Alpha-synuclein
| VALUE_ERROR (nil) | |||||||
|---|---|---|---|---|---|---|---|
| Identifiers | |||||||
| Aliases | |||||||
| External IDs | GeneCards: [1] | ||||||
| Orthologs | |||||||
| Species | Human | Mouse | |||||
| Entrez |
|
| |||||
| Ensembl |
|
| |||||
| UniProt |
|
| |||||
| RefSeq (mRNA) |
|
| |||||
| RefSeq (protein) |
|
| |||||
| Location (UCSC) | n/a | n/a | |||||
| PubMed search | n/a | n/a | |||||
| Wikidata | |||||||
| |||||||
Alpha-synuclein is a protein that, in humans, is encoded by the SNCA gene.[1] It is abundant in the brain while smaller amounts are found in the heart, muscles, and other tissues. In the brain, alpha-synuclein is found mainly at the tips of nerve cells (neurons) in specialized structures called presynaptic terminals.[1] Within these structures, alpha-synuclein interacts with phospholipids[2] and proteins.[1] Presynaptic terminals release chemical messengers, called neurotransmitters, from compartments known as synaptic vesicles. The release of neurotransmitters relays signals between neurons and is critical for normal brain function.[1]
Although the function of alpha-synuclein is not well understood, studies suggest that it plays a role in maintaining a supply of synaptic vesicles in presynaptic terminals by clustering synaptic vesicles.[3] It may also help regulate the release of dopamine, a type of neurotransmitter that is critical for controlling the start and stop of voluntary and involuntary movements.[1]
The human alpha-synuclein protein is made of 140 amino acids and is encoded by the SNCA gene.[4][5][6] An alpha-synuclein fragment, known as the non-Abeta component (NAC) of Alzheimer's disease amyloid, originally found in an amyloid-enriched fraction, was shown to be a fragment of its precursor protein, NACP.[4] It was later determined that NACP was the human homologue of Torpedo synuclein. Therefore, NACP is now referred to as human alpha-synuclein.
Tissue expression
Alpha-synuclein is a synuclein protein of unknown function primarily found in neural tissue, making up as much as 1% of all proteins in the cytosol of brain cells.[7] It is predominantly expressed in the neocortex, hippocampus, substantia nigra, thalamus, and cerebellum. It is predominantly a neuronal protein, but can also be found in the neuroglial cells.[citation needed] In melanocytic cells, SNCA protein expression may be regulated by MITF.[8]
It has been established that alpha-synuclein is extensively localized in the nucleus of mammalian brain neurons, suggesting a role of alpha-synuclein in the nucleus.[9] Synuclein is however found predominantly in the presynaptic termini, in both free or membrane-bound forms,[10] with roughly 15% of synuclein being membrane-bound in any moment in neurons.[11]
It has also been shown that alpha-synuclein is localized in neuronal mitochondria.[12][13] Alpha-synuclein is highly expressed in the mitochondria in olfactory bulb, hippocampus, striatum and thalamus, where the cytosolic alpha-synuclein is also rich. However, the cerebral cortex and cerebellum are two exceptions, which contain rich cytosolic alpha-synuclein but very low levels of mitochondrial alpha-synuclein. It has been shown that alpha-synuclein is localized in the inner membrane of mitochondria, and that the inhibitory effect of alpha-synuclein on complex I activity of mitochondrial respiratory chain is dose-dependent. Thus, it is suggested that alpha-synuclein in mitochondria is differentially expressed in different brain regions and the background levels of mitochondrial alpha-synuclein may be a potential factor affecting mitochondrial function and predisposing some neurons to degeneration.[13]
At least three isoforms of synuclein are produced through alternative splicing.[14] The majority form of the protein, and the one most investigated, is the full-length protein of 140 amino acids. Other isoforms are alpha-synuclein-126, which lacks residues 41-54 due to loss of exon 3; and alpha-synuclein-112,[15] which lacks residue 103-130 due to loss of exon 5.[14]
Structure
Alpha-synuclein in solution is considered to be an intrinsically disordered protein, i.e. it lacks a single stable 3D structure.[16] As of 2014, an increasing number of reports suggest, however, the presence of partial structures or mostly structured oligomeric states in the solution structure of alpha-synuclein even in the absence of lipids. This trend is also supported by a large number of single molecule (optical tweezers) measurements on single copies of monomeric alpha-synuclein as well as covalently enforced dimers or tetramers of alpha-synuclein.[17]
Function
Alpha-synuclein is specifically upregulated in a discrete population of presynaptic terminals of the brain during a period of acquisition-related synaptic rearrangement.[18] It has been shown that alpha-synuclein significantly interacts with tubulin,[19] and that alpha-synuclein may have activity as a potential microtubule-associated protein, like tau.[20]
Recent evidence suggests that alpha-synuclein functions as a molecular chaperone in the formation of SNARE complexes.[21][22] In particular, it simultaneously binds to phospholipids of the plasma membrane via its N-terminus domain and to synaptobrevin-2 via its C-terminus domain, with increased importance during synaptic activity.[23] Indeed, there is growing evidence that alpha-synuclein is involved in the functioning of the neuronal Golgi apparatus and vesicle trafficking.[24]
Apparently, alpha-synuclein is essential for normal development of the cognitive functions. Knock-out mice with the targeted inactivation of the expression of alpha-synuclein show impaired spatial learning and working memory.[25]
Interaction with lipid membranes
Experimental evidence has been collected on the interaction of alpha-synuclein with membrane and its involvement with membrane composition and turnover. Yeast genome screening has found that several genes that deal with lipid metabolism play a role in alpha-synuclein toxicity.[26] Conversely, alpha-synuclein expression levels can affect the viscosity and the relative amount of fatty acids in the lipid bilayer.[27]
