 (95% CI 4, 53)]. After discontinuation of Bevacizumab treatment, recovery of ovarian function at all time points during the post-treatment period was demonstrated in 22% (7/32) of the Bevacizumab-treated women. Recovery of ovarian function is defined as resumption of menses, a positive serum β-HCG pregnancy test, or a FSH level < 30 mIU/mL during the post-treatment period. Long term effects of Bevacizumab exposure on fertility are unknown. [See Warnings and Precautions (5.10), Use in Specific Populations (8.6).]
Metastatic Colorectal Cancer (mCRC)
The data in Table 1 and Table 2 were obtained in Study 1, a randomized, double-blind, controlled trial comparing chemotherapy plus Bevacizumab with chemotherapy plus placebo. Bevacizumab was administered at 5 mg/kg every 2 weeks. All Grade 3–4 adverse events and selected Grade 1–2 adverse events (hypertension, proteinuria, thromboembolic events) were collected in the entire study population. Severe and life-threatening (Grade 3–4) adverse events, which occurred at a higher incidence ( ≥ 2%) in patients receiving bolus-IFL plus Bevacizumab as compared to bolus-IFL plus placebo, are presented in Table 1.
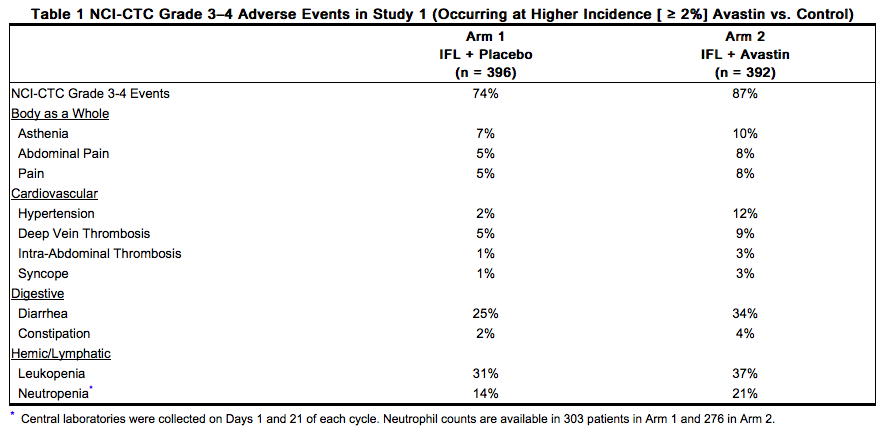
Grade 1–4 adverse events which occurred at a higher incidence ( ≥ 5%) in patients receiving bolus-IFL plus Bevacizumab as compared to the bolus-IFL plus placebo arm are presented in Table 2. Grade 1–4 adverse events were collected for the first approximately 100 patients in each of the three treatment arms who were enrolled until enrollment in Arm 3 (5-FU/LV + Bevacizumab) was discontinued.

Bevacizumab in Combination with FOLFOX4 in Second-line mCRC
Only Grade 3-5 non-hematologic and Grade 4–5 hematologic adverse events related to treatment were collected in Study 2. The most frequent adverse events (selected Grade 3–5 non-hematologic and Grade 4–5 hematologic adverse events) occurring at a higher incidence ( ≥ 2%) in 287 patients receiving FOLFOX4 plus Bevacizumab compared to 285 patients receiving FOLFOX4 alone were fatigue (19% vs. 13%), diarrhea (18% vs. 13%), sensory neuropathy (17% vs. 9%), nausea (12% vs. 5%), vomiting (11% vs. 4%), dehydration (10% vs. 5%), hypertension (9% vs. 2%), abdominal pain (8% vs. 5%), hemorrhage (5% vs. 1%), other neurological (5% vs. 3%), ileus (4% vs. 1%) and headache (3% vs. 0%). These data are likely to under-estimate the true adverse event rates due to the reporting mechanisms used in Study 2. Bevacizumab in Combination with Fluoropyrimidine-Irinotecan or Fluoropyrimidine-Oxaliplatin Based Chemotherapy in Second-line mCRC Patients who have Progressed on an Bevacizumab Containing Regimen in First-line mCRC: No new safety signals were observed in Study 4 when Bevacizumab was administered in second line mCRC patients who progressed on an Bevacizumab containing regimen in first line mCRC. The safety data was consistent with the known safety profile established in first and second line mCRC.
Unresectable Non-Squamous Non-Small Cell Lung Cancer (NSCLC)
Only Grade 3-5 non-hematologic and Grade 4-5 hematologic adverse events were collected in Study 5. Grade 3–5 non-hematologic and Grade 4–5 hematologic adverse events (occurring at a higher incidence (≥ 2%) in 427 patients receiving PC plus Bevacizumab compared with 441 patients receiving PC alone were neutropenia (27% vs. 17%), fatigue (16% vs. 13%), hypertension (8% vs. 0.7%), infection without neutropenia (7% vs. 3%), venous thrombus/embolism (5% vs. 3%), febrile neutropenia (5% vs. 2%), pneumonitis/pulmonary infiltrates (5% vs. 3%), infection with Grade 3 or 4 neutropenia (4% vs. 2%), hyponatremia (4% vs. 1%), headache (3% vs. 1%) and proteinuria (3% vs. 0%).
Glioblastoma
All adverse events were collected in 163 patients enrolled in Study 6 who either received Bevacizumab alone or Bevacizumab plus irinotecan. All patients received prior radiotherapy and temozolomide. Bevacizumab was administered at 10 mg/kg every 2 weeks alone or in combination with irinotecan. Bevacizumab was discontinued due to adverse events in 4.8% of patients treated with Bevacizumab alone. In patients receiving Bevacizumab alone (N = 84), the most frequently reported adverse events of any grade were infection (55%), fatigue (45%), headache (37%), hypertension (30%), epistaxis (19%) and diarrhea (21%). Of these, the incidence of Grade ≥ 3 adverse events was infection (10%), fatigue (4%), headache (4%), hypertension (8%) and diarrhea (1%). Two deaths on study were possibly related to Bevacizumab: one retroperitoneal hemorrhage and one neutropenic infection. In patients receiving Bevacizumab alone or Bevacizumab plus irinotecan (N = 163), the incidence of Bevacizumab-related adverse events (Grade 1–4) were bleeding/hemorrhage (40%), epistaxis (26%), CNS hemorrhage (5%), hypertension (32%), venous thromboembolic event (8%), arterial thromboembolic event (6%), wound-healing complications (6%), proteinuria (4%), gastrointestinal perforation (2%), and RPLS (1%). The incidence of Grade 3–5 events in these 163 patients were bleeding/hemorrhage (2%), CNS hemorrhage (1%), hypertension (5%), venous thromboembolic event (7%), arterial thromboembolic event (3%), wound-healing complications (3%), proteinuria (1%), and gastrointestinal perforation (2%).
