Tenecteplase
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]; Associate Editor(s)-in-Chief: Alejandro Lemor, M.D. [2]
WikiDoc MAKES NO GUARANTEE OF VALIDITY. WikiDoc is not a professional health care provider, nor is it a suitable replacement for a licensed healthcare provider. WikiDoc is intended to be an educational tool, not a tool for any form of healthcare delivery. The educational content on WikiDoc drug pages is based upon the FDA package insert, National Library of Medicine content and practice guidelines / consensus statements. WikiDoc does not promote the administration of any medication or device that is not consistent with its labeling. Please read our full disclaimer here.
Overview
Tenecteplase is a tissue plasminogen activator that is FDA approved for the {{{indicationType}}} of acute myocardial infarction. Common adverse reactions include hematoma, gastrointestinal bleeding, bleeding.
Adult Indications and Dosage
FDA-Labeled Indications and Dosage (Adult)
Acute Myocardial Infarction
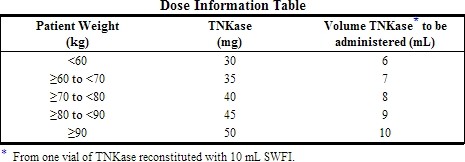
- The recommended total dose should not exceed 50 mg and is based upon patient weight.
- A single bolus dose should be administered over 5 seconds based on patient weight.
Off-Label Use and Dosage (Adult)
Guideline-Supported Use
There is limited information regarding Off-Label Guideline-Supported Use of Tenecteplase in adult patients.
Non–Guideline-Supported Use
There is limited information regarding Off-Label Non–Guideline-Supported Use of Tenecteplase in adult patients.
Pediatric Indications and Dosage
FDA-Labeled Indications and Dosage (Pediatric)
There is limited information regarding Tenecteplase FDA-Labeled Indications and Dosage (Pediatric) in the drug label.
Off-Label Use and Dosage (Pediatric)
Guideline-Supported Use
There is limited information regarding Off-Label Guideline-Supported Use of Tenecteplase in pediatric patients.
Non–Guideline-Supported Use
There is limited information regarding Off-Label Non–Guideline-Supported Use of Tenecteplase in pediatric patients.
Contraindications
Tenecteplase therapy in patients with acute myocardial infarction is contraindicated in the following situations because of an increased risk of bleeding:
- Active internal bleeding
- History of cerebrovascular accident
- Intracranial or intraspinal surgery or trauma within 2 months
- Intracranial neoplasm, arteriovenous malformation, or aneurysm
- Known bleeding diathesis
- Severe uncontrolled hypertension
Warnings
Bleeding
The most common complication encountered during Tenecteplase therapy is bleeding. The type of bleeding associated with thrombolytic therapy can be divided into two broad categories:
- Internal bleeding, involving intracranial and retroperitoneal sites, or the gastrointestinal, genitourinary, or respiratory tracts.
- Superficial or surface bleeding, observed mainly at vascular puncture and access sites (e.g., venous cutdowns, arterial punctures) or sites of recent surgical intervention.
Should serious bleeding (not controlled by local pressure) occur, any concomitant heparin or antiplatelet agents should be discontinued immediately.
In clinical studies of Tenecteplase, patients were treated with both aspirin and heparin. heparin may contribute to the bleeding risks associated with Tenecteplase. The safety of the use of Tenecteplase with other antiplatelet agents has not been adequately studied. Intramuscular injections and nonessential handling of the patient should be avoided for the first few hours following treatment with Tenecteplase. Venipunctures should be performed and monitored carefully.
Should an arterial puncture be necessary during the first few hours following Tenecteplase therapy, it is preferable to use an upper extremity vessel that is accessible to manual compression. Pressure should be applied for at least 30 minutes, a pressure dressing applied, and the puncture site checked frequently for evidence of bleeding.
