Adrenalone
 | |
| Clinical data | |
|---|---|
| Pregnancy category |
|
| Routes of administration | Topical |
| ATC code | |
| Pharmacokinetic data | |
| Metabolism | MAO, COMT |
| Excretion | Renal |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEMBL | |
| E number | {{#property:P628}} |
| ECHA InfoCard | {{#property:P2566}}Lua error in Module:EditAtWikidata at line 36: attempt to index field 'wikibase' (a nil value). |
| Chemical and physical data | |
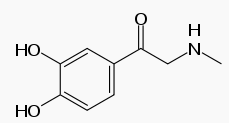
| Formula | C9H11NO3 |
| Molar mass | 181.189 g/mol |
| 3D model (JSmol) | |
| Melting point | 235 to 236 °C (Expression error: Unrecognized word "to". °F) (decomposes) |
| |
| |
| | |
|
WikiDoc Resources for Adrenalone |
|
Articles |
|---|
|
Most recent articles on Adrenalone |
|
Media |
|
Evidence Based Medicine |
|
Clinical Trials |
|
Ongoing Trials on Adrenalone at Clinical Trials.gov Clinical Trials on Adrenalone at Google
|
|
Guidelines / Policies / Govt |
|
US National Guidelines Clearinghouse on Adrenalone
|
|
Books |
|
News |
|
Commentary |
|
Definitions |
|
Patient Resources / Community |
|
Patient resources on Adrenalone Discussion groups on Adrenalone Patient Handouts on Adrenalone Directions to Hospitals Treating Adrenalone Risk calculators and risk factors for Adrenalone
|
|
Healthcare Provider Resources |
|
Causes & Risk Factors for Adrenalone |
|
Continuing Medical Education (CME) |
|
International |
|
|
|
Business |
|
Experimental / Informatics |
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]
Overview
Adrenalone is an adrenergic agonist used as a topical vasoconstrictