Streptococcus pneumoniae
|
Streptococcus pneumoniae infection Microchapters |
| Streptococcus pneumoniae | ||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
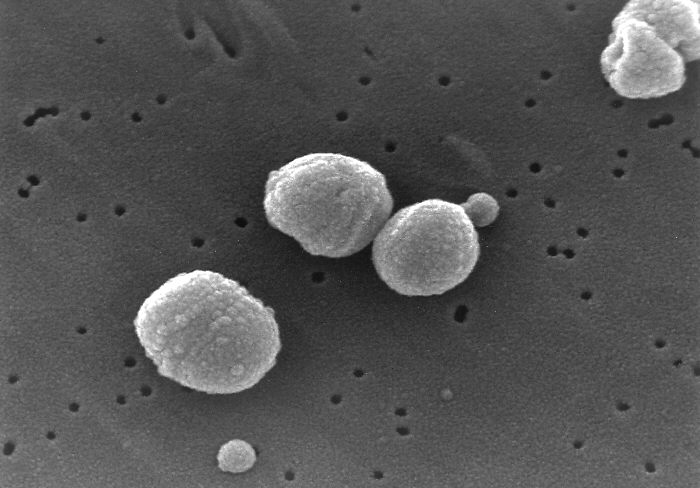 SEM micrograph of S. pneumoniae.
| ||||||||||||||
| Scientific classification | ||||||||||||||
| ||||||||||||||
| Binomial name | ||||||||||||||
| Streptococcus pneumoniae (Klein 1884) Chester 1901 |
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]
Overview
Streptococcus pneumoniae, or pneumococcus, is a Gram-positive, alpha-hemolytic, facultative anaerobic member of the genus Streptococcus.[1] A significant human pathogenic bacterium, S. pneumoniae was recognized as a major cause of pneumonia in the late 19th century, and is the subject of many humoral immunity studies.
S. pneumoniae resides asymptomatically in the nasopharynx of healthy carriers. The respiratory tract, sinuses, and nasal cavity are the parts of host body that are usually infected. However, in susceptible individuals, such as elderly and immunocompromised people and children, the bacterium may become pathogenic, spread to other locations and cause disease. S. pneumoniae is the main cause of community acquired pneumonia and meningitis in children and the elderly, and of septicemia in HIV-infected persons. The methods of transmission include sneezing, coughing, and direct contact with an infected person.
Despite the name, the organism causes many types of pneumococcal infections other than pneumonia. These invasive pneumococcal diseases include bronchitis, rhinitis, acute sinusitis, otitis media, conjunctivitis, meningitis, bacteremia, sepsis, osteomyelitis, septic arthritis, endocarditis, peritonitis, pericarditis, cellulitis, and brain abscess.[2]
S. pneumoniae is one of the most common causes of bacterial meningitis in adults and young adults, along with Neisseria meningitidis, and is the leading cause of bacterial meningitis in adults in the USA. It is also one of the top two isolates found in ear infection, otitis media.[3] Pneumococcal pneumonia is more common in the very young and the very old. It also is a major bacterium for invasive diseases like pneumonia and meningitis in South Asian children 12 years of age, though the evidence is of low quality and scarce.[4]
S. pneumoniae can be differentiated from Streptococcus viridans, some of which are also alpha-hemolytic, using an optochin test, as S. pneumoniae is optochin-sensitive. S. pneumoniae can also be distinguished based on its sensitivity to lysis by bile, the so-called "bile solubility test". The encapsulated, Gram-positive coccoid bacteria have a distinctive morphology on Gram stain, lancet-shaped diplococci. They have a polysaccharide capsule that acts as a virulence factor for the organism; more than 90 different serotypes are known, and these types differ in virulence, prevalence, and extent of drug resistance.
History
In 1881, the organism, known later in 1886 as the pneumococcus[5] for its role as an [etiologic agent] of pneumonia, was first isolated simultaneously and independently by the U.S. Army physician George Sternberg[6] and the French chemist Louis Pasteur.[7]
The organism was termed Diplococcus pneumoniae from 1920[8] because of its characteristic appearance in Gram-stained sputum. It was renamed Streptococcus pneumoniae in 1974 because it was very similar to streptococci.[5][9]
S. pneumoniae played a central role in demonstrating genetic material consists of DNA. In 1928, Frederick Griffith demonstrated transformation of life, turning harmless pneumococcus into a lethal form by co-inoculating the live pneumococci into a mouse along with heat-killed, virulent pneumococci.[10] In 1944, Oswald Avery, Colin MacLeod, and Maclyn McCarty demonstrated the transforming factor in Griffith's experiment was DNA, not protein, as was widely believed at the time.[11] Avery's work marked the birth of the molecular era of genetics.[12]
Virulence Factors
S. pneumoniae expresses different virulence factors on its cell surface and inside the organism. These virulence factors contribute to some of the clinical manifestations during infection with S. pneumoniae.
