G protein
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]
Overview
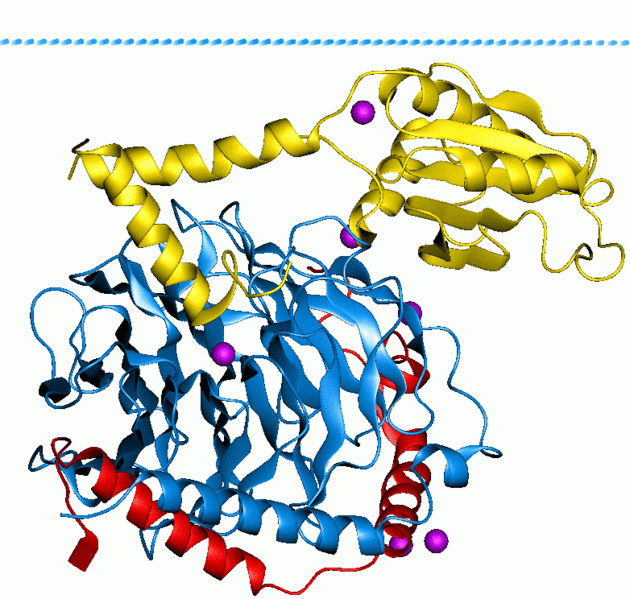
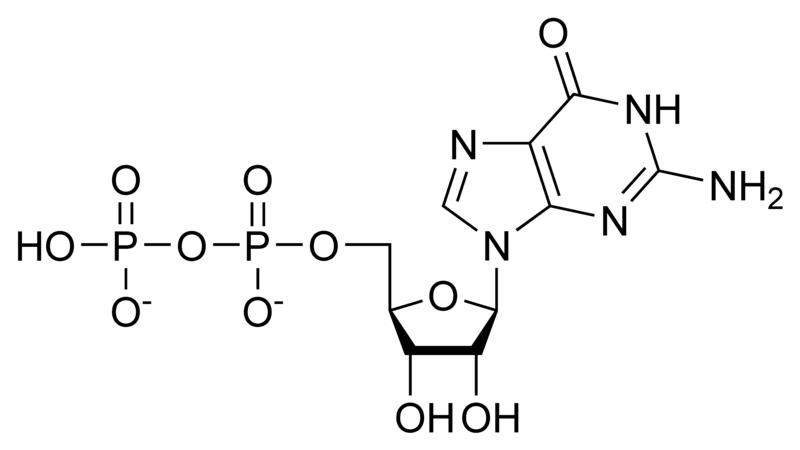
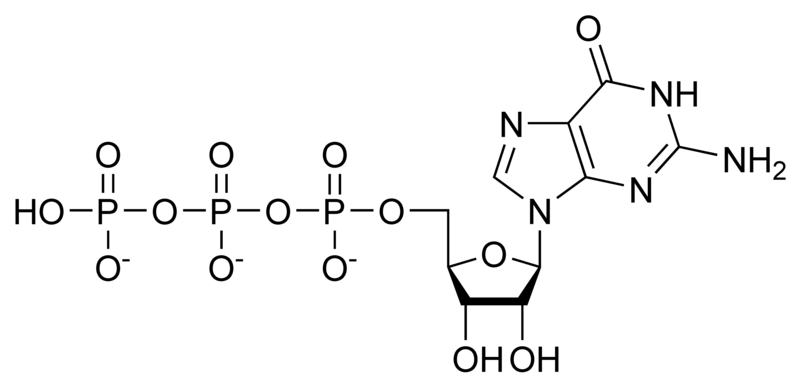
G proteins, short for guanine nucleotide binding proteins, are a family of proteins involved in second messenger cascades. Their nomenclature originates from their ability to function as "molecular switches", alternating between an inactive guanosine diphosphate (GDP) and activated guanosine triphosphate (GTP) bound state, ultimately going on to regulate downstream cell processes.
G proteins belong to the larger group of enzymes called GTPases.
History
Alfred Gilman and Martin Rodbell were awarded the Nobel Prize in Physiology or Medicine in 1994 for their discovery of and research on G proteins.
Function
G proteins are important signal transducing molecules in cells. In fact, diseases such as diabetes and certain forms of cancer, among other pathologies, are thought to arise due to derangement of G protein signaling.
Types of G protein signaling
G protein can refer to two distinct families of proteins. Heterotrimeric G proteins, sometimes referred to as the "large" G proteins that are activated by G protein-coupled receptors and made up of alpha (α), beta (β) and gamma (γ) subunits. There are also "small" G proteins (20-25kDa) that belong to the Ras superfamily of small GTPases. These proteins are homologous to the alpha (α) subunit found in heterotrimers, and are in fact monomeric. However, they also bind GTP and GDP and are involved in signal transduction.
Heterotrimeric G proteins
Heterotrimeric G proteins share a common mode of action, i.e. activation in response to a conformation change in the G-protein coupled receptor, exchange of GTP for GDP and dissociation in order to activate further proteins in the signal transduction pathway. However, the specific mechanism differs between different types of G proteins.