Each patient being considered for therapy with Tenecteplase should be carefully evaluated and anticipated benefits weighed against potential risks associated with therapy. In the following conditions, the risk of Tenecteplase therapy may be increased and should be weighed against the anticipated benefits:
- Recent major surgery, e.g., coronary artery bypass graft, obstetrical delivery, organ biopsy, previous puncture of noncompressible vessels
- Cerebrovascular disease
- Recent gastrointestinal or genitourinary bleeding
- Recent trauma
- hypertension: systolic BP ≥180 mm Hg and/or diastolic BP ≥110 mm Hg
- High likelihood of left heart thrombus, e.g., mitral stenosis with atrial fibrillation
- Acute pericarditis
- Subacute bacterial endocarditis
- Hemostatic defects, including those secondary to severe hepatic or renal disease
- Severe hepatic dysfunction
- Pregnancy
- Diabetic hemorrhagic retinopathy or other hemorrhagic ophthalmic conditions
- Septic thrombophlebitis or occluded AV cannula at seriously infected site
- Advanced age
- Patients currently receiving oral anticoagulants, e.g., warfarin sodium
- Recent administration of GP IIb/IIIa inhibitors
- Any other condition in which bleeding constitutes a significant hazard or would be particularly difficult to manage because of its location
Cholesterol Embolization
Cholesterol embolism has been reported rarely in patients treated with all types of thrombolytic agents; the true incidence is unknown. This serious condition, which can be lethal, is also associated with invasive vascular procedures (e.g., cardiac catheterization, angiography, vascular surgery) and/or anticoagulant therapy. Clinical features of cholesterol embolism may include livedo reticularis, "purple toe" syndrome, acute renal failure, gangrenous digits, hypertension, pancreatitis, myocardial infarction, cerebral infarction, spinal cord infarction, retinal artery occlusion, bowel infarction, and rhabdomyolysis.
Arrhythmias
Coronary thrombolysis may result in arrhythmias associated with reperfusion. These arrhythmias (such as sinus bradycardia, accelerated idioventricular rhythm, ventricular premature depolarizations, ventricular tachycardia) are not different from those often seen in the ordinary course of acute myocardial infarction and may be managed with standard anti‑arrhythmic measures. It is recommended that anti‑arrhythmic therapy for bradycardia and/or ventricular irritability be available when Tenecteplase is administered.
Use with Percutaneous Coronary Intervention (PCI)
In patients with large ST segment elevation myocardial infarction, physicians should choose either thrombolysis or PCI as the primary treatment strategy for reperfusion. Rescue PCI or subsequent elective PCI may be performed after administration of thrombolytic therapies if medically appropriate; however, the optimal use of adjunctive antithrombotic and antiplatelet therapies in this setting is unknown.
Adverse Reactions
Clinical Trials Experience
Adverse Reactions
Bleeding
The most frequent adverse reaction associated with Tenecteplase is bleeding.
Should serious bleeding occur, concomitant heparin and antiplatelet therapy should be discontinued. Death or permanent disability can occur in patients who experience stroke or serious bleeding episodes.
For Tenecteplase-treated patients in ASSENT-2, the incidence of intracranial hemorrhage was 0.9% and any stroke was 1.8%. The incidence of all strokes, including intracranial bleeding, increases with increasing age.
In the ASSENT-2 study, the following bleeding events were reported (see Table 3).
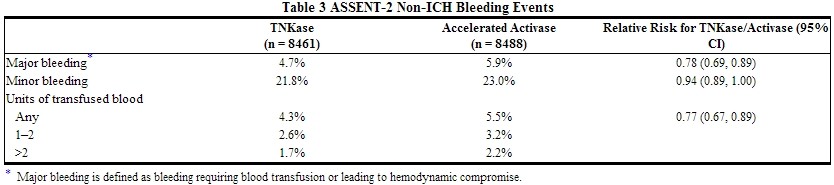
Non-intracranial major bleeding and the need for blood transfusions were lower in patients treated with Tenecteplase.