- Polysaccharide capsule -prevents phagocytosis by host immune cells by inhibiting C3b opsonization of the bacterial cells
- Pneumolysin (Ply) -a 53-kDa protein that can cause lysis of host cells and activate complement
- Autolysin (LytA) -activation of this protein lyses the bacteria releasing its internal contents (i.e. pneumolysin)
- Hydrogen peroxide - causes damage to host cells (can cause apoptosis in neuronal cells during meningitis) and has bactericidal effects against competing bacteria (Staphylococcus aureus)[13]
- Pili - hair-like structures that extend from the surface of many strains of S. pneumoniae. They contribute to colonization of upper respiratory tract and increase the formation of large amounts of TNF by the immune system during sepsis, raising the possibility of septic shock[14]
- Choline binding protein A (CbpA) -an adhesin that can interact with carbohydrates on the cell surface of pulmonary epithelial cells
- Protective Antigen (PspA) -can inhibit complement-mediated opsonization of pneumococci
Pathogenesis
S. pneumoniae is normally found in the nasopharynx of 5-10% of healthy adults, and 20-40% of healthy children. It can be found in higher amounts in certain environments, especially those where people are spending a great deal of time in close proximity to each other (day cares, army barracks). It attaches to nasopharyngeal cells through interaction of bacterial surface adhesins. This normal colonization can become infectious if the organisms are carried into areas such as the Eustachian tube or nasal sinuses where it can cause otitis media and sinusitis, respectively. Pneumonia occurs if the organisms are inhaled into the lungs and not cleared (again, viral infection, or smoking-induced ciliary paralysis might be contributing factors). Once the organism makes its way to a site where it is not normally found, it activates the complement protein group, stimulates cytokine production, and attracts white blood cells (specifically neutrophils). The organism's polysaccharide capsule makes it resistant to phagocytosis, and if there is no pre-existing anticapsular antibody, alveolar macrophages cannot adequately kill the pneumococci. The organism spreads to the blood stream (where it can cause bacteremia) and is carried to the meninges, joint spaces, bones, and peritoneal cavity, and may result in meningitis, brain abscess, septic arthritis, or osteomyelitis.
S. pneumoniae has several virulence factors, including the polysaccharide capsule mentioned earlier, that help it evade a host's immune system. It has pneumococcal surface proteins that inhibit complement-mediated opsonization, and it secretes IgA1 protease that will destroy secretory IgA produced by the body.
Humoral immunity
In the 19th century, it was demonstrated that immunization of rabbits with killed pneumococci protected them against subsequent challenge with viable pneumococci. Serum from immunized rabbits or from humans who had recovered from pneumococcal pneumonia also conferred protection. In the 20th century, the efficacy of immunization was demonstrated in South African miners.
It was discovered that the pneumococcus's capsule made it resistant to phagocytosis, and in the 1920s it was shown that an antibody specific for capsular polysaccharide aided the killing of S. pneumoniae. In 1936, a pneumococcal capsular polysaccharide vaccine was used to abort an epidemic of pneumococcal pneumonia. In the 1940s, experiments on capsular transformation by pneumococci first identified DNA as the material that carries genetic information.
In 1900, it was recognized that different serovars of pneumococci exist, and that immunization with a given serovar did not protect against infection with other serovars. Since then over ninety serovars have been discovered, each with a unique polysaccharide capsule which can be identified by the quellung reaction. Because some of these serovars cause disease more commonly than others, it is possible to provide reasonable protection by immunizing with less than 90 serovars; the current vaccine contains 23 serovars (i.e., it is "23-valent").
The serovars are numbered according to two systems: the American system, which numbers them in the order in which they were discovered, and the Danish system which groups them according to antigenic similarities.
Genetics
The genome of S. pneumoniae is a closed, circular DNA structure that contains between 2.0 and 2.1 million base pairs, depending on the strain. It has a core set of 1553 genes, plus 154 genes in its virulome, which contribute to virulence, and 176 genes that maintain a noninvasive phenotype. Genetic information can vary up to 10% between strains.[15]
Transformation in S. pneumoniae
Natural bacterial transformation involves the transfer of DNA from one bacterium to another through the surrounding medium. Transformation is a complex, developmental process requiring energy, dependent on expression of numerous genes. In S. pneumoniae at least 23 genes are required. In order for a bacterium to bind, take up and recombine exogenous DNA into its chromosome it must enter a special physiological state, called competence.