Metastatic Renal Cell Carcinoma (mRCC)
All grade adverse events were collected in Study 8. Grade 3–5 adverse events occurring at a higher incidence ( ≥ 2%) in 337 patients receiving interferon alfa (IFN-α) plus Bevacizumab compared to 304 patients receiving IFN-α plus placebo arm were fatigue (13% vs. 8%), asthenia (10% vs. 7%), proteinuria (7% vs. 0%), hypertension (6% vs. 1%; including hypertension and hypertensive crisis), and hemorrhage (3% vs. 0.3%; including epistaxis, small intestinal hemorrhage, aneurysm ruptured, gastric ulcer hemorrhage, gingival bleeding, haemoptysis, hemorrhage intracranial, large intestinal hemorrhage, respiratory tract hemorrhage, and traumatic hematoma). Grade 1–5 adverse events occurring at a higher incidence ( ≥ 5%) in patients receiving IFN-α plus Bevacizumab compared to the IFN-α plus placebo arm are presented in Table 3.

The following adverse events were reported at a 5-fold greater incidence in the IFN-α plus Bevacizumab arm compared to IFN-α alone and not represented in Table 3: gingival bleeding (13 patients vs. 1 patient); rhinitis (9 vs.0 ); blurred vision (8 vs. 0); gingivitis (8 vs. 1); gastroesophageal reflux disease (8 vs.1 ); tinnitus (7 vs. 1); tooth abscess (7 vs.0); mouth ulceration (6 vs. 0); acne (5 vs. 0); deafness (5 vs. 0); gastritis (5 vs. 0); gingival pain (5 vs. 0) and pulmonary embolism (5 vs. 1).
Immunogenicity
As with all therapeutic proteins, there is a potential for an immune response to Bevacizumab. In clinical trials of adjuvant colon carcinoma, 14 of 2233 evaluable patients (0.63%) tested positive for treatment-emergent anti-bevacizumab antibodies detected by an electrochemiluminescent (ECL) based assay. Among these 14 patients, three tested positive for neutralizing antibodies against bevacizumab using an enzyme-linked immunosorbent assay (ELISA). The clinical significance of these anti-product antibody responses to bevacizumab is unknown. Immunogenicity assay results are highly dependent on the sensitivity and specificity of the test method and may be influenced by several factors, including sample handling, timing of sample collection, concomitant medications, and underlying disease. For these reasons, comparison of the incidence of antibodies to Bevacizumab with the incidence of antibodies to other products may be misleading.
Postmarketing Experience
The following adverse reactions have been identified during post-approval use of Bevacizumab. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Body as a Whole: Polyserositis Cardiovascular: Pulmonary hypertension, RPLS, Mesenteric venous occlusion Eye disorders (from unapproved intravitreal use for treatment of various ocular disorders): Permanent loss of vision; Endophthalmitis (infectious and sterile); Intraocular inflammation; Retinal detachment; Increased intraocular pressure; Hemorrhage including conjunctival, vitreous hemorrhage or retinal hemorrhage; Vitreous floaters; Ocular hyperemia; Ocular pain or discomfort Gastrointestinal: Gastrointestinal ulcer, Intestinal necrosis, Anastomotic ulceration Hemic and lymphatic: Pancytopenia Hepatobiliary disorders: Gallbladder perforation Infections and infestations: Necrotizing fasciitis, usually secondary to wound healing complications, gastrointestinal perforation or fistula formation Musculoskeletal: Osteonecrosis of the jaw Renal: Renal thrombotic microangiopathy (manifested as severe proteinuria) Respiratory: Nasal septum perforation, dysphonia Systemic Events (from unapproved intravitreal use for treatment of various ocular disorders): Arterial thromboembolic events, Hypertension, Gastrointestinal perforation, Hemorrhage
Drug Interactions
A drug interaction study was performed in which irinotecan was administered as part of the FOLFIRI regimen with or without Bevacizumab. The results demonstrated no significant effect of bevacizumab on the pharmacokinetics of irinotecan or its active metabolite SN38. In a randomized study in 99 patients with NSCLC, based on limited data, there did not appear to be a difference in the mean exposure of either carboplatin or paclitaxel when each was administered alone or in combination with Bevacizumab. However, 3 of the 8 patients receiving Bevacizumab plus paclitaxel/carboplatin had substantially lower paclitaxel exposure after four cycles of treatment (at Day 63) than those at Day 0, while patients receiving paclitaxel/carboplatin without Bevacizumab had a greater paclitaxel exposure at Day 63 than at Day 0. In Study 8, there was no difference in the mean exposure of interferon alfa administered in combination with Bevacizumab when compared to interferon alfa alone.
Use in Specific Populations
Pregnancy
Pregnancy Category (FDA): C
There are no adequate or well controlled studies of bevacizumab in pregnant women. While it is not known if bevacizumab crosses the placenta, human IgG is known to cross the placenta Reproduction studies in rabbits treated with approximately 1 to 12 times the recommended human dose of bevacizumab demonstrated teratogenicity, including an increased incidence of specific gross and skeletal fetal alterations. Adverse fetal outcomes were observed at all doses tested. Other observed effects included decreases in maternal and fetal body weights and an increased number of fetal resorptions.
Because of the observed teratogenic effects of bevacizumab in animals and of other inhibitors of angiogenesis in humans, bevacizumab should be used during pregnancy only if the potential benefit to the pregnant woman justifies the potential risk to the fetus.
Pregnancy Category (AUS):
There is no Australian Drug Evaluation Committee (ADEC) guidance on usage of Bevacizumab in women who are pregnant.
Labor and Delivery
There is no FDA guidance on use of Bevacizumab during labor and delivery.
Nursing Mothers
It is not known whether Bevacizumab is secreted in human milk. Human IgG is excreted in human milk, but published data suggest that breast milk antibodies do not enter the neonatal and infant circulation in substantial amounts. Because many drugs are secreted in human milk and because of the potential for serious adverse reactions in nursing infants from bevacizumab, a decision should be made whether to discontinue nursing or discontinue drug, taking into account the half-life of the bevacizumab (approximately 20 days [range 11–50 days]) and the importance of the drug to the mother.