Types of major bleeding reported in 1% or more of the patients were hematoma (1.7%) and gastrointestinal tract (1%). Types of major bleeding reported in less than 1% of the patients were urinary tract, puncture site (including cardiac catheterization site), retroperitoneal, respiratory tract, and unspecified. Types of minor bleeding reported in 1% or more of the patients were hematoma (12.3%), urinary tract (3.7%), puncture site (including cardiac catheterization site) (3.6%), pharyngeal (3.1%), gastrointestinal tract (1.9%), epistaxis (1.5%), and unspecified (1.3%).
Allergic Reactions
Allergic-type reactions (e.g., anaphylaxis, angioedema, laryngeal edema, rash, and urticaria) have rarely (< 1%) been reported in patients treated with Tenecteplase. Anaphylaxis was reported in < 0.1% of patients treated with Tenecteplase; however, causality was not established. When such reactions occur, they usually respond to conventional therapy.
Other Adverse Reactions
The following adverse reactions have been reported among patients receiving Tenecteplase in clinical trials. These reactions are frequent sequelae of the underlying disease, and the effect of Tenecteplase on the incidence of these events is unknown.
These events include cardiogenic shock, arrhythmias, atrioventricular block, pulmonary edema, heart failure, cardiac arrest, recurrent myocardial ischemia, myocardial reinfarction, myocardial rupture, cardiac tamponade, pericarditis, pericardial effusion, mitral regurgitation, thrombosis, embolism, and electromechanical dissociation. These events can be life-threatening and may lead to death. Nausea and/or vomiting,hypotension, and fever have also been reported.
Postmarketing Experience
There is limited information regarding Tenecteplase Postmarketing Experience in the drug label.
Drug Interactions
Formal interaction studies of Tenecteplase with other drugs have not been performed. Patients studied in clinical trials of Tenecteplase were routinely treated with heparin and aspirin. Anticoagulants (such as heparin and vitamin K antagonists) and drugs that alter platelet function (such as acetylsalicylic acid, dipyridamole, and GP IIb/IIIa inhibitors) may increase the risk of bleeding if administered prior to, during, or after Tenecteplase therapy.
Use in Specific Populations
Pregnancy
Pregnancy Category (FDA): C
Tenecteplase has been shown to elicit maternal and embryo toxicity in rabbits given multiple IV administrations. In rabbits administered 0.5, 1.5, and 5.0 mg/kg/day, vaginal hemorrhageresulted in maternal deaths. Subsequent embryonic deaths were secondary to maternal hemorrhage and no fetal anomalies were observed. Tenecteplase does not elicit maternal and embryo toxicity in rabbits following a single IV administration. Thus, in developmental toxicity studies conducted in rabbits, the no observable effect level (NOEL) of a single IV administration of Tenecteplase on maternal or developmental toxicity was 5 mg/kg (approximately 8–10 times the human dose). There are no adequate and well‑controlled studies in pregnant women. Tenecteplase should be given to pregnant women only if the potential benefits justify the potential risk to the fetus.
Pregnancy Category (AUS):
There is no Australian Drug Evaluation Committee (ADEC) guidance on usage of Tenecteplase in women who are pregnant.
Labor and Delivery
There is no FDA guidance on use of Tenecteplase during labor and delivery.
Nursing Mothers
It is not known if Tenecteplase is excreted in human milk. Because many drugs are excreted in human milk, caution should be exercised when Tenecteplase is administered to a nursing woman.
Pediatric Use
The safety and effectiveness of Tenecteplase in pediatric patients have not been established.
Geriatic Use
Of the patients in ASSENT-2 who received Tenecteplase, 4,958 (59%) were under the age of 65; 2,256 (27%) were between the ages of 65 and 74; and 1,244 (15%) were 75 and over. The 30-day mortality rates by age were 2.5% in patients under the age of 65, 8.5% in patients between the ages of 65 and 74, and 16.2% in patients age 75 and over. The ICH rates were 0.4% in patients under the age of 65, 1.6% in patients between the ages of 65 and 74, and 1.7% in patients age 75 and over. The rates of any stroke were 1.0% in patients under the age of 65, 2.9% in patients between the ages of 65 and 74, and 3.0% in patients age 75 and over. Major bleeding rates, defined as bleeding requiring blood transfusion or leading to hemodynamic compromise, were 3.1% in patients under the age of 65, 6.4% in patients between the ages of 65 and 74, and 7.7% in patients age 75 and over. In elderly patients, the benefits of Tenecteplase on mortality should be carefully weighed against the risk of increased adverse events, including bleeding.