Competence, in S. pneumoniae, is induced by DNA-damaging agents such as mitomycin C, a DNA inter-strand cross-linking agent, and the fluoroquinolone antibiotics norfloxacin, levofloxacin and moxifloxacin, topoisomerase inhibitors that cause double-strand breaks.[16] Transformation protects S. pneumoniae against the bactericidal effect of mitomycin C.[17] Michod et al.[18] summarized evidence that induction of competence in S. pneumoniae is associated with increased resistance to oxidative stress and increased expression of the RecA protein, a key component of the recombinational repair machinery for removing DNA damages. On the basis of these findings, they suggested that transformation is an adaptation for repairing oxidative DNA damages. S. pneumoniae infection stimulates polymorphonuclear leukocytes (granulocyte) to produce an oxidative burst that is potentially lethal to the bacteria. The ability of S. pneumoniae to repair the oxidative DNA damages in its genome, caused by this host defense, likely contributes to this pathogen’s virulence.
Infection
S. pneumoniae is part of the normal upper respiratory tract flora, but, as with many natural flora, it can become pathogenic under the right conditions, like if the immune system of the host is suppressed. Invasins, such as pneumolysin, an anti-phagocytic capsule, various adhesins and immunogenic cell wall components are all major virulence factors.
Risk Factors
The risk of pneumococcal infection is much increased in persons with impaired IgG synthesis, impaired phagocytosis, or defective clearance of pneumococci. In particular, the absence of a functional spleen, through congenital asplenia, splenectomy, or sickle-cell disease predisposes one to a more severe course of infection (Overwhelming post-splenectomy infection) and prevention measures are indicated (see asplenia).
Vaccine
Interaction with Haemophilus influenzae
Both Haemophilus influenzae (H. influenzae) and S. pneumoniae can be found in the human upper respiratory system. A study of competition in vitro revealed S. pneumoniae overpowered H. influenzae by attacking it with hydrogen peroxide.[19]
When both bacteria are placed together into the nasal cavity of a mouse, within 2 weeks, only H. influenzae survives. When both are placed separately into a nasal cavity, each one survives. Upon examining the upper respiratory tissue from mice exposed to both bacteria, an extraordinarily large number of neutrophil immune cells were found. In mice exposed to only one bacterium, the cells were not present.
Lab tests show neutrophils that were exposed to already-dead H. influenzae were more aggressive in attacking S. pneumoniae than unexposed neutrophils. Exposure to killed H. influenzae had no effect on live H. influenzae.
Two scenarios may be responsible for this response:
- When H. influenzae is attacked by S. pneumoniae, it signals the immune system to attack the S. pneumoniae
- The combination of the two species sets off an immune system alarm that is not set off by either species individually.
It is unclear why H. influenzae is not affected by the immune system response.[20]
See also
References
- ↑ Ryan KJ; Ray CG (editors) (2004). Sherris Medical Microbiology. McGraw Hill. ISBN 0-8385-8529-9.
- ↑ Siemieniuk, Reed A.C. (Nov 2011). "The persisting burden of invasive pneumococcal disease in HIV patients: an observational cohort study". BMC Infectious Diseases. 11: 314. doi:10.1186/1471-2334-11-314. PMC 3226630. PMID 22078162. Unknown parameter
|coauthors=ignored (help) - ↑ Dagan R (2000). "Treatment of acute otitis media—challenges in the era of antibiotic resistance". Vaccine. 19 (Suppl 1): S9–S16. doi:10.1016/S0264-410X(00)00272-3. PMID 11163457.
- ↑ Jaiswal, Nishant; Singh, Meenu; Thumburu, Kiran Kumar; Bharti, Bhavneet; Agarwal, Amit; Kumar, Ajay; Kaur, Harpreet; Chadha, Neelima; et al. (5 May 2014). "Burden of Invasive Pneumococcal Disease in Children Aged 1 Month to 12 Years Living in South Asia: A Systematic Review". PLoS ONE. 9 (5): e96282. doi:10.1371/journal.pone.0096282.
- ↑ 5.0 5.1 Plotkin, SA; Orenstein, W; Offit, PA (September 22, 2012). Vaccines. Elsevier – Saunders. p. 542. ISBN 978-1455700905. Retrieved July 2, 2015.
- ↑ Sternberg, George Miller (30 April 1881). "A fatal form of septicaemia in the rabbit produced by the subcutaneous injection of human saliva. An experimental research". Bulletin of the National Board of Health. Baltimore, Maryland.
- ↑ Pasteur, Louis (1881). "Sur une maladie nouvelle provoquée par la salive d'un enfant mort de rage". Acad. D. Sc. De Paris. Paris, France. 92: 159.
- ↑ Winslow, C., and J. Broadhurst (1920). "The Families and Genera of the Bacteria: Final Report of the Committee of the Society of American Bacteriologists on Characterization and Classification of Bacterial Types". J Bacteriol. 5 (3): 191–229. PMC 378870. PMID 16558872.