Alpha-synuclein is known to directly bind to lipid membranes, associating with the negatively charged surfaces of phospholipids.[27] Alpha-synuclein forms an extended helical structure on small unilamellar vesicles.[28] A preferential binding to small vesicles has been found.[29] The binding of alpha-synuclein to lipid membranes has complex effects on the latter, altering the bilayer structure and leading to the formation of small vesicles.[30] Alpha-synuclein has been shown to bend membranes of negatively charged phospholipid vesicles and form tubules from large lipid vesicles.[31] Using cryo-EM it was shown that these are micellar tubes of ~5-6 nm diameter.[32] Alpha-synuclein has also been shown to form lipid disc-like particles similar to apolipoproteins.[33] EPR studies have shown that the structure of alpha synuclein is dependent on the binding surface.[34] The protein adopts a broken-helical conformation on lipoprotein particles while it forms an extended helical structure on lipid vesicles and membrane tubes.[34] Studies have also suggested a possible antioxidant activity of alpha-synuclein in the membrane.[35]
Membrane interaction of alpha-synuclein modulates or affects its rate of aggregation.[36] The membrane-mediated modulation of aggregation is very similar to that observed for other amyloid proteins such as IAPP and abeta.[36] Aggregated states of alpha-synuclein permeate the membrane of lipid vesicles.[37] They are formed upon interaction with peroxidation-prone polyunsaturated fatty acids (PUFA) but not with monounsaturated fatty acids[38] and the binding of lipid autoxidation-promoting transition metals such as iron or copper provokes oligomerization of alpha-synuclein.[39] The aggregated alpha-synuclein has a specific activity for peroxidized lipids and induces lipid autoxidation in PUFA-rich membranes of both neurons and astrocytes, decreasing resistance to apoptosis.[40] Lipid autoxidation is inhibited if the cells are pre-incubated with isotope-reinforced PUFAs (D-PUFA).[41]
Sequence
Alpha-synuclein primary structure is usually divided in three distinct domains:
- Residues 1-60: An amphipathic N-terminal region dominated by four 11-residue repeats including the consensus sequence KTKEGV. This sequence has a structural alpha helix propensity similar to apolipoproteins-binding domains[42]
- Residues 61-95: A central hydrophobic region which includes the non-amyloid-β component (NAC) region, involved in protein aggregation[4]
- Residues 96-140: a highly acidic and proline-rich region which has no distinct structural propensity
Autoproteolytic activity
The use of high-resolution ion-mobility mass spectrometry (IMS-MS) on HPLC-purified alpha-synuclein in vitro has shown alpha-synuclein to be autoproteolytic (self-proteolytic), generating a variety of small molecular weight fragments upon incubation.[43] The 14.46 kDa protein was found to generate numerous smaller fragments, including 12.16 kDa (amino acids 14-133) and 10.44 kDa (40-140) fragments formed through C- and N-terminal truncation and a 7.27 kDa C-terminal fragment (72-140). The 7.27 kDa fragment, which contains the majority of the NAC region, aggregated considerably faster than full-length alpha-synuclein. It is possible that these autoproteolytic products play a role as intermediates or cofactors in the aggregation of alpha-synuclein in vivo.
Clinical significance
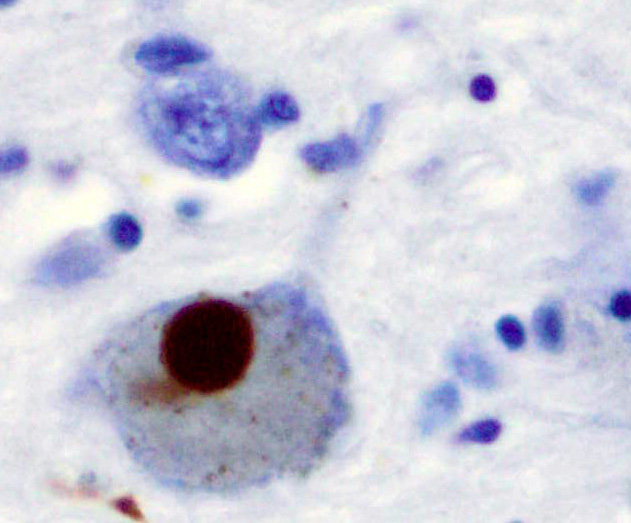
Classically considered an unstructured soluble protein, unmutated α-synuclein forms a stably folded tetramer that resists aggregation.[44] This observation, though reproduced and extended by several labs,[45][46][47] is still a matter of debate in the field due to conflicting reports.[48][49][50] Nevertheless, alpha-synuclein aggregates to form insoluble fibrils in pathological conditions characterized by Lewy bodies, such as Parkinson's disease, dementia with Lewy bodies and multiple system atrophy.[51][52] These disorders are known as synucleinopathies. Alpha-synuclein is the primary structural component of Lewy body fibrils. Occasionally, Lewy bodies contain tau protein;[53] however, alpha-synuclein and tau constitute two distinctive subsets of filaments in the same inclusion bodies.[54] Alpha-synuclein pathology is also found in both sporadic and familial cases with Alzheimer's disease.[55]
The aggregation mechanism of alpha-synuclein is uncertain. There is evidence of a structured intermediate rich in beta structure that can be the precursor of aggregation and, ultimately, Lewy bodies.[56] A single molecule study in 2008 suggests alpha-synuclein exists as a mix of unstructured, alpha-helix, and beta-sheet-rich conformers in equilibrium. Mutations or buffer conditions known to improve aggregation strongly increase the population of the beta conformer, thus suggesting this could be a conformation related to pathogenic aggregation.[57] One theory is that the majority of alpha-synuclein aggregates are located in the presynapse as smaller deposits which causes synaptic dysfunction.[58] Among the strategies for treating synucleinopathies are compounds that inhibit aggregation of alpha-synuclein. It has been shown that the small molecule cuminaldehyde inhibits fibrillation of alpha-synuclein.[59] The Epstein-Barr virus has been implicated in these disorders.[60]
In rare cases of familial forms of Parkinson's disease, there is a mutation in the gene coding for alpha-synuclein. Five point mutations have been identified thus far: A53T,[61] A30P,[62] E46K,[63] H50Q,[64] and G51D.[65] It has been reported that some mutations influence the initiation and amplification steps of the aggregation process.[66] Genomic duplication and triplication of the gene appear to be a rare cause of Parkinson's disease in other lineages, although more common than point mutations.[67] Hence certain mutations of alpha-synuclein may cause it to form amyloid-like fibrils that contribute to Parkinson's disease.