Pediatric Use
The safety, effectiveness and pharmacokinetic profile of Bevacizumab in pediatric patients have not been established. Antitumor activity was not observed among eight children with relapsed glioblastoma treated with bevacizumab and irinotecan. There is insufficient information to determine the safety and efficacy of Bevacizumab in children with glioblastoma. Juvenile cynomolgus monkeys with open growth plates exhibited physeal dysplasia following 4 to 26 weeks exposure at 0.4 to 20 times the recommended human dose (based on mg/kg and exposure). The incidence and severity of physeal dysplasia were dose-related and were partially reversible upon cessation of treatment.
Geriatic Use
In Study 1, severe adverse events that occurred at a higher incidence ( ≥ 2%) in patients aged ≥ 65 years as compared to younger patients were asthenia, sepsis, deep thrombophlebitis, hypertension, hypotension, myocardial infarction, congestive heart failure, diarrhea, constipation, anorexia, leukopenia, anemia, dehydration, hypokalemia, and hyponatremia. The effect of Bevacizumab on overall survival was similar in elderly patients as compared to younger patients. In Study 2, patients aged ≥ 65 years receiving Bevacizumab plus FOLFOX4 had a greater relative risk as compared to younger patients for the following adverse events: nausea, emesis, ileus, and fatigue. In Study 5, patients aged ≥ 65 years receiving carboplatin, paclitaxel, and Bevacizumab had a greater relative risk for proteinuria as compared to younger patients Of the 742 patients enrolled in Genentech-sponsored clinical studies in which all adverse events were captured, 212 (29%) were age 65 or older and 43 (6%) were age 75 or older. Adverse events of any severity that occurred at a higher incidence in the elderly as compared to younger patients, in addition to those described above, were dyspepsia, gastrointestinal hemorrhage, edema, epistaxis, increased cough, and voice alteration. In an exploratory, pooled analysis of 1745 patients treated in five randomized, controlled studies, there were 618 (35%) patients aged ≥ 65 years and 1127 patients < 65 years of age. The overall incidence of arterial thromboembolic events was increased in all patients receiving Bevacizumab with chemotherapy as compared to those receiving chemotherapy alone, regardless of age. However, the increase in arterial thromboembolic events incidence was greater in patients aged ≥ 65 years (8.5% vs. 2.9%) as compared to those < 65 years (2.1% vs. 1.4%)
Gender
There is no FDA guidance on the use of Bevacizumab with respect to specific gender populations.
Race
There is no FDA guidance on the use of Bevacizumab with respect to specific racial populations.
Renal Impairment
There is no FDA guidance on the use of Bevacizumab in patients with renal impairment.
Hepatic Impairment
There is no FDA guidance on the use of Bevacizumab in patients with hepatic impairment.
Females of Reproductive Potential and Males
Bevacizumab increases the risk of ovarian failure and may impair fertility. Inform females of reproductive potential of the risk of ovarian failure prior to starting treatment with Bevacizumab. Long term effects of Bevacizumab exposure on fertility are unknown. In a prospectively designed substudy of 179 premenopausal women randomized to receive chemotherapy with or without Bevacizumab, the incidence of ovarian failure was higher in the Bevacizumab arm (34%) compared to the control arm (2%). After discontinuation of Bevacizumab and chemotherapy, recovery of ovarian function occurred in 22% (7/32) of these Bevacizumab-treated patients.
Immunocompromised Patients
There is no FDA guidance one the use of Bevacizumab in patients who are immunocompromised.
Administration and Monitoring
Administration
- Administration
- Do not administer as an intravenous push or bolus. Administer only as an intravenous (IV) infusion.
- Do not initiate Bevacizumab until at least 28 days following major surgery. Administer Bevacizumab after the surgical incision has fully healed.
- First infusion: Administer infusion over 90 minutes.
- Subsequent infusions: Administer second infusion over 60 minutes if first infusion is tolerated; administer all subsequent infusions over 30 minutes if infusion over 60 minutes is tolerated.
- Preparation for Administration
- Use appropriate aseptic technique. Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit. Withdraw necessary amount of Bevacizumab and dilute in a total volume of 100 mL of 0.9% Sodium Chloride Injection, USP. Discard any unused portion left in a vial, as the product contains no preservatives.
- DO NOT ADMINISTER OR MIX WITH DEXTROSE SOLUTION.
Monitoring
FDA Package Insert for Bevacizumab contains no information regarding drug monitoring.
IV Compatibility
There is limited information about the IV Compatibility.
Overdosage
The highest dose tested in humans (20 mg/kg IV) was associated with headache in nine of 16 patients and with severe headache in three of 16 patients.
Pharmacology
Bevacizumab?
| |
| Therapeutic monoclonal antibody | |
| Source | zu/o |
| Target | VEGF-A |
| Identifiers | |
| CAS number | |
| ATC code | L01 |
| PubChem | ? |
| DrugBank | |
| Chemical data | |
| Formula | Template:OrganicBox atomTemplate:OrganicBox atomTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBox atomTemplate:OrganicBoxTemplate:OrganicBox atomTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBox atomTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBox |
| Mol. mass | approx. 149 kDa |
| Pharmacokinetic data | |
| Bioavailability | 100% (IV only) |
| Metabolism | ? |
| Half life | 20 days (range: 11–50 days) |
| Excretion | ? |
| Therapeutic considerations | |
| Licence data |
, |
| Pregnancy cat. |
C(US) |
| Legal status |
Template:Unicode Prescription only |
| Routes | Intravenous |
Mechanism of Action
Bevacizumab binds VEGF and prevents the interaction of VEGF to its receptors (Flt-1 and KDR) on the surface of endothelial cells. The interaction of VEGF with its receptors leads to endothelial cell proliferation and new blood vessel formation in in vitro models of angiogenesis. Administration of bevacizumab to xenotransplant models of colon cancer in nude (athymic) mice caused reduction of microvascular growth and inhibition of metastatic disease progression.
Structure
Bevacizumab (bevacizumab) is a recombinant humanized monoclonal IgG1 antibody that binds to and inhibits the biologic activity of human vascular endothelial growth factor (VEGF) in in vitro and in vivo assay systems. Bevacizumab contains human framework regions and the complementarity-determining regions of a murine antibody that binds to VEGF. Bevacizumab has an approximate molecular weight of 149 kD. Bevacizumab is produced in a mammalian cell (Chinese Hamster Ovary) expression system in a nutrient medium containing the antibiotic gentamicin. Gentamicin is not detectable in the final product. Bevacizumab is a clear to slightly opalescent, colorless to pale brown, sterile, pH 6.2 solution for intravenous infusion. Bevacizumab is supplied in 100 mg and 400 mg preservative-free, single-use vials to deliver 4 mL or 16 mL of Bevacizumab (25 mg/mL). The 100 mg product is formulated in 240 mg α,α-trehalose dihydrate, 23.2 mg sodium phosphate (monobasic, monohydrate), 4.8 mg sodium phosphate (dibasic, anhydrous), 1.6 mg polysorbate 20, and Water for Injection, USP. The 400 mg product is formulated in 960 mg α,α-trehalose dihydrate, 92.8 mg sodium phosphate (monobasic, monohydrate), 19.2 mg sodium phosphate (dibasic, anhydrous), 6.4 mg polysorbate 20, and Water for Injection, USP.