Gender
There is no FDA guidance on the use of Tenecteplase with respect to specific gender populations.
Race
There is no FDA guidance on the use of Tenecteplase with respect to specific racial populations.
Renal Impairment
There is no FDA guidance on the use of Tenecteplase in patients with renal impairment.
Hepatic Impairment
There is no FDA guidance on the use of Tenecteplase in patients with hepatic impairment.
Females of Reproductive Potential and Males
There is no FDA guidance on the use of Tenecteplase in women of reproductive potentials and males.
Immunocompromised Patients
There is no FDA guidance one the use of Tenecteplase in patients who are immunocompromised.
Administration and Monitoring
Administration
1.The product should be visually inspected prior to administration for particulate matter and discoloration. Tenecteplase may be administered as reconstituted at 5 mg/mL.
2.Precipitation may occur when Tenecteplase is administered in an IV line containing dextrose. Dextrose-containing lines should be flushed with a saline-containing solution prior to and following single bolus administration of Tenecteplase.
3.Reconstituted Tenecteplase should be administered as a single IV bolus over 5 seconds.
4.Because Tenecteplase contains no antibacterial preservatives, it should be reconstituted immediately before use. If the reconstituted Tenecteplase is not used immediately, refrigerate the Tenecteplase vial at 2–8°C (36–46°F) and use within 8 hours.
5.Although the supplied syringe is compatible with a conventional needle, this syringe is designed to be used with needleless IV systems. From the information below, follow the instructions applicable to the IV system in use.

6.Dispose of the syringe, cannula and shield per established procedures.
The safety and efficacy of Tenecteplase have only been investigated with concomitant administration of heparin and aspirin.
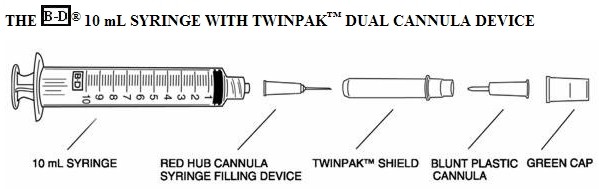
Reconstitution
NOTE: Read all instructions completely before beginning reconstitution and administration.
1.Remove the shield assembly from the supplied B-D® 10 mL syringe with TwinPak™ Dual Cannula Device (see figure) and aseptically withdraw 10 mL of Sterile Water for Injection (SWFI), USP, from the supplied diluent vial using the red hub cannula syringe filling device. Do not use Bacteriostatic Water for Injection, USP.
Note: Do not discard the shield assembly.
2.Inject the entire contents of the syringe (10 mL) into the Tenecteplase vial directing the diluent stream into the powder. Slight foaming upon reconstitution is not unusual; any large bubbles will dissipate if the product is allowed to stand undisturbed for several minutes.
3.Gently swirl until contents are completely dissolved. DO NOT SHAKE. The reconstituted preparation results in a colorless to pale yellow transparent solution containing Tenecteplase at 5 mg/mL at a pH of approximately 7.3. The osmolality of this solution is approximately 290 mOsm/kg.
4.Determine the appropriate dose of Tenecteplase (see Dose Information Table) and withdraw this volume (in milliliters) from the reconstituted vial with the syringe. Any unused solution should be discarded.
5.Once the appropriate dose of Tenecteplase is drawn into the syringe, stand the shield vertically on a flat surface (with green side down) and passively recap the red hub cannula.
6.Remove the entire shield assembly, including the red hub cannula, by twisting counterclockwise. Note: The shield assembly also contains the clear-ended blunt plastic cannula; retain for split septum IV access.