Certain sections of the alpha-synuclein protein may play a role in the tauopathies.[68]
A prion form of the protein alpha-synuclein may be a causal agent for the disease multiple system atrophy.[69][70][71]
This section needs additional citations for verification. (November 2015) (Learn how and when to remove this template message) |
Antibodies against alpha-synuclein have replaced antibodies against ubiquitin as the gold standard for immunostaining of Lewy bodies.[73] The central panel in the figure to the right shows the major pathway for protein aggregation. Monomeric α-synuclein is natively unfolded in solution but can also bind to membranes in an α-helical form. It seems likely that these two species exist in equilibrium within the cell, although this is unproven. From in vitro work, it is clear that unfolded monomer can aggregate first into small oligomeric species that can be stabilized by β-sheet-like interactions and then into higher molecular weight insoluble fibrils. In a cellular context, there is some evidence that the presence of lipids can promote oligomer formation: α-synuclein can also form annular, pore-like structures that interact with membranes. The deposition of α-synuclein into pathological structures such as Lewy bodies is probably a late event that occurs in some neurons. On the left hand side are some of the known modifiers of this process. Electrical activity in neurons changes the association of α-synuclein with vesicles and may also stimulate polo-like kinase 2 (PLK2), which has been shown to phosphorylate α-synuclein at Ser129. Other kinases have also been proposed to be involved. As well as phosphorylation, truncation through proteases such as calpains, and nitration, probably through nitric oxide (NO) or other reactive nitrogen species that are present during inflammation, all modify synuclein such that it has a higher tendency to aggregate. The addition of ubiquitin (shown as a black spot) to Lewy bodies is probably a secondary process to deposition. On the right are some of the proposed cellular targets for α-synuclein mediated toxicity, which include (from top to bottom) ER-golgi transport, synaptic vesicles, mitochondria and lysosomes and other proteolytic machinery. In each of these cases, it is proposed that α-synuclein has detrimental effects, listed below each arrow, although at this time it is not clear if any of these are either necessary or sufficient for toxicity in neurons.
Protein-protein interactions
Alpha-synuclein has been shown to interact with
- Dopamine transporter,[74][75]
- Parkin (ligase),[76][77]
- Phospholipase D1,[78]
- SNCAIP,[79][80][81][82]
- Tau protein.[83][84]
- Beta amyloid[85]
See also
- Synuclein
- Contursi Terme - the village in Italy where a mutation in the α-synuclein gene led to a family history of Parkinson's disease
References
- ↑ 1.0 1.1 1.2 1.3 1.4 "Genetics Home Reference: SNCA". U.S. National Library of Medicine. 12 Nov 2013. Retrieved 14 Nov 2013.
- ↑ Chandra S, Chen X, Rizo J, Jahn R, Südhof TC (April 2003). "A broken alpha -helix in folded alpha -Synuclein". The Journal of Biological Chemistry. 278 (17): 15313–8. doi:10.1074/jbc.M213128200. PMID 12586824.
- ↑ Diao J, Burré J, Vivona S, Cipriano DJ, Sharma M, Kyoung M, Südhof TC, Brunger AT (April 2013). "Native α-synuclein induces clustering of synaptic-vesicle mimics via binding to phospholipids and synaptobrevin-2/VAMP2". eLife. 2: e00592. doi:10.7554/eLife.00592. PMC 3639508. PMID 23638301.
- ↑ 4.0 4.1 4.2 Uéda K, Fukushima H, Masliah E, Xia Y, Iwai A, Yoshimoto M, Otero DA, Kondo J, Ihara Y, Saitoh T (December 1993). "Molecular cloning of cDNA encoding an unrecognized component of amyloid in Alzheimer disease". Proceedings of the National Academy of Sciences of the United States of America. 90 (23): 11282–6. doi:10.1073/pnas.90.23.11282. PMC 47966. PMID 8248242.
- ↑ Xia Y, Saitoh T, Uéda K, Tanaka S, Chen X, Hashimoto M, Hsu L, Conrad C, Sundsmo M, Yoshimoto M, Thal L, Katzman R, Masliah E (October 2001). "Characterization of the human alpha-synuclein gene: Genomic structure, transcription start site, promoter region and polymorphisms". Journal of Alzheimer's Disease. 3 (5): 485–494. PMID 12214035. Archived from the original on 2016-05-14.
- ↑ Xia Y, Saitoh T, Uéda K, Tanaka S, Chen X, Hashimoto M, Hsu L, Conrad C, Sundsmo M, Yoshimoto M, Thal L, Katzman R, Masliah E (2002). "Characterization of the human alpha-synuclein gene: Genomic structure, transcription start site, promoter region and polymorphisms: Erratum p489 Fig 3". J. Alzheimers Dis. 4 (4): 337. Archived from the original on 2016-05-14.
- ↑ Iwai A, Masliah E, Yoshimoto M, Ge N, Flanagan L, de Silva HA, Kittel A, Saitoh T (February 1995). "The precursor protein of non-A beta component of Alzheimer's disease amyloid is a presynaptic protein of the central nervous system". Neuron. 14 (2): 467–75. doi:10.1016/0896-6273(95)90302-X. PMID 7857654.
- ↑ Hoek KS, Schlegel NC, Eichhoff OM, Widmer DS, Praetorius C, Einarsson SO, Valgeirsdottir S, Bergsteinsdottir K, Schepsky A, Dummer R, Steingrimsson E (December 2008). "Novel MITF targets identified using a two-step DNA microarray strategy". Pigment Cell & Melanoma Research. 21 (6): 665–76. doi:10.1111/j.1755-148X.2008.00505.x. PMID 19067971.
- ↑ Yu S, Li X, Liu G, Han J, Zhang C, Li Y, Xu S, Liu C, Gao Y, Yang H, Uéda K, Chan P (March 2007). "Extensive nuclear localization of alpha-synuclein in normal rat brain neurons revealed by a novel monoclonal antibody". Neuroscience. 145 (2): 539–55. doi:10.1016/j.neuroscience.2006.12.028. PMID 17275196.
- ↑ McLean PJ, Kawamata H, Ribich S, Hyman BT (March 2000). "Membrane association and protein conformation of alpha-synuclein in intact neurons. Effect of Parkinson's disease-linked mutations". The Journal of Biological Chemistry. 275 (12): 8812–6. doi:10.1074/jbc.275.12.8812. PMID 10722726.
- ↑ Lee HJ, Choi C, Lee SJ (January 2002). "Membrane-bound alpha-synuclein has a high aggregation propensity and the ability to seed the aggregation of the cytosolic form". The Journal of Biological Chemistry. 277 (1): 671–8. doi:10.1074/jbc.M107045200. PMID 11679584.
- ↑ Zhang L, Zhang C, Zhu Y, Cai Q, Chan P, Uéda K, Yu S, Yang H (December 2008). "Semi-quantitative analysis of alpha-synuclein in subcellular pools of rat brain neurons: an immunogold electron microscopic study using a C-terminal specific monoclonal antibody". Brain Research. 1244: 40–52. doi:10.1016/j.brainres.2008.08.067. PMID 18817762.