Pharmacodynamics
FDA Package Insert for Bevacizumab contains no information regarding pharmacodynamics.
Pharmacokinetics
The pharmacokinetic profile of bevacizumab was assessed using an assay that measures total serum bevacizumab concentrations (i.e., the assay did not distinguish between free bevacizumab and bevacizumab bound to VEGF ligand). Based on a population pharmacokinetic analysis of 491 patients who received 1 to 20 mg/kg of Bevacizumab weekly, every 2 weeks, or every 3 weeks, the estimated half-life of bevacizumab was approximately 20 days (range 11–50 days). The predicted time to reach steady state was 100 days. The accumulation ratio following a dose of 10 mg/kg of bevacizumab every 2 weeks was 2.8. The clearance of bevacizumab varied by body weight, gender, and tumor burden. After correcting for body weight, males had a higher bevacizumab clearance (0.262 L/day vs. 0.207 L/day) and a larger Vc (3.25 L vs. 2.66 L) than females. Patients with higher tumor burden (at or above median value of tumor surface area) had a higher bevacizumab clearance (0.249 L/day vs. 0.199 L/day) than patients with tumor burdens below the median. In Study 1, there was no evidence of lesser efficacy (hazard ratio for overall survival) in males or patients with higher tumor burden treated with Bevacizumab as compared to females and patients with low tumor burden. The relationship between bevacizumab exposure and clinical outcomes has not been explored.
Nonclinical Toxicology
Carcinogenesis, Mutagenesis, Impairment of Fertility
No carcinogenicity or mutagenicity studies of bevacizumab have been conducted. Bevacizumab may impair fertility. Female cynomolgus monkeys treated with 0.4 to 20 times the recommended human dose of bevacizumab exhibited arrested follicular development or absent corpora lutea as well as dose-related decreases in ovarian and uterine weights, endometrial proliferation, and the number of menstrual cycles. Following a 4- or 12-week recovery period, there was a trend suggestive of reversibility. After the 12-week recovery period, follicular maturation arrest was no longer observed, but ovarian weights were still moderately decreased. Reduced endometrial proliferation was no longer observed at the 12-week recovery time point; however, decreased uterine weight, absent corpora lutea, and reduced number of menstrual cycles remained evident.
Animal Toxicology and/or Pharmacology
In cynomolgus monkeys, when bevacizumab was administered at doses of 0.4 to 20 times the weekly human exposure, anatomical pathology revealed several adverse effects on general growth and skeletal development, fertility and wound healing capacity. Severe physeal dysplasia was consistently reported in juvenile monkeys with open growth plates receiving 0.4 to 20 times the human dose. The physeal dysplasia was characterized by a linear cessation of growth line and chondrocyte hyperplasia which did not completely resolve after the 4 to 12 weeks recovery period without drug exposure. Rabbits dosed with bevacizumab exhibited reduced wound healing capacity. Using full-thickness skin incision and partial thickness circular dermal wound models, bevacizumab dosing resulted in reductions in wound tensile strength, decreased granulation and re-epithelialization, and delayed time to wound closure.
Reproductive and Developmental Toxicology
Pregnant rabbits dosed with 1 to 12 times the human dose of bevacizumab every three days during the period of organogenesis (gestation day 6–18) exhibited teratogenic effects, decreases in maternal and fetal body weights, and increased number of fetal resorptions. Teratogenic effects included: reduced or irregular ossification in the skull, jaw, spine, ribs, tibia and bones of the paws; meningocele; fontanel, rib and hindlimb deformities; corneal opacity; and absent hindlimb phalanges. There are no data available regarding the level of bevacizumab exposure in the offspring.
Clinical Studies
Study 1
In this double-blind, active-controlled study, patients were randomized (1:1:1) to IV bolus-IFL (irinotecan 125 mg/m2, 5-FU 500 mg/m2, and leucovorin (LV) 20 mg/m2 given once weekly for 4 weeks every 6 weeks) plus placebo (Arm 1), bolus-IFL plus Bevacizumab (5 mg/kg every 2 weeks) (Arm 2), or 5-FU/LV plus Bevacizumab (5 mg/kg every 2 weeks) (Arm 3). Enrollment in Arm 3 was discontinued, as pre-specified, when the toxicity of Bevacizumab in combination with the bolus-IFL regimen was deemed acceptable. The main outcome measure was overall survival (OS). Of the 813 patients randomized to Arms 1 and 2, the median age was 60, 40% were female, 79% were Caucasian, 57% had an ECOG performance status of 0, 21% had a rectal primary and 28% received prior adjuvant chemotherapy. In 56% of the patients, the dominant site of disease was extra-abdominal, while the liver was the dominant site in 38% of patients. The addition of Bevacizumab resulted in an improvement in survival across subgroups defined by age ( < 65 yrs, ≥ 65 yrs) and gender. Results are presented in Table 4 and Figure 1.

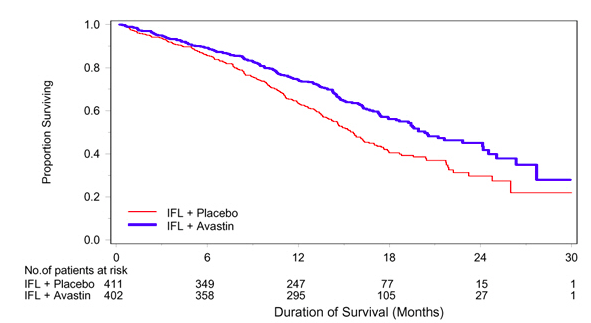
Among the 110 patients enrolled in Arm 3, median OS was 18.3 months, median progression-free survival (PFS) was 8.8 months, objective response rate (ORR) was 39%, and median duration of response was 8.5 months.