Common mechanism
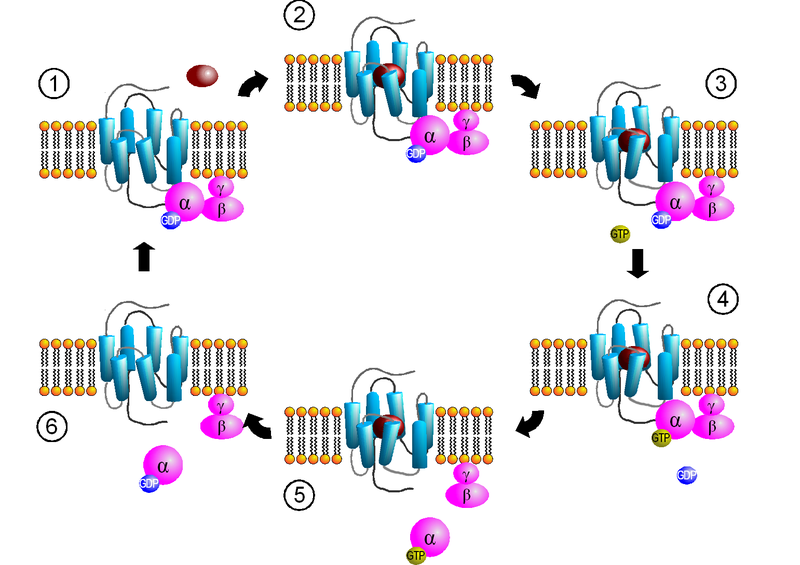
Receptor activated G proteins are bound to the inside surface of the cell membrane. They consist of the Gα and the tightly associated Gβγ subunits. Presently, four main families exist for Gα subunits: Gαs, Gαi, Gαq/11, and Gα12/13. These groups differ primarily in effector recognition, but share a similar mechanism of activation.
Activation
When a ligand activates the G protein-coupled receptor, it induces a conformation change in the receptor (a change in shape) that allows the receptor to function as a guanine nucleotide exchange factor (GEF) that exchanges GTP for GDP on the Gα subunit. In the traditional view of heterotrimeric protein activation, this exchange triggers the dissociation of the Gα subunit from the Gβγ dimer and the receptor. However, models that suggest molecular rearrangement, reorganization and pre-complexing of effector molecules are beginning to be accepted. Regardless, both, Gα-GTP and Gβγ, can then activate different signaling cascades (or second messenger pathways) and effector proteins, while the receptor is able to activate the next G protein.
Termination
The Gα subunit will eventually hydrolyze the attached GTP to GDP by its inherent enzymatic activity, allowing it to re-associate with Gβγ and starting a new cycle. Although, there do exist groups of proteins called RBMs that act as GTPase activating proteins (GAPs) which are specific for Gα subunits, which act to accelerate hydrolysis and terminate the transduced signal. In some cases the effector itself may possess intrinsic GAP activity, which helps deactivate the pathway. This is true in the case of phospholipase C beta which possesses GAP activity within its C-terminal region. This is an alternate form of regulation for the Gα subunit.
Specific mechanisms
- Gαs stimulates the production of cAMP from ATP. This is accomplished by direct stimulation of the membrane associated enzyme adenylate cyclase. cAMP acts as a second messenger which goes on to interact with and activate protein kinase A (PKA). PKA can then phosphorylate a myriad of downstream targets.
- Gαi inhibits the production of cAMP from ATP.
- Gαq/11 stimulates membrane bound phospholipase C beta which then cleaves PIP2 (a minor membrane phosphoinositol) into two second messengers, IP3 and diacylglycerol (DAG).
- Gα12/13 are involved in Rho family GTPase signaling (through RhoGEF superfamily) and control cell cytoskeleton remodeling, thus regulating cell migration.
- Gβγ sometimes also have active functions, e.g. coupling to L-type calcium channels.
Small GTPases
Small GTPases also bind GTP and GDP and are involved in signal transduction. These proteins are homologous to the alpha (α) subunit found in heterotrimers, and are in fact monomeric. However, they also bind GTP and GDP and are involved in signal transduction. They are small (20-25 kDa) proteins that bind to guanosine triphosphate (GTP). This family of proteins is homologous to Ras GTPases and is also called the Ras superfamily GTPases.
Lipidation
In order to associate with the inner leaflet of the plasma membrane, many G proteins are covalently modified with lipid extensions, i.e. they are lipidated.
- Heterotrimeric G protein subunits may be myristolated, palmitoylated, or prenylated.
- Small G proteins may be prenylated.
References
- Eric R. Kandel, James H. Schwartz, Thomas M. Jessell (2000). Principles of neural science. New York: McGraw-Hill. ISBN 0-8385-7701-6.
- Lodish et al. 2000. Molecular Cell Biology 4th ed. W.H. Freeman and Company, New York.
- Voet, Donald and Judith G. Voet. 1995. Biochemistry 2nd ed. John Wilely & Sons, New York.
- Gilman A (1987). "G proteins: transducers of receptor-generated signals". Annu Rev Biochem. 56: 615–49. PMID 3113327.
- Neves S.R., Ram P.T., Iyengar R. (2002). "G Protein Pathways". Science. 296: 1636–9. PMID: 12040175[2].
ca:Proteïna G de:G-Protein it:Proteina G he:חלבון G nl:G-proteïne fi:G-proteiini sv:G-protein