- ↑ 13.0 13.1 Liu G, Zhang C, Yin J, Li X, Cheng F, Li Y, Yang H, Uéda K, Chan P, Yu S (May 2009). "alpha-Synuclein is differentially expressed in mitochondria from different rat brain regions and dose-dependently down-regulates complex I activity". Neuroscience Letters. 454 (3): 187–92. doi:10.1016/j.neulet.2009.02.056. PMID 19429081.
- ↑ 14.0 14.1 Beyer K (September 2006). "Alpha-synuclein structure, posttranslational modification and alternative splicing as aggregation enhancers". Acta Neuropathologica. 112 (3): 237–51. doi:10.1007/s00401-006-0104-6. PMID 16845533.
- ↑ Uéda K, Saitoh T, Mori H (December 1994). "Tissue-dependent alternative splicing of mRNA for NACP, the precursor of non-A beta component of Alzheimer's disease amyloid". Biochemical and Biophysical Research Communications. 205 (2): 1366–72. doi:10.1006/bbrc.1994.2816. PMID 7802671.
- ↑ van Rooijen BD, van Leijenhorst-Groener KA, Claessens MM, Subramaniam V (December 2009). "Tryptophan fluorescence reveals structural features of alpha-synuclein oligomers". Journal of Molecular Biology. 394 (5): 826–33. doi:10.1016/j.jmb.2009.10.021. PMID 19837084.
- ↑ Neupane K, Solanki A, Sosova I, Belov M, Woodside MT (January 2014). "Diverse metastable structures formed by small oligomers of α-synuclein probed by force spectroscopy". PLOS One. 9 (1): e86495. doi:10.1371/journal.pone.0086495. PMC 3901707. PMID 24475132.
- ↑ George JM, Jin H, Woods WS, Clayton DF (August 1995). "Characterization of a novel protein regulated during the critical period for song learning in the zebra finch". Neuron. 15 (2): 361–72. doi:10.1016/0896-6273(95)90040-3. PMID 7646890.
- ↑ Alim MA, Hossain MS, Arima K, Takeda K, Izumiyama Y, Nakamura M, Kaji H, Shinoda T, Hisanaga S, Ueda K (January 2002). "Tubulin seeds alpha-synuclein fibril formation". The Journal of Biological Chemistry. 277 (3): 2112–7. doi:10.1074/jbc.M102981200. PMID 11698390.
- ↑ Alim MA, Ma QL, Takeda K, Aizawa T, Matsubara M, Nakamura M, Asada A, Saito T, Kaji H, Yoshii M, Hisanaga S, Uéda K (August 2004). "Demonstration of a role for alpha-synuclein as a functional microtubule-associated protein". Journal of Alzheimer's Disease. 6 (4): 435–42, discussion 443–9. PMID 15345814.
- ↑ Bonini NM, Giasson BI (November 2005). "Snaring the function of alpha-synuclein". Cell. 123 (3): 359–61. doi:10.1016/j.cell.2005.10.017. PMID 16269324.
- ↑ Chandra S, Gallardo G, Fernández-Chacón R, Schlüter OM, Südhof TC (November 2005). "Alpha-synuclein cooperates with CSPalpha in preventing neurodegeneration". Cell. 123 (3): 383–96. doi:10.1016/j.cell.2005.09.028. PMID 16269331.
- ↑ Burré J, Sharma M, Tsetsenis T, Buchman V, Etherton MR, Südhof TC (September 2010). "Alpha-synuclein promotes SNARE-complex assembly in vivo and in vitro". Science. 329 (5999): 1663–7. doi:10.1126/science.1195227. PMC 3235365. PMID 20798282.
- ↑ Cooper AA, Gitler AD, Cashikar A, Haynes CM, Hill KJ, Bhullar B, Liu K, Xu K, Strathearn KE, Liu F, Cao S, Caldwell KA, Caldwell GA, Marsischky G, Kolodner RD, Labaer J, Rochet JC, Bonini NM, Lindquist S (July 2006). "Alpha-synuclein blocks ER-Golgi traffic and Rab1 rescues neuron loss in Parkinson's models". Science. 313 (5785): 324–8. doi:10.1126/science.1129462. PMC 1983366. PMID 16794039.
- ↑ Kokhan VS, Afanasyeva MA, Van'kin GI (May 2012). "α-Synuclein knockout mice have cognitive impairments". Behavioural Brain Research. 231 (1): 226–30. doi:10.1016/j.bbr.2012.03.026. PMID 22469626.
- ↑ Willingham S, Outeiro TF, DeVit MJ, Lindquist SL, Muchowski PJ (December 2003). "Yeast genes that enhance the toxicity of a mutant huntingtin fragment or alpha-synuclein". Science. 302 (5651): 1769–72. doi:10.1126/science.1090389. PMID 14657499.
- ↑ 27.0 27.1 Uversky VN (October 2007). "Neuropathology, biochemistry, and biophysics of alpha-synuclein aggregation". Journal of Neurochemistry. 103 (1): 17–37. doi:10.1111/j.1471-4159.2007.04764.x. PMID 17623039.
- ↑ Jao CC, Hegde BG, Chen J, Haworth IS, Langen R (December 2008). "Structure of membrane-bound alpha-synuclein from site-directed spin labeling and computational refinement". Proceedings of the National Academy of Sciences of the United States of America. 105 (50): 19666–71. doi:10.1073/pnas.0807826105. PMC 2605001. PMID 19066219.
- ↑ Zhu M, Li J, Fink AL (October 2003). "The association of alpha-synuclein with membranes affects bilayer structure, stability, and fibril formation". The Journal of Biological Chemistry. 278 (41): 40186–97. doi:10.1074/jbc.M305326200. PMID 12885775.
- ↑ Madine J, Doig AJ, Middleton DA (May 2006). "A study of the regional effects of alpha-synuclein on the organization and stability of phospholipid bilayers". Biochemistry. 45 (18): 5783–92. doi:10.1021/bi052151q. PMID 16669622.
- ↑ Varkey J, Isas JM, Mizuno N, Jensen MB, Bhatia VK, Jao CC, Petrlova J, Voss JC, Stamou DG, Steven AC, Langen R (October 2010). "Membrane curvature induction and tubulation are common features of synucleins and apolipoproteins". The Journal of Biological Chemistry. 285 (42): 32486–93. doi:10.1074/jbc.M110.139576. PMC 2952250. PMID 20693280.
- ↑ Mizuno N, Varkey J, Kegulian NC, Hegde BG, Cheng N, Langen R, Steven AC (August 2012). "Remodeling of lipid vesicles into cylindrical micelles by α-synuclein in an extended α-helical conformation". The Journal of Biological Chemistry. 287 (35): 29301–11. doi:10.1074/jbc.M112.365817. PMC 3436199. PMID 22767608.