Study 2
Study 2 was a randomized, open-label, active-controlled trial in patients who were previously treated with irinotecan ± 5-FU for initial therapy for metastatic disease or as adjuvant therapy. Patients were randomized (1:1:1) to IV FOLFOX4 (Day 1: oxaliplatin 85 mg/m2 and LV 200 mg/m2 concurrently, then 5-FU 400 mg/m2 bolus followed by 600 mg/m2 continuously; Day 2: LV 200 mg/m2, then 5-FU 400 mg/m2 bolus followed by 600 mg/m2 continuously; repeated every 2 weeks), FOLFOX4 plus Bevacizumab (10 mg/kg every 2 weeks prior to FOLFOX4 on Day 1), or Bevacizumab monotherapy(10 mg/kg every 2 weeks). The main outcome measure was OS. The Bevacizumab monotherapy arm was closed to accrual after enrollment of 244 of the planned 290 patients following a planned interim analysis by the data monitoring committee based on evidence of decreased survival compared to FOLFOX4 alone. Of the 829 patients randomized to the three arms, the median age was 61 years, 40% were female, 87% were Caucasian, 49% had an ECOG performance status of 0, 26% received prior radiation therapy, and 80% received prior adjuvant chemotherapy, 99% received prior irinotecan, with or without 5-FU as therapy for metastatic disease, and 1% received prior irinotecan and 5-FU as adjuvant therapy. The addition of Bevacizumab to FOLFOX4 resulted in significantly longer survival as compared to FOLFOX4 alone (median OS 13.0 months vs. 10.8 months; hazard ratio 0.75 [95% CI 0.63, 0.89], p = 0.001 stratified log rank test) with clinical benefit seen in subgroups defined by age (< 65 yrs, ≥ 65 yrs) and gender. PFS and ORR based on investigator assessment were higher in the Bevacizumab plus FOLFOX4 arm.
Study 3
The activity of Bevacizumab in combination with bolus or infusional 5-FU/LV was evaluated in a single arm study enrolling 339 patients with mCRC with disease progression following both irinotecan- and oxaliplatin-containing chemotherapy regimens. Seventy-three percent of patients received concurrent bolus 5-FU/LV. One objective partial response was verified in the first 100 evaluable patients for an overall response rate of 1% (95% CI 0–5.5%).
Study 4
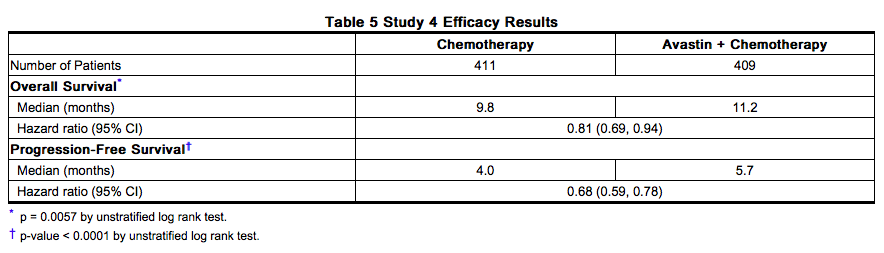
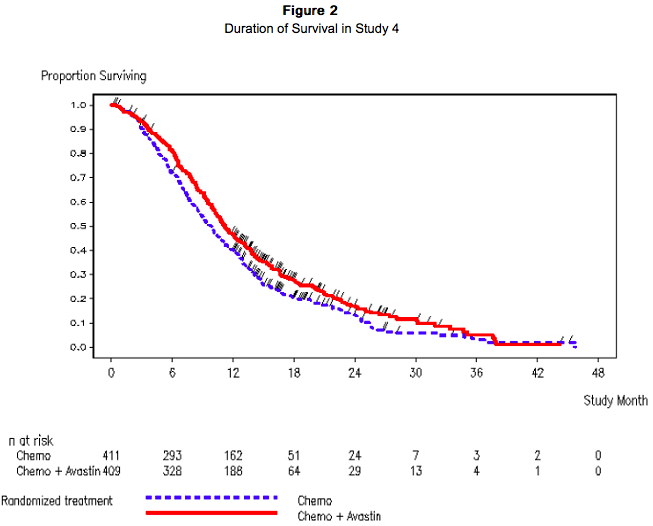
Study 4 was a prospective, randomized, open-label, multinational, controlled trial in patients with histologically confirmed metastatic colorectal cancer who had progressed on a first-line Bevacizumab containing regimen. Patients were excluded if they progressed within 3 months of initiating first-line chemotherapy and if they received Bevacizumab for less than 3 consecutive months in the first-line setting. Patients were randomized (1:1) within 3 months after discontinuation of Bevacizumab as first-line therapy to receive fluoropyrimidine/oxaliplatin- or fluoropyrimidine/irinotecan-based chemotherapy with or without Bevacizumab administered at 5 mg/kg every 2 weeks or 7.5 mg/kg every 3 weeks. The choice of second line therapy was contingent upon first-line chemotherapy treatment. Second-line treatment was administered until progressive disease or unacceptable toxicity. The main outcome measure was OS defined as the time from randomization until death from any cause. Of the 820 patients randomized, the majority of patients were male (64%) and the median age was 63.0 years (range 21 to 84 years). At baseline, 52% of patients were ECOG performance status (PS) 1, 44% were ECOG PS 0, 58% received irinotecan-based therapy as first-line treatment, 55% progressed on first-line treatment within 9 months, and 77% received their last dose of Bevacizumab as first-line treatment within 42 days of being randomized. Second-line chemotherapy regimens were generally balanced between each treatment arm. The addition of Bevacizumab to fluoropyrimidine-based chemotherapy resulted in a statistically significant prolongation of survival and PFS; there was no significant difference in overall response rate, a key secondary outcome measure. Results are presented in Table 5 and Figure 2.