- ↑ Wainer H (2014). Medical Illuminations: Using Evidence, Visualization and Statistical Thinking to Improve Healthcare. Oxford University Press. p. 53. ISBN 978-0199668793. Retrieved July 4, 2015.
- ↑ Griffith F (January 1928). "The Significance of Pneumococcal Types" (PDF). Journal of Hygiene. Cambridge University Press. 27 (2): 113–159. doi:10.1017/S0022172400031879. PMC 2167760. Retrieved July 3, 2015.
- ↑ Avery OT, MacLeod CM, and McCarty M (1944). "Studies on the chemical nature of the substance inducing transformation of pneumococcal types: induction of transformation by a desoxyribonucleic acid fraction isolated from pneumococcus type III". J Exp Med. 79 (2): 137–158. doi:10.1084/jem.79.2.137. PMC 2135445. PMID 19871359.
- ↑ Lederberg J (1994). "The Transformation of Genetics by DNA: An Anniversary Celebration of Avery, Macleod and Mccarty (1944)". Genetics. 136 (2): 423&ndash, 6. PMC 1205797. PMID 8150273.
- ↑ Pericone, Christopher D., Overweg, Karin, Hermans, Peter W. M., Weiser, Jeffrey N. (2000). "Inhibitory and Bactericidal Effects of Hydrogen Peroxide Production by Streptococcus pneumoniae on Other Inhabitants of the Upper Respiratory Tract". Infect Immun. 68 (7): 3990–3997. PMID 10858213.
- ↑ Barocchi M, Ries J, Zogaj X, Hemsley C, Albiger B, Kanth A, Dahlberg S, Fernebro J, Moschioni M, Masignani V, Hultenby K, Taddei A, Beiter K, Wartha F, von Euler A, Covacci A, Holden D, Normark S, Rappuoli R, Henriques-Normark B (2006). "A pneumococcal pilus influences virulence and host inflammatory responses". Proc Natl Acad Sci U S A. 103 (8): 2857–2862. PMID 16481624.
- ↑ van der Poll T, Opal SM (2009). "Pathogenesis, treatment, and prevention of pneumococcal pneumonia". Lancet. 374 (9700): 1543–56. doi:10.1016/S0140-6736(09)61114-4. PMID 19880020.
- ↑ Claverys JP, Prudhomme M, Martin B (2006). "Induction of competence regulons as a general response to stress in gram-positive bacteria". Annu. Rev. Microbiol. 60: 451–75. doi:10.1146/annurev.micro.60.080805.142139. PMID 16771651.
- ↑ Engelmoer DJ, Rozen DE (December 2011). "Competence increases survival during stress in Streptococcus pneumoniae". Evolution. 65 (12): 3475–85. doi:10.1111/j.1558-5646.2011.01402.x. PMID 22133219.
- ↑ Michod RE, Bernstein H, Nedelcu AM (May 2008). "Adaptive value of sex in microbial pathogens". Infect. Genet. Evol. 8 (3): 267–85. doi:10.1016/j.meegid.2008.01.002. PMID 18295550.http://www.hummingbirds.arizona.edu/Faculty/Michod/Downloads/IGE%20review%20sex.pdf
- ↑ Pericone, Christopher D., Overweg, Karin, Hermans, Peter W. M., Weiser, Jeffrey N. (2000). "Inhibitory and Bactericidal Effects of Hydrogen Peroxide Production by Streptococcus pneumoniae on Other Inhabitants of the Upper Respiratory Tract". Infect Immun. 68 (7): 3990&ndash, 3997. doi:10.1128/IAI.68.7.3990-3997.2000. PMC 101678. PMID 10858213.
- ↑ Lysenko ES, Ratner AJ, Nelson AL, Weiser JN (2005). "The Role of Innate Immune Responses in the Outcome of Interspecies Competition for Colonization of Mucosal Surfaces". PLoS Pathog. 1 (1): e1. doi:10.1371/journal.ppat.0010001. PMC 1238736. PMID 16201010. Full text
External links
- GAVI Alliance
- PneumoADIP
- PATH's Vaccine Resource Library pneumococcal resources
- Centers for Disease Control and Prevention (2012). "Ch. 16: Pneumococcal Disease". In Atkinson W, Wolfe S, Hamborsky J. Epidemiology and Prevention of Vaccine-Preventable Diseases (12th ed.). Washington DC: Public Health Foundation. pp. 233–248.
- CS1 maint: Extra text: authors list
- Pages with citations using unsupported parameters
- Articles with inconsistent citation formats
- CS1: Julian–Gregorian uncertainty
- CS1 maint: Multiple names: authors list
- CS1 maint: Multiple names: editors list
- Streptococcaceae
- Polysaccharide encapsulated bacteria
- Pneumonia
- Gram-positive bacteria