- ↑ Varkey J, Mizuno N, Hegde BG, Cheng N, Steven AC, Langen R (June 2013). "α-Synuclein oligomers with broken helical conformation form lipoprotein nanoparticles". The Journal of Biological Chemistry. 288 (24): 17620–30. doi:10.1074/jbc.M113.476697. PMC 3682563. PMID 23609437.
- ↑ 34.0 34.1 Varkey J, Langen R (July 2017). "Membrane remodeling by amyloidogenic and non-amyloidogenic proteins studied by EPR". Journal of Magnetic Resonance. 280: 127–139. doi:10.1016/j.jmr.2017.02.014. PMID 28579098.
- ↑ Zhu M, Qin ZJ, Hu D, Munishkina LA, Fink AL (July 2006). "Alpha-synuclein can function as an antioxidant preventing oxidation of unsaturated lipid in vesicles". Biochemistry. 45 (26): 8135–42. doi:10.1021/bi052584t. PMID 16800638.
- ↑ 36.0 36.1 Rawat A, Langen R, Varkey J (April 2018). "Membranes as modulators of amyloid protein misfolding and target of toxicity". Biochimica et Biophysica Acta. 1860 (9): 1863–1875. doi:10.1016/j.bbamem.2018.04.011. PMID 29702073.
- ↑ Flagmeier P, De S, Wirthensohn DC, Lee SF, Vincke C, Muyldermans S, Knowles TP, Gandhi S, Dobson CM, Klenerman D (June 2017). "2+ Influx into Lipid Vesicles Induced by Protein Aggregates". Angewandte Chemie. 56 (27): 7750–7754. doi:10.1002/anie.201700966. PMC 5615231. PMID 28474754.
- ↑ Sharon R, Bar-Joseph I, Frosch MP, Walsh DM, Hamilton JA, Selkoe DJ (February 2003). "The formation of highly soluble oligomers of alpha-synuclein is regulated by fatty acids and enhanced in Parkinson's disease". Neuron. 37 (4): 583–95. doi:10.1016/s0896-6273(03)00024-2. PMID 12597857.
- ↑ Amer DA, Irvine GB, El-Agnaf OM (August 2006). "Inhibitors of alpha-synuclein oligomerization and toxicity: a future therapeutic strategy for Parkinson's disease and related disorders". Experimental Brain Research. 173 (2): 223–33. doi:10.1007/s00221-006-0539-y. PMID 16733698.
- ↑ Ruipérez V, Darios F, Davletov B (October 2010). "Alpha-synuclein, lipids and Parkinson's disease". Progress in Lipid Research. 49 (4): 420–8. doi:10.1016/j.plipres.2010.05.004. PMID 20580911.
- ↑ Angelova PR, Horrocks MH, Klenerman D, Gandhi S, Abramov AY, Shchepinov MS (May 2015). "Lipid peroxidation is essential for α-synuclein-induced cell death". Journal of Neurochemistry. 133 (4): 582–9. doi:10.1111/jnc.13024. PMC 4471127. PMID 25580849.
- ↑ Clayton DF, George JM (1998). "The synucleins: a family of proteins involved in synaptic function, plasticity, neurodegeneration and disease". Trends in Neurosciences. 21 (6): 249–254. doi:10.1016/S0166-2236(97)01213-7.
- ↑ Vlad C, Lindner K, Karreman C, Schildknecht S, Leist M, Tomczyk N, Rontree J, Langridge J, Danzer K, Ciossek T, Petre A, Gross ML, Hengerer B, Przybylski M (December 2011). "Autoproteolytic fragments are intermediates in the oligomerization/aggregation of the Parkinson's disease protein alpha-synuclein as revealed by ion mobility mass spectrometry". Chembiochem. 12 (18): 2740–4. doi:10.1002/cbic.201100569. PMC 3461308. PMID 22162214.
- ↑ Bartels T, Choi JG, Selkoe DJ (August 2011). "α-Synuclein occurs physiologically as a helically folded tetramer that resists aggregation". Nature. 477 (7362): 107–10. doi:10.1038/nature10324. PMC 3166366. PMID 21841800. Lay summary – Harvard Medical School New Focus.
- ↑ Dettmer U, Newman AJ, Luth ES, Bartels T, Selkoe D (March 2013). "In vivo cross-linking reveals principally oligomeric forms of α-synuclein and β-synuclein in neurons and non-neural cells". The Journal of Biological Chemistry. 288 (9): 6371–85. doi:10.1074/jbc.M112.403311. PMC 3585072. PMID 23319586.
- ↑ Westphal CH, Chandra SS (January 2013). "Monomeric synucleins generate membrane curvature". The Journal of Biological Chemistry. 288 (3): 1829–40. doi:10.1074/jbc.M112.418871. PMC 3548493. PMID 23184946.
- ↑ Trexler AJ, Rhoades E (May 2012). "N-Terminal acetylation is critical for forming α-helical oligomer of α-synuclein". Protein Science. 21 (5): 601–5. doi:10.1002/pro.2056. PMC 3403458. PMID 22407793.
- ↑ Fauvet B, Mbefo MK, Fares MB, Desobry C, Michael S, Ardah MT, Tsika E, Coune P, Prudent M, Lion N, Eliezer D, Moore DJ, Schneider B, Aebischer P, El-Agnaf OM, Masliah E, Lashuel HA (May 2012). "α-Synuclein in central nervous system and from erythrocytes, mammalian cells, and Escherichia coli exists predominantly as disordered monomer". The Journal of Biological Chemistry. 287 (19): 15345–64. doi:10.1074/jbc.M111.318949. PMC 3346117. PMID 22315227.
- ↑ Burré J, Vivona S, Diao J, Sharma M, Brunger AT, Südhof TC (June 2013). "Properties of native brain α-synuclein". Nature. 498 (7453): E4–6, discussion E6-7. doi:10.1038/nature12125. PMC 4255827. PMID 23765500.
- ↑ Theillet FX, Binolfi A, Bekei B, Martorana A, Rose HM, Stuiver M, Verzini S, Lorenz D, van Rossum M, Goldfarb D, Selenko P (February 2016). "Structural disorder of monomeric α-synuclein persists in mammalian cells". Nature. 530 (7588): 45–50. doi:10.1038/nature16531. PMID 26808899.
- ↑ Spillantini MG, Schmidt ML, Lee VM, Trojanowski JQ, Jakes R, Goedert M (August 1997). "Alpha-synuclein in Lewy bodies". Nature. 388 (6645): 839–40. doi:10.1038/42166. PMID 9278044.