Lack of Efficacy in Adjuvant Treatment of Colon Cancer
Lack of efficacy of Bevacizumab as an adjunct to standard chemotherapy for the adjuvant treatment of colon cancer was determined in two randomized, open-label, multicenter clinical trials. The first study conducted in 3451 patients with high risk stage II and III colon cancer, who had undergone surgery for colon cancer with curative intent, was a 3-arm study of Bevacizumab administered at a dose equivalent to 2.5 mg/kg/week on either a 2-weekly schedule in combination with FOLFOX4, or on a 3-weekly schedule in combination with XELOX and FOLFOX4 alone. Patients were randomized as follows: 1151 patients to FOLFOX4 arm, 1155 to FOLFOX4 plus Bevacizumab arm, and 1145 to XELOX plus Bevacizumab arm. The median age was 58 years, 54% were male, 84% were Caucasian and 29% were ≥ age 65. Eighty-three percent had stage III disease. The main efficacy outcome of the study was disease free survival (DFS) in patients with stage III colon cancer. Addition of Bevacizumab to chemotherapy did not improve DFS. As compared to the control arm, the proportion of stage III patients with disease recurrence or with death due to disease progression were numerically higher in the FOLFOX4 plus Bevacizumab and in the XELOX plus Bevacizumab arms. The hazard ratios for DFS were 1.17 (95% CI: 0.98–1.39) for the FOLFOX4 plus Bevacizumab versus FOLFOX4 and 1.07 (95% CI: 0.90–1.28) for the XELOX plus Bevacizumab versus FOLFOX4. The hazard ratios for overall survival were 1.31 (95% CI=1.03, 1.67) and 1.27 (95% CI=1.00, 1.62) for the comparison of Bevacizumab plus FOLFOX4 versus FOLFOX4 and Bevacizumab plus XELOX versus FOLFOX4, respectively. Similar lack of efficacy for DFS were observed in the Bevacizumab-containing arms compared to control in the high-risk stage II cohort. In a second study, 2710 patients with stage II and III colon cancer who had undergone surgery with curative intent, were randomized to receive either Bevacizumab administered at a dose equivalent to 2.5 mg/kg/week in combination with mFOLFOX6 (N=1354) or mFOLFOX6 alone (N=1356). The median age was 57 years, 50% were male and 87% Caucasian. Seventy-five percent had stage III disease. The main efficacy outcome was DFS among stage III patients. The hazard ratio for DFS was 0.92 (95% CI: 0.77, 1.10). Overall survival, an additional efficacy outcome, was not significantly improved with the addition of Bevacizumab to mFOLFOX6 (HR=0.96, 95% CI=[0.75,1.22].
Unresectable Non–Squamous Non–Small Cell Lung Cancer (NSCLC)
Study 5
The safety and efficacy of Bevacizumab as first-line treatment of patients with locally advanced, metastatic, or recurrent non–squamous NSCLC was studied in a single, large, randomized, active-controlled, open-label, multicenter study. Chemotherapy-naïve patients with locally advanced, metastatic or recurrent non–squamous NSCLC were randomized (1:1) to receive six 21-day cycles of paclitaxel 200 mg/m2 and carboplatin AUC = 6.0, by IV on day 1 (PC) or PC in combination with Bevacizumab 15 mg/kg by IV on day 1 (PC plus Bevacizumab). After completion or upon discontinuation of chemotherapy, patients in the PC plus Bevacizumab arm continued to receive Bevacizumab alone until disease progression or until unacceptable toxicity. Patients with predominant squamous histology (mixed cell type tumors only), central nervous system (CNS) metastasis, gross hemoptysis ( ≥ 1/2 tsp of red blood), unstable angina, or receiving therapeutic anticoagulation were excluded. The main outcome measure was duration of survival. Of the 878 patients randomized, the median age was 63, 46% were female, 43% were ≥ age 65, and 28% had ≥ 5% weight loss at study entry. Eleven percent had recurrent disease and of the 89% with newly diagnosed NSCLC, 12% had Stage IIIB with malignant pleural effusion and 76% had Stage IV disease. The results are presented in Figure 3. OS was statistically significantly higher among patients receiving PC plus Bevacizumab compared with those receiving PC alone; median OS was 12.3 months vs. 10.3 months [hazard ratio 0.80 (repeated 95% CI 0.68, 0.94), final p- value 0.013, stratified log-rank test]. Based on investigator assessment which was not independently verified, patients were reported to have longer PFS with Bevacizumab in combination with PC compared to PC alone.

In an exploratory analyses across patient subgroups, the impact of Bevacizumab on OS was less robust in the following: women [HR = 0.99 (95% CI: 0.79, 1.25)], age ≥ 65 years [HR = 0.91 (95% CI: 0.72, 1.14)] and patients with ≥ 5% weight loss at study entry [HR = 0.96 (95% CI: 0.73, 1.26)]. The safety and efficacy of Bevacizumab in patients with locally advanced, metastatic or recurrent non-squamous NSCLC, who had not received prior chemotherapy was studied in another randomized, double-blind, placebo controlled, three-arm study of Bevacizumab in combination with cisplatin and gemcitabine (CG) versus placebo and CG. A total of 1043 patients were randomized 1:1:1 to receive placebo plus CG, Bevacizumab 7.5 mg/kg plus CG or Bevacizumab 15.0 mg/kg plus CG. The median age was 58 years, 36% were female, and 29% were ≥ age 65. Eight percent had recurrent disease and 77% had Stage IV disease. Progression-free survival, the main efficacy outcome measure, was significantly higher in both Bevacizumab containing arms compared to the placebo arm [HR 0.75 (95% CI 0.62, 0.91), p = 0.0026 for the Bevacizumab 7.5 mg/kg plus CG arm and HR 0.82 (95% CI 0.68; 0.98), p = 0.0301 for the Bevacizumab 15.0 mg/kg plus CG arm]. The addition of Bevacizumab to CG chemotherapy failed to demonstrate an improvement in the duration of overall survival, an additional efficacy outcome measure, [HR 0.93 (95% CI 0.78; 1.11), p = 0.4203 for the Bevacizumab 7.5 mg/kg plus CG arm and HR 1.03 (95% CI 0.86; 1.23), p = 0.7613 for the Bevacizumab 15.0 mg/kg plus CG arm].
Study 6
The efficacy and safety of Bevacizumab was evaluated in Study 6, an open-label, multicenter, randomized, non-comparative study of patients with previously treated glioblastoma. Patients received Bevacizumab (10 mg/kg IV) alone or Bevacizumab plus irinotecan every 2 weeks until disease progression or until unacceptable toxicity. All patients received prior radiotherapy (completed at least 8 weeks prior to receiving Bevacizumab) and temozolomide. Patients with active brain hemorrhage were excluded. Of the 85 patients randomized to the Bevacizumab arm, the median age was 54 years, 32% were female, 81% were in first relapse, Karnofsky performance status was 90–100 for 45% and 70–80 for 55%. The efficacy of Bevacizumab was demonstrated using response assessment based on both WHO radiographic criteria and by stable or decreasing corticosteroid use, which occurred in 25.9% (95% CI 17.0%, 36.1%) of the patients. Median duration of response was 4.2 months (95% CI 3.0, 5.7). Radiologic assessment was based on MRI imaging (using T1 and T2/FLAIR). MRI does not necessarily distinguish between tumor, edema, and radiation necrosis.