Readministration
Readministration of plasminogen activators, including tenecteplase, to patients who have received prior plasminogen activator therapy has not been systematically studied. Three of 487 patients tested for antibody formation to tenecteplase had a positive antibody titer at 30 days. The data reflect the percentage of patients whose test results were considered positive for antibodies to tenecteplase in a radioimmunoprecipitation assay, and are highly dependent on the sensitivity and specificity of the assay. Additionally, the observed incidence of antibody positivity in an assay may be influenced by several factors including sample handling, concomitant medications, and underlying disease. For these reasons, comparison of the incidence of antibodies to tenecteplase with the incidence of antibodies to other products may be misleading. Although sustained antibody formation in patients receiving one dose of tenecteplase has not been documented, readministration should be undertaken with caution. If an anaphylactic reaction occurs, appropriate therapy should be administered.
Monitoring
There is limited information regarding Monitoring of Tenecteplase in the drug label.
IV Compatibility
There is limited information regarding the compatibility of Tenecteplase and IV administrations.
Overdosage
There is limited information regarding Tenecteplase overdosage. If you suspect drug poisoning or overdose, please contact the National Poison Help hotline (1-800-222-1222) immediately.
Pharmacology
Tenecteplase
| |
| Systematic (IUPAC) name | |
| Human tissue plasminogen activator | |
| Identifiers | |
| CAS number | |
| ATC code | B01 |
| PubChem | ? |
| DrugBank | |
| Chemical data | |
| Formula | Template:OrganicBox atomTemplate:OrganicBox atomTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBox atomTemplate:OrganicBoxTemplate:OrganicBox atomTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBox atomTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBox |
| Mol. mass | 58951.2 g/mol |
| Pharmacokinetic data | |
| Bioavailability | ? |
| Metabolism | ? |
| Half life | ? |
| Excretion | Liver |
| Therapeutic considerations | |
| Pregnancy cat. |
? |
| Legal status |
Template:Unicode Prescription only |
| Routes | ? |
Mechanism of Action
Tenecteplase is a modified form of human tissue plasminogen activator (tPA) that binds to fibrin and converts plasminogen to plasmin. In the presence of fibrin, in vitro studies demonstrate that Tenecteplase conversion of plasminogen to plasmin is increased relative to its conversion in the absence of fibrin. This fibrin specificity decreases systemic activation of plasminogen and the resulting degradation of circulating fibrinogen as compared to a molecule lacking this property. Following administration of 30, 40, or 50 mg of Tenecteplase, there are decreases in circulating fibrinogen (4%–15%) and plasminogen (11%–24%). The clinical significance of fibrin-specificity on safety (e.g., bleeding) or efficacy has not been established. Biological potency is determined by an in vitro clot lysis assay and is expressed in Tenecteplase-specific units. The specific activity of Tenecteplase has been defined as 200 units/mg.
Structure
Tenecteplase is a tissue plasminogen activator (tPA) produced by recombinant DNA technology using an established mammalian cell line (Chinese Hamster Ovary cells). Tenecteplase is a 527 amino acid glycoprotein developed by introducing the following modifications to the complementary DNA (cDNA) for natural human tPA: a substitution of threonine 103 with asparagine, and a substitution of asparagine 117 with glutamine, both within the kringle 1 domain, and a tetra-alanine substitution at amino acids 296–299 in the protease domain.
Pharmacodynamics
In patients with acute myocardial infarction (AMI), Tenecteplase administered as a single bolus exhibits a biphasic disposition from the plasma. Tenecteplase was cleared from the plasma with an initial half-life of 20 to 24 minutes. The terminal phase half-life of Tenecteplase was 90 to 130 minutes. In 99 of 104 patients treated with Tenecteplase, mean plasma clearance ranged from 99 to 119 mL/min.
The initial volume of distribution is weight related and approximates plasma volume. Liver metabolism is the major clearance mechanism for Tenecteplase
Pharmacokinetics
There is limited information regarding Pharmacokinetics of Tenecteplase in the drug label.