- ↑ Mezey E, Dehejia A, Harta G, Papp MI, Polymeropoulos MH, Brownstein MJ (July 1998). "Alpha synuclein in neurodegenerative disorders: murderer or accomplice?". Nature Medicine. 4 (7): 755–7. doi:10.1038/nm0798-755. PMID 9662355.
- ↑ Arima K, Hirai S, Sunohara N, Aoto K, Izumiyama Y, Uéda K, Ikeda K, Kawai M (October 1999). "Cellular co-localization of phosphorylated tau- and NACP/alpha-synuclein-epitopes in lewy bodies in sporadic Parkinson's disease and in dementia with Lewy bodies". Brain Research. 843 (1–2): 53–61. doi:10.1016/S0006-8993(99)01848-X. PMID 10528110.
- ↑ Arima K, Mizutani T, Alim MA, Tonozuka-Uehara H, Izumiyama Y, Hirai S, Uéda K (August 2000). "NACP/alpha-synuclein and tau constitute two distinctive subsets of filaments in the same neuronal inclusions in brains from a family of parkinsonism and dementia with Lewy bodies: double-immunolabeling fluorescence and electron microscopic studies". Acta Neuropathologica. 100 (2): 115–21. doi:10.1007/s004010050002. PMID 10963357.
- ↑ Yokota O, Terada S, Ishizu H, Ujike H, Ishihara T, Nakashima H, Yasuda M, Kitamura Y, Uéda K, Checler F, Kuroda S (December 2002). "NACP/alpha-synuclein, NAC, and beta-amyloid pathology of familial Alzheimer's disease with the E184D presenilin-1 mutation: a clinicopathological study of two autopsy cases". Acta Neuropathologica. 104 (6): 637–48. doi:10.1007/s00401-002-0596-7. PMID 12410385.
- ↑ Kim HY, Heise H, Fernandez CO, Baldus M, Zweckstetter M (September 2007). "Correlation of amyloid fibril beta-structure with the unfolded state of alpha-synuclein". Chembiochem. 8 (14): 1671–4. doi:10.1002/cbic.200700366. PMID 17722123.
- ↑ Sandal M, Valle F, Tessari I, Mammi S, Bergantino E, Musiani F, Brucale M, Bubacco L, Samorì B (January 2008). "Conformational equilibria in monomeric alpha-synuclein at the single-molecule level". PLoS Biology. 6 (1): e6. doi:10.1371/journal.pbio.0060006. PMC 2174973. PMID 18198943.
- ↑ Schulz-Schaeffer WJ (August 2010). "The synaptic pathology of alpha-synuclein aggregation in dementia with Lewy bodies, Parkinson's disease and Parkinson's disease dementia". Acta Neuropathologica. 120 (2): 131–43. doi:10.1007/s00401-010-0711-0. PMC 2892607. PMID 20563819.
- ↑ Morshedi D, Aliakbari F (Spring 2012). "The Inhibitory Effects of Cuminaldehyde on Amyloid Fibrillation and Cytotoxicity of Alpha-synuclein". Modares Journal of Medical Sciences: Pathobiology. 15 (1): 45–60.
- ↑ Woulfe J, Hoogendoorn H, Tarnopolsky M, Muñoz DG (November 2000). "Monoclonal antibodies against Epstein-Barr virus cross-react with alpha-synuclein in human brain". Neurology. 55 (9): 1398–401. doi:10.1212/WNL.55.9.1398. PMID 11087792.
- ↑ Polymeropoulos MH, Lavedan C, Leroy E, Ide SE, Dehejia A, Dutra A, Pike B, Root H, Rubenstein J, Boyer R, Stenroos ES, Chandrasekharappa S, Athanassiadou A, Papapetropoulos T, Johnson WG, Lazzarini AM, Duvoisin RC, Di Iorio G, Golbe LI, Nussbaum RL (June 1997). "Mutation in the alpha-synuclein gene identified in families with Parkinson's disease". Science. 276 (5321): 2045–7. doi:10.1126/science.276.5321.2045. PMID 9197268.
- ↑ Krüger R, Kuhn W, Müller T, Woitalla D, Graeber M, Kösel S, Przuntek H, Epplen JT, Schöls L, Riess O (February 1998). "Ala30Pro mutation in the gene encoding alpha-synuclein in Parkinson's disease". Nature Genetics. 18 (2): 106–8. doi:10.1038/ng0298-106. PMID 9462735.
- ↑ Zarranz JJ, Alegre J, Gómez-Esteban JC, Lezcano E, Ros R, Ampuero I, Vidal L, Hoenicka J, Rodriguez O, Atarés B, Llorens V, Gomez Tortosa E, del Ser T, Muñoz DG, de Yebenes JG (February 2004). "The new mutation, E46K, of alpha-synuclein causes Parkinson and Lewy body dementia". Annals of Neurology. 55 (2): 164–73. doi:10.1002/ana.10795. PMID 14755719.
- ↑ Appel-Cresswell S, Vilarino-Guell C, Encarnacion M, Sherman H, Yu I, Shah B, Weir D, Thompson C, Szu-Tu C, Trinh J, Aasly JO, Rajput A, Rajput AH, Jon Stoessl A, Farrer MJ (June 2013). "Alpha-synuclein p.H50Q, a novel pathogenic mutation for Parkinson's disease". Movement Disorders. 28 (6): 811–3. doi:10.1002/mds.25421. PMID 23457019.
- ↑ Lesage S, Anheim M, Letournel F, Bousset L, Honoré A, Rozas N, Pieri L, Madiona K, Dürr A, Melki R, Verny C, Brice A (April 2013). "G51D α-synuclein mutation causes a novel parkinsonian-pyramidal syndrome". Annals of Neurology. 73 (4): 459–71. doi:10.1002/ana.23894. PMID 23526723.
- ↑ Flagmeier P, Meisl G, Vendruscolo M, Knowles TP, Dobson CM, Buell AK, Galvagnion C (September 2016). "Mutations associated with familial Parkinson's disease alter the initiation and amplification steps of α-synuclein aggregation". Proceedings of the National Academy of Sciences of the United States of America. 113 (37): 10328–33. doi:10.1073/pnas.1604645113. PMC 5027465. PMID 27573854.
