NADH dehydrogenase
Overview


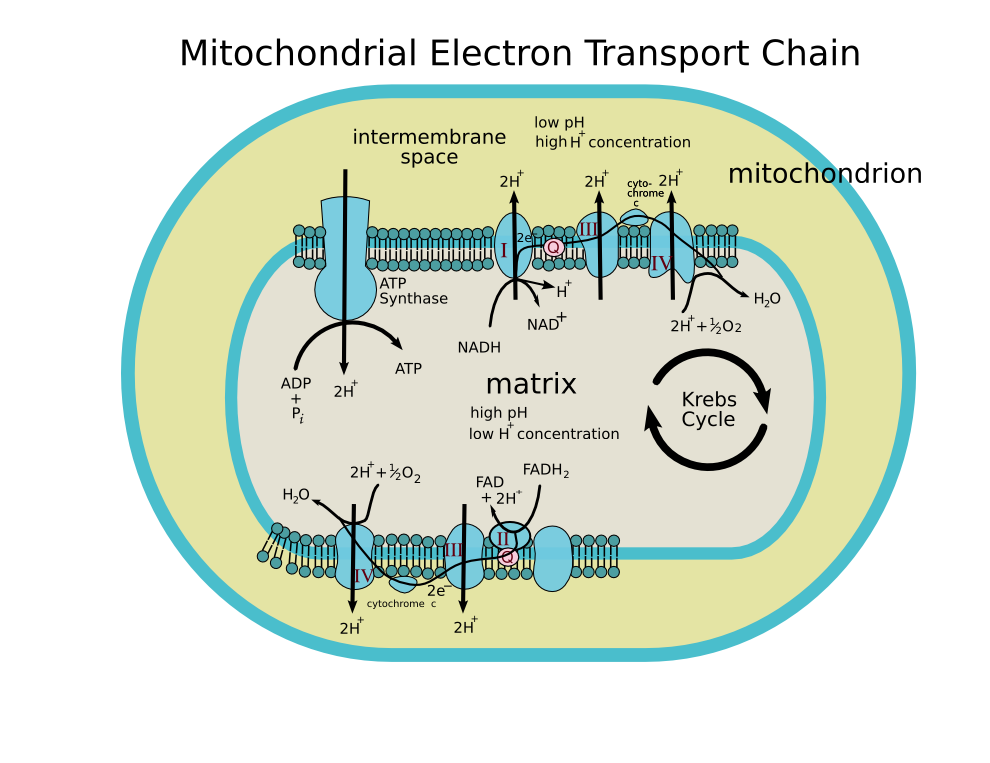
NADH dehydrogenase (EC 1.6.5.3) is an enzyme located in the inner mitochodrial membrane that catalyzes the transfer of electrons from NADH to coenzyme Q (CoQ). It is also called the NADH:quinone oxidoreductase.
Reaction
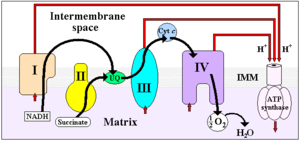
It is the first enzyme (complex I) of the mitochondrial electron transport chain.
- NADH + CoQ + 5H+ → NAD+ + CoQH2 + 4H+
In this process, the complex translocates protons across the inner membrane, helping to build the electrochemical potential used to produce ATP. The exact catalytic mechanism remains unknown.
Composition and structure
It is the largest of the respiratory complexes, the mammalian enzyme containing 42 separate polypeptide chains. Of particular functional importance are the flavin prosthetic group and eight iron-sulfur clusters. Of the 42 subunits, seven are encoded by the mitochondrial genome [1].
The structure is a L shape with a long membrane domain (with around 60 trans-membrane helices) and a hydrophilic peripheral domain which includes the NAD reducing activity. While the structure of the eukaryotic complex is not well characterised the peripheral/hydrophilic domain of the complex from a bacterium (Thermus thermophilus) has been crystallised (PDB: 2FUG) [2].
Inhibitor
The best known inhibitor of complex I is Rotenone (used as an organic pesticide). It is thought to bind to the ubiquinone binding site.
Piericidin A is a more potent inhibitor and is a close structural homologue of ubiquinone.
Pathology
Mutations in the subunits of complex I can cause mitochondrial diseases, including Leigh syndrome.
There is some evidence that complex I defects may play a role in the etiology of Parkinson's disease, perhaps because of reactive oxygen species (complex I can, like complex II, leak electron to oxygen, forming highly toxic superoxide). In fact, recent investigations suggest that reverse electron transfer through Complex I might be the most important site of superoxide production within mitochondria.
Genes
- NDUFS1 - NADH dehydrogenase (ubiquinone) Fe-S protein 1, 75kDa (NADH-coenzyme Q reductase)
- NDUFS2 - NADH dehydrogenase (ubiquinone) Fe-S protein 2, 49kDa (NADH-coenzyme Q reductase)
- NDUFV1 - NADH dehydrogenase (ubiquinone) flavoprotein 1, 51kDa
- NDUFV2 - NADH dehydrogenase (ubiquinone) flavoprotein 2, 24kDa
- NDUFV3 - NADH dehydrogenase (ubiquinone) flavoprotein 3, 10kDa
- MT-ND4 - mitochondrially encoded NADH dehydrogenase 4
References
Additional images
-
ETC
External links
- Interactive Molecular model of NADH dehydrogenase (Requires MDL Chime)
- Complex I home page at The Scripps Research Institute
- Electron+Transport+Complex+I at the US National Library of Medicine Medical Subject Headings (MeSH)
- NADH+Dehydrogenase at the US National Library of Medicine Medical Subject Headings (MeSH)