Nonclinical Toxicology
Carcinogenesis, Mutagenesis, Impairment of Fertility
Studies in animals have not been performed to evaluate the carcinogenic potential, mutagenicity, or the effect on fertility.
Clinical Studies
ASSENT-2 was an international, randomized, double-blind trial that compared 30-day mortality rates in 16,949 patients assigned to receive an IV bolus dose of Tenecteplase or an accelerated infusion of Activase® (alteplase). Eligibility criteria included onset of chest pain within 6 hours of randomization and ST-segment elevation or left bundle branch block on electrocardiogram (ECG). Patients were to be excluded from the trial if they received GP IIb/IIIainhibitors within the previous 12 hours. Tenecteplase was dosed using actual or estimated weight in a weight-tiered fashion. All patients were to receive 150–325 mg of aspirin administered as soon as possible, followed by 150–325 mg daily. Intravenous heparin was to be administered as soon as possible: for patients weighing ≤67 kg, heparin was administered as a 4000 unit IV bolus followed by infusion at 800 U/hr; for patients weighing >67 kg, heparin was administered as a 5000 unit IV bolus followed by infusion at 1000 U/hr. heparin was continued for 48 to 72 hours with infusion adjusted to maintain aPTT at 50–75 seconds. The use of GP IIb/IIIa inhibitors was discouraged for the first 24 hours following randomization. The results of the primary endpoint (30-day mortality rates with non-parametric adjustment for the covariates of age, Killip class, heart rate, systolic blood pressure and infarct location) along with selected other 30-day endpoints are shown in Table 1.
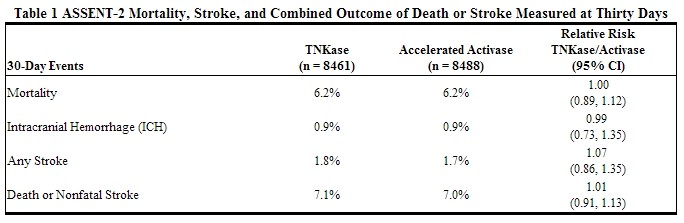
Rates of mortality and the combined endpoint of death or stroke among pre-specified subgroups, including age, gender, time to treatment, infarct location, and history of previousmyocardial infarction, demonstrate consistent relative risks across these subgroups. There was insufficient enrollment of non-Caucasian patients to draw any conclusions regarding relative efficacy in racial subsets.
Rates of in-hospital procedures, including percutaneous transluminal coronary angioplasty (PTCA), stent placement, intra-aortic balloon pump(IABP) use, and coronary artery bypass graft (CABG) surgery, were similar between the Tenecteplase and Activase®(Alteplase) groups.
TIMI 10B was an open-label, controlled, randomized, dose-ranging, angiography study which utilized a blinded core laboratory for review of coronary arteriograms. Patients (n = 837) presenting within 12 hours of symptom onset were treated with fixed doses of 30, 40, or 50 mg of Tenecteplase or the accelerated infusion of activase and underwent coronary arteriography at 90 minutes. The results showed that the 40 mg and 50 mg doses were similar to accelerated infusion of activase in restoring patency. TIMI Grade 3 flow and TIMI Grade 2/3 flow at 90 minutes are shown in Table 2. The exact relationship between coronary artery patency and clinical activity has not been established.
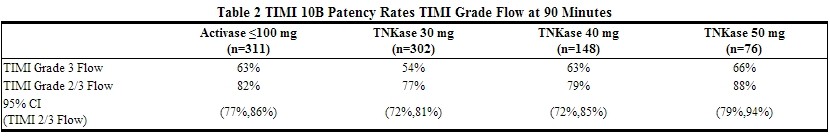
The angiographic results from TIMI 10B and the safety data from ASSENT-1, an additional uncontrolled safety study of 3,235 Tenecteplase-treated patients, provided the framework to develop a weight-tiered Tenecteplase dose regimen.3 Exploratory analyses suggested that a weight-adjusted dose of 0.5 mg/kg to 0.6 mg/kg of Tenecteplase resulted in a better patency to bleeding relationship than fixed doses of Tenecteplase across a broad range of patient weights.
The Assessment of the Safety and Efficacy of a New Treatment Strategy with Percutaneous Coronary Intervention (ASSENT 4 PCI) was a Phase IIIb/IV study designed to assess the safety and effectiveness of a strategy of administering full dose Tenecteplase with a single bolus of 4000 U of unfractionated heparin in patients with ST segment elevation AMI, in whom primary percutaneous coronary intervention (PCI) was planned, but in whom a delay of 1–3 hours was anticipated before PCI. The trial was prematurely terminated with 1667 randomized patients (75 of whom were treated in the United States) due to a numerically higher mortality in the patients receiving Tenecteplase prior to primary PCI versus PCI without Tenecteplase (median time from randomization to balloon of 115 minutes). The incidence of the 90‑day primary endpoint, a composite of death or cardiogenic shock or congestive heart failure (CHF) within 90 days, was 18.6% in patients treated with Tenecteplase plus PCI versus 13.4% in those treated with PCI alone (p=0.0055; OR 1.39 (95% C.I. 1.11–1.74)).
There were trends toward worse outcomes in the individual components of the primary endpoint between Tenecteplase plus PCI versus PCI alone (mortality 6.7% vs. 5.0%, respectively; cardiogenic shock 6.1% vs. 4.8%, respectively; and CHF 12.1% vs. 9.4%, respectively). In addition, there were trends towards worse outcomes in recurrent MI (6.1% vs. 3.5%, respectively; p=0.03) and repeat target vessel revascularization (6.6% vs. 3.6%, respectively; p=0.005) in patients receiving Tenecteplase plus PCI versus PCI alone.
There was no difference in in‑hospital major bleeding between the two groups (5.6% vs. 4.4% for Tenecteplase plus PCI vs. PCI alone, respectively). For patients treated with Tenecteplase plus PCI, in‑hospital rates of intracranial hemorrhage and total stroke were similar to those observed in previous trials (0.97% and 1.8%, respectively); however, none of the patients treated with PCI alone experienced a stroke (ischemic, hemorrhagic or other).
How Supplied
Tenecteplase is supplied as a sterile, lyophilized powder in a 50 mg vial under partial vacuum. Each 50 mg vial of Tenecteplase is packaged with one 10 mL vial of Sterile Water for Injection, USP for reconstitution, the B-D® 10 mL syringe with TwinPak™ Dual Cannula Device, and three alcohol prep pads. NDC 50242-038-61.
Storage
Store lyophilized Tenecteplase at controlled room temperature not to exceed 30°C (86°F) or under refrigeration 2–8°C (36–46°F). Do not use beyond the expiration date stamped on the vial.
Images
Drug Images
{{#ask: Page Name::Tenecteplase |?Pill Name |?Drug Name |?Pill Ingred |?Pill Imprint |?Pill Dosage |?Pill Color |?Pill Shape |?Pill Size (mm) |?Pill Scoring |?NDC |?Drug Author |format=template |template=DrugPageImages |mainlabel=- |sort=Pill Name }}
Package and Label Display Panel
{{#ask: Label Page::Tenecteplase |?Label Name |format=template |template=DrugLabelImages |mainlabel=- |sort=Label Page }}
Patient Counseling Information
There is limited information regarding Patient Counseling Information of Tenecteplase in the drug label.
Precautions with Alcohol
Alcohol-Tenecteplase interaction has not been established. Talk to your doctor about the effects of taking alcohol with this medication.
Brand Names
TNKase®
Look-Alike Drug Names
There is limited information regarding Tenecteplase Look-Alike Drug Names in the drug label.
Price
References
The contents of this FDA label are provided by the National Library of Medicine.
{{#subobject:
|Label Page=Tenecteplase |Label Name=Tenecteplase07.jpg
}}