- ↑ Singleton AB, Farrer M, Johnson J, Singleton A, Hague S, Kachergus J, Hulihan M, Peuralinna T, Dutra A, Nussbaum R, Lincoln S, Crawley A, Hanson M, Maraganore D, Adler C, Cookson MR, Muenter M, Baptista M, Miller D, Blancato J, Hardy J, Gwinn-Hardy K (October 2003). "alpha-Synuclein locus triplication causes Parkinson's disease". Science. 302 (5646): 841. doi:10.1126/science.1090278. PMID 14593171.
- ↑ Takeda A, Hashimoto M, Mallory M, Sundsumo M, Hansen L, Masliah E (March 2000). "C-terminal alpha-synuclein immunoreactivity in structures other than Lewy bodies in neurodegenerative disorders". Acta Neuropathologica. 99 (3): 296–304. doi:10.1007/PL00007441. PMID 10663973.
- ↑ Prusiner SB, Woerman AL, Mordes DA, Watts JC, Rampersaud R, Berry DB, Patel S, Oehler A, Lowe JK, Kravitz SN, Geschwind DH, Glidden DV, Halliday GM, Middleton LT, Gentleman SM, Grinberg LT, Giles K (September 2015). "Evidence for α-synuclein prions causing multiple system atrophy in humans with parkinsonism". Proceedings of the National Academy of Sciences of the United States of America. 112 (38): E5308–17. doi:10.1073/pnas.1514475112. PMC 4586853. PMID 26324905.
- ↑ Weiler N (31 August 2015). "New Type of Prion May Cause, Transmit Neurodegeneration".
- ↑ Rettner R (31 August 2015). "Another Fatal Brain Disease May Come from the Spread of 'Prion' Proteins". Wired Science.
- ↑ Cookson MR (February 2009). "alpha-Synuclein and neuronal cell death". Molecular Neurodegeneration. 4 (1): 9. doi:10.1186/1750-1326-4-9. PMC 2646729. PMID 19193223.
- ↑ Fujiwara H, Hasegawa M, Dohmae N, Kawashima A, Masliah E, Goldberg MS, Shen J, Takio K, Iwatsubo T (February 2002). "alpha-Synuclein is phosphorylated in synucleinopathy lesions". Nature Cell Biology. 4 (2): 160–4. doi:10.1038/ncb748. PMID 11813001.
- ↑ Wersinger C, Sidhu A (April 2003). "Attenuation of dopamine transporter activity by alpha-synuclein". Neuroscience Letters. 340 (3): 189–92. doi:10.1016/S0304-3940(03)00097-1. PMID 12672538.
- ↑ Lee FJ, Liu F, Pristupa ZB, Niznik HB (April 2001). "Direct binding and functional coupling of alpha-synuclein to the dopamine transporters accelerate dopamine-induced apoptosis". FASEB Journal. 15 (6): 916–26. doi:10.1096/fj.00-0334com. PMID 11292651.
- ↑ Choi P, Golts N, Snyder H, Chong M, Petrucelli L, Hardy J, Sparkman D, Cochran E, Lee JM, Wolozin B (September 2001). "Co-association of parkin and alpha-synuclein". NeuroReport. 12 (13): 2839–43. doi:10.1097/00001756-200109170-00017. PMID 11588587.
- ↑ Kawahara K, Hashimoto M, Bar-On P, Ho GJ, Crews L, Mizuno H, Rockenstein E, Imam SZ, Masliah E (March 2008). "alpha-Synuclein aggregates interfere with Parkin solubility and distribution: role in the pathogenesis of Parkinson disease". The Journal of Biological Chemistry. 283 (11): 6979–87. doi:10.1074/jbc.M710418200. PMID 18195004.
- ↑ Ahn BH, Rhim H, Kim SY, Sung YM, Lee MY, Choi JY, Wolozin B, Chang JS, Lee YH, Kwon TK, Chung KC, Yoon SH, Hahn SJ, Kim MS, Jo YH, Min DS (April 2002). "alpha-Synuclein interacts with phospholipase D isozymes and inhibits pervanadate-induced phospholipase D activation in human embryonic kidney-293 cells". The Journal of Biological Chemistry. 277 (14): 12334–42. doi:10.1074/jbc.M110414200. PMID 11821392.
- ↑ Neystat M, Rzhetskaya M, Kholodilov N, Burke RE (June 2002). "Analysis of synphilin-1 and synuclein interactions by yeast two-hybrid beta-galactosidase liquid assay". Neuroscience Letters. 325 (2): 119–23. doi:10.1016/S0304-3940(02)00253-7. PMID 12044636.
- ↑ Reed JC, Meister L, Tanaka S, Cuddy M, Yum S, Geyer C, Pleasure D (December 1991). "Differential expression of bcl2 protooncogene in neuroblastoma and other human tumor cell lines of neural origin". Cancer Research. 51 (24): 6529–38. PMID 1742726.
- ↑ Kawamata H, McLean PJ, Sharma N, Hyman BT (May 2001). "Interaction of alpha-synuclein and synphilin-1: effect of Parkinson's disease-associated mutations". Journal of Neurochemistry. 77 (3): 929–34. doi:10.1046/j.1471-4159.2001.00301.x. PMID 11331421.
- ↑ Engelender S, Kaminsky Z, Guo X, Sharp AH, Amaravi RK, Kleiderlein JJ, Margolis RL, Troncoso JC, Lanahan AA, Worley PF, Dawson VL, Dawson TM, Ross CA (May 1999). "Synphilin-1 associates with alpha-synuclein and promotes the formation of cytosolic inclusions". Nature Genetics. 22 (1): 110–4. doi:10.1038/8820. PMID 10319874.
- ↑ Jensen PH, Hager H, Nielsen MS, Hojrup P, Gliemann J, Jakes R (September 1999). "alpha-synuclein binds to Tau and stimulates the protein kinase A-catalyzed tau phosphorylation of serine residues 262 and 356". The Journal of Biological Chemistry. 274 (36): 25481–9. doi:10.1074/jbc.274.36.25481. PMID 10464279.
- ↑ Giasson BI, Lee VM, Trojanowski JQ (2003). "Interactions of amyloidogenic proteins". Neuromolecular Medicine. 4 (1–2): 49–58. doi:10.1385/NMM:4:1-2:49. PMID 14528052.
- ↑ Ono K, Takahashi R, Ikeda T, Yamada M (September 2012). "Cross-seeding effects of amyloid β-protein and α-synuclein". Journal of Neurochemistry. 122 (5): 883–90. doi:10.1111/j.1471-4159.2012.07847.x. PMID 22734715.
Further reading
- Blakeslee S (2002-05-27). "In Folding Proteins, Clues to Many Diseases -". New York Times.