Study 7
Study 7, was a single-arm, single institution trial with 56 patients with glioblastoma. All patients had documented disease progression after receiving temozolomide and radiation therapy. Patients received Bevacizumab 10 mg/kg IV every 2 weeks until disease progression or unacceptable toxicity. The median age was 54, 54% were male, 98% Caucasian, and 68% had a Karnofsky Performance Status of 90–100. The efficacy of Bevacizumab was supported by an objective response rate of 19.6% (95% CI 10.9%, 31.3%) using the same response criteria as in Study 6. Median duration of response was 3.9 months (95% CI 2.4, 17.4).
Metastatic Renal Cell Carcinoma (mRCC)
Study 8
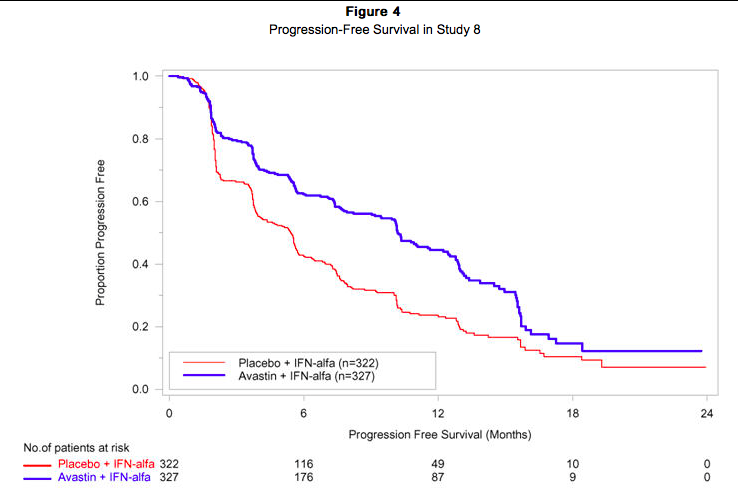
Patients with treatment-naïve mRCC were evaluated in a multicenter, randomized, double-blind, international study comparing Bevacizumab plus interferon alfa 2a (IFN-α2a) versus placebo plus IFN-α2a. A total of 649 patients who had undergone a nephrectomy were randomized (1:1) to receive either Bevacizumab (10 mg/kg IV infusion every 2 weeks; n = 327) or placebo (IV every 2 weeks; n = 322) in combination with IFN-α2a (9 MIU subcutaneously three times weekly, for a maximum of 52 weeks). Patients were treated until disease progression or unacceptable toxicity. The main outcome measure of the study was investigator-assessed PFS. Secondary outcome measures were ORR and OS. The median age was 60 years (range 18–82), 96% were white, and 70% were male. The study population was characterized by Motzer scores as follows: 28% favorable (0), 56% intermediate (1-2), 8% poor (3–5), and 7% missing. The results are presented in Figure 4. PFS was statistically significantly prolonged among patients receiving Bevacizumab plus IFN-α2a compared to those receiving IFN-α2a alone; median PFS was 10.2 months vs. 5.4 months [HR 0.60 (95% CI 0.49, 0.72), p-value < 0.0001, stratified log-rank test]. Among the 595 patients with measurable disease, ORR was also significantly higher (30% vs. 12%, p < 0.0001, stratified CMH test). There was no improvement in OS based on the final analysis conducted after 444 deaths, with a median OS of 23 months in the Bevacizumab plus IFN-α2a arm and 21 months in the IFN-α2a plus placebo arm [HR 0.86, (95% CI 0.72, 1.04)].
How Supplied
Bevacizumab vials [100 mg (NDC 50242-060-01) and 400 mg (NDC 50242-061-01)] are stable at 2–8°C (36–46°F). Bevacizumab vials should be protected from light. Do not freeze or shake.
Storage
Diluted Bevacizumab solutions may be stored at 2–8°C (36–46°F) for up to 8 hours. Store in the original carton until time of use. No incompatibilities between Bevacizumab and polyvinylchloride or polyolefin bags have been observed.
Images
Drug Images
{{#ask: Page Name::Bevacizumab |?Pill Name |?Drug Name |?Pill Ingred |?Pill Imprint |?Pill Dosage |?Pill Color |?Pill Shape |?Pill Size (mm) |?Pill Scoring |?NDC |?Drug Author |format=template |template=DrugPageImages |mainlabel=- |sort=Pill Name }}
Package and Label Display Panel
{{#ask: Label Page::Bevacizumab |?Label Name |format=template |template=DrugLabelImages |mainlabel=- |sort=Label Page }}
Patient Counseling Information
Advise patients:
- To undergo routine blood pressure monitoring and to contact their health care provider if blood pressure is elevated.
- To immediately contact their health care provider for unusual bleeding, high fever, rigors, sudden onset of worsening neurological function, or persistent or severe abdominal pain, severe constipation, or vomiting.
- Of increased risk of wound healing complications during and following Bevacizumab.
- Of increased risk of an arterial thromboembolic event.
- Of the potential risk to the fetus during and following Bevacizumab and the need to continue adequate contraception for at least 6 months following last dose of Bevacizumab.
- Of the increased risk for ovarian failure following Bevacizumab treatment.
Precautions with Alcohol
Alcohol-Bevacizumab interaction has not been established. Talk to your doctor about the effects of taking alcohol with this medication.
Brand Names
Avastin
Look-Alike Drug Names
Avastin - Astelin[20]
Price
References
The contents of this FDA label are provided by the National Library of Medicine.
- ↑ El Matri L, Kort F, Bouraoui R, Karim B, Chebil A, Chaker N (2011). "Intravitreal bevacizumab for the treatment of choroidal neovascularization secondary to angioid streaks: one year of follow-up". Acta Ophthalmol. 89 (7): 641–6. doi:10.1111/j.1755-3768.2009.01836.x. PMID 21155980.
- ↑ Donati MC, Virgili G, Bini A, Giansanti F, Rapizzi E, Giacomelli G; et al. (2009). "Intravitreal bevacizumab (Bevacizumab) for choroidal neovascularization in angioid streaks: a case series". Ophthalmologica. 223 (1): 24–7. doi:10.1159/000161879. PMID 18849633.
- ↑ Hikichi T, Higuchi M, Matsushita T, Kosaka S, Matsushita R, Takami K; et al. (2014). "Two-year outcomes of intravitreal bevacizumab therapy for macular oedema secondary to branch retinal vein occlusion". Br J Ophthalmol. 98 (2): 195–9. doi:10.1136/bjophthalmol-2013-303121. PMC 3913112. PMID 24215032.