- Polymeropoulos MH, Lavedan C, Leroy E, Ide SE, Dehejia A, Dutra A, Pike B, Root H, Rubenstein J, Boyer R, Stenroos ES, Chandrasekharappa S, Athanassiadou A, Papapetropoulos T, Johnson WG, Lazzarini AM, Duvoisin RC, Di Iorio G, Golbe LI, Nussbaum RL (June 1997). "Mutation in the alpha-synuclein gene identified in families with Parkinson's disease". Science. 276 (5321): 2045–7. doi:10.1126/science.276.5321.2045. PMID 9197268.
- Neumann M, Kahle PJ, Giasson BI, Ozmen L, Borroni E, Spooren W, Müller V, Odoy S, Fujiwara H, Hasegawa M, Iwatsubo T, Trojanowski JQ, Kretzschmar HA, Haass C (November 2002). "Misfolded proteinase K-resistant hyperphosphorylated alpha-synuclein in aged transgenic mice with locomotor deterioration and in human alpha-synucleinopathies". The Journal of Clinical Investigation. 110 (10): 1429–39. doi:10.1172/JCI15777. PMC 151810. PMID 12438441.
- George JM (2001). "The synucleins". Genome Biology. 3 (1): REVIEWS3002. doi:10.1186/gb-2001-3-1-reviews3002. PMC 150459. PMID 11806835.
- Lavedan C (September 1998). "The synuclein family". Genome Research. 8 (9): 871–80. doi:10.1101/gr.8.9.871. PMID 9750188.
- Ozawa T, Wakabayashi K, Oyanagi K (February 2002). "[Recent progress in the research of multiple system atrophy with special references to alpha-synuclein and suprachiasmatic nucleus]". No to Shinkei = Brain and Nerve. 54 (2): 111–7. PMID 11889756.
- Cole NB, Murphy DD (2002). "The cell biology of alpha-synuclein: a sticky problem?". Neuromolecular Medicine. 1 (2): 95–109. doi:10.1385/NMM:1:2:95. PMID 12025860.
- Iwatsubo T (June 2002). "[alpha-synuclein and Parkinson's disease]". Seikagaku. The Journal of Japanese Biochemical Society. 74 (6): 477–82. PMID 12138709.
- Trojanowski JQ, Lee VM (October 2002). "Parkinson's disease and related synucleinopathies are a new class of nervous system amyloidoses". Neurotoxicology. 23 (4–5): 457–60. doi:10.1016/S0161-813X(02)00065-7. PMID 12428717.
- Alves da Costa C (February 2003). "Recent advances on alpha-synuclein cell biology: functions and dysfunctions". Current Molecular Medicine. 3 (1): 17–24. doi:10.2174/1566524033361690. PMID 12558071.
- Ma QL, Chan P, Yoshii M, Uéda K (April 2003). "Alpha-synuclein aggregation and neurodegenerative diseases". Journal of Alzheimer's Disease. 5 (2): 139–48. PMID 12719631.
- Di Rosa G, Puzzo D, Sant'Angelo A, Trinchese F, Arancio O (October 2003). "Alpha-synuclein: between synaptic function and dysfunction". Histology and Histopathology. 18 (4): 1257–66. doi:10.14670/HH-18.1257. PMID 12973692.
- Baptista MJ, Cookson MR, Miller DW (February 2004). "Parkin and alpha-synuclein: opponent actions in the pathogenesis of Parkinson's disease". The Neuroscientist. 10 (1): 63–72. doi:10.1177/1073858403260392. PMID 14987449.
- Kim S, Seo JH, Suh YH (May 2004). "Alpha-synuclein, Parkinson's disease, and Alzheimer's disease". Parkinsonism & Related Disorders. 10 Suppl 1: S9–13. doi:10.1016/j.parkreldis.2003.11.005. PMID 15109581.
- Sidhu A, Wersinger C, Vernier P (May 2004). "alpha-Synuclein regulation of the dopaminergic transporter: a possible role in the pathogenesis of Parkinson's disease". FEBS Letters. 565 (1–3): 1–5. doi:10.1016/j.febslet.2004.03.063. PMID 15135042.
- Vekrellis K, Rideout HJ, Stefanis L (August 2004). "Neurobiology of alpha-synuclein". Molecular Neurobiology. 30 (1): 1–21. doi:10.1385/MN:30:1:001. PMID 15247485.
- Chiba-Falek O, Nussbaum RL (2004). "Regulation of alpha-synuclein expression: implications for Parkinson's disease". Cold Spring Harbor Symposia on Quantitative Biology. 68: 409–15. doi:10.1101/sqb.2003.68.409. PMID 15338643.
- Pankratz N, Foroud T (April 2004). "Genetics of Parkinson disease". NeuroRx. 1 (2): 235–42. doi:10.1602/neurorx.1.2.235. PMC 534935. PMID 15717024.
- Singleton AB (August 2005). "Altered alpha-synuclein homeostasis causing Parkinson's disease: the potential roles of dardarin". Trends in Neurosciences. 28 (8): 416–21. doi:10.1016/j.tins.2005.05.009. PMID 15955578.
- Yu S, Uéda K, Chan P (2005). "Alpha-synuclein and dopamine metabolism". Molecular Neurobiology. 31 (1–3): 243–54. doi:10.1385/MN:31:1-3:243. PMID 15953825.
- Lee HG, Zhu X, Takeda A, Perry G, Smith MA (July 2006). "Emerging evidence for the neuroprotective role of alpha-synuclein". Experimental Neurology. 200 (1): 1–7. doi:10.1016/j.expneurol.2006.04.024. PMID 16780837.
- Giorgi FS, Bandettini di Poggio A, Battaglia G, Pellegrini A, Murri L, Ruggieri S, Paparelli A, Fornai F (2006). "A short overview on the role of alpha-synuclein and proteasome in experimental models of Parkinson's disease". Journal of Neural Transmission. Supplementum. 70 (70): 105–9. doi:10.1007/978-3-211-45295-0_17. PMID 17017516.
External links
Media related to alpha synuclein at Wikimedia Commons
- alpha-Synuclein at the US National Library of Medicine Medical Subject Headings (MeSH)
- Human SNCA genome location and SNCA gene details page in the UCSC Genome Browser.
- Pages with broken file links
- Genes on human chromosome
- All articles with unsourced statements
- Articles with unsourced statements from March 2012
- Articles with invalid date parameter in template
- Articles needing additional references from November 2015
- All articles needing additional references
- Peripheral membrane proteins
- Lewy body dementia