- ↑ Epstein DL, Algvere PV, von Wendt G, Seregard S, Kvanta A (2012). "Bevacizumab for macular edema in central retinal vein occlusion: a prospective, randomized, double-masked clinical study". Ophthalmology. 119 (6): 1184–9. doi:10.1016/j.ophtha.2012.01.022. PMID 22424833.
- ↑ Iacono P, Parodi MB, Papayannis A, Kontadakis S, Sheth S, Cascavilla ML; et al. (2012). "Intravitreal ranibizumab versus bevacizumab for treatment of myopic choroidal neovascularization". Retina. 32 (8): 1539–46. doi:10.1097/IAE.0b013e31826956b7. PMID 22922846.
- ↑ Michaelides M, Kaines A, Hamilton RD, Fraser-Bell S, Rajendram R, Quhill F; et al. (2010). "A prospective randomized trial of intravitreal bevacizumab or laser therapy in the management of diabetic macular edema (BOLT study) 12-month data: report 2". Ophthalmology. 117 (6): 1078–1086.e2. doi:10.1016/j.ophtha.2010.03.045. PMID 20416952.
- ↑ Zhang ZH, Liu HY, Hernandez-Da Mota SE, Romano MR, Falavarjani KG, Ahmadieh H; et al. (2013). "Vitrectomy with or without preoperative intravitreal bevacizumab for proliferative diabetic retinopathy: a meta-analysis of randomized controlled trials". Am J Ophthalmol. 156 (1): 106–115.e2. doi:10.1016/j.ajo.2013.02.008. PMID 23791371.
- ↑ Karnezis TT, Davidson TM (2012). "Treatment of hereditary hemorrhagic telangiectasia with submucosal and topical bevacizumab therapy". Laryngoscope. 122 (3): 495–7. doi:10.1002/lary.22501. PMID 22147664.
- ↑ Ohtsu A, Shah MA, Van Cutsem E, Rha SY, Sawaki A, Park SR; et al. (2011). "Bevacizumab in combination with chemotherapy as first-line therapy in advanced gastric cancer: a randomized, double-blind, placebo-controlled phase III study". J Clin Oncol. 29 (30): 3968–76. doi:10.1200/JCO.2011.36.2236. PMID 21844504.
- ↑ Robert NJ, Diéras V, Glaspy J, Brufsky AM, Bondarenko I, Lipatov ON; et al. (2011). "RIBBON-1: randomized, double-blind, placebo-controlled, phase III trial of chemotherapy with or without bevacizumab for first-line treatment of human epidermal growth factor receptor 2-negative, locally recurrent or metastatic breast cancer". J Clin Oncol. 29 (10): 1252–60. doi:10.1200/JCO.2010.28.0982. PMID 21383283.
- ↑ Miles DW, Chan A, Dirix LY, Cortés J, Pivot X, Tomczak P; et al. (2010). "Phase III study of bevacizumab plus docetaxel compared with placebo plus docetaxel for the first-line treatment of human epidermal growth factor receptor 2-negative metastatic breast cancer". J Clin Oncol. 28 (20): 3239–47. doi:10.1200/JCO.2008.21.6457. PMID 20498403.
- ↑ Costagliola C, Cipollone U, Rinaldi M, della Corte M, Semeraro F, Romano MR (2008). "Intravitreal bevacizumab (Bevacizumab) injection for neovascular glaucoma: a survey on 23 cases throughout 12-month follow-up". Br J Clin Pharmacol. 66 (5): 667–73. doi:10.1111/j.1365-2125.2008.03278.x. PMC 2661982. PMID 19032174.
- ↑ Burger RA, Brady MF, Bookman MA, Fleming GF, Monk BJ, Huang H; et al. (2011). "Incorporation of bevacizumab in the primary treatment of ovarian cancer". N Engl J Med. 365 (26): 2473–83. doi:10.1056/NEJMoa1104390. PMID 22204724.
- ↑ Perren TJ, Swart AM, Pfisterer J, Ledermann JA, Pujade-Lauraine E, Kristensen G; et al. (2011). "A phase 3 trial of bevacizumab in ovarian cancer". N Engl J Med. 365 (26): 2484–96. doi:10.1056/NEJMoa1103799. PMID 22204725.
- ↑ Backes FJ, Richardson DL, McCann GA, Smith B, Salani R, Eisenhauer EL; et al. (2013). "Should bevacizumab be continued after progression on bevacizumab in recurrent ovarian cancer?". Int J Gynecol Cancer. 23 (5): 833–8. doi:10.1097/IGC.0b013e318290ea69. PMID 23640292.
- ↑ Aghajanian C, Blank SV, Goff BA, Judson PL, Teneriello MG, Husain A; et al. (2012). "OCEANS: a randomized, double-blind, placebo-controlled phase III trial of chemotherapy with or without bevacizumab in patients with platinum-sensitive recurrent epithelial ovarian, primary peritoneal, or fallopian tube cancer". J Clin Oncol. 30 (17): 2039–45. doi:10.1200/JCO.2012.42.0505. PMC 3646321. PMID 22529265.
- ↑ Garcia AA, Hirte H, Fleming G, Yang D, Tsao-Wei DD, Roman L; et al. (2008). "Phase II clinical trial of bevacizumab and low-dose metronomic oral cyclophosphamide in recurrent ovarian cancer: a trial of the California, Chicago, and Princess Margaret Hospital phase II consortia". J Clin Oncol. 26 (1): 76–82. doi:10.1200/JCO.2007.12.1939. PMID 18165643.
- ↑ Wu WC, Yeh PT, Chen SN, Yang CM, Lai CC, Kuo HK (2011). "Effects and complications of bevacizumab use in patients with retinopathy of prematurity: a multicenter study in taiwan". Ophthalmology. 118 (1): 176–83. doi:10.1016/j.ophtha.2010.04.018. PMID 20673589.
- ↑ Xu Y, Zhang Q, Kang X, Zhu Y, Li J, Chen Y; et al. (2013). "Early vitreoretinal surgery on vascularly active stage 4 retinopathy of prematurity through the preoperative intravitreal bevacizumab injection". Acta Ophthalmol. 91 (4): e304–10. doi:10.1111/aos.12055. PMID 23647944.
- ↑ "https://www.ismp.org". External link in
|title=(help)
{{#subobject:
|Label Page=Bevacizumab |Label Name=Bevacizumab_label_01.jpg
}}
{{#subobject:
|Label Page=Bevacizumab |Label Name=Bevacizumab_label_02.jpg
}}
{{#subobject:
|Label Page=Bevacizumab |Label Name=Bevacizumab_panel_01.png
}}
{{#subobject:
|Label Page=Bevacizumab |Label Name=Bevacizumab_panel_02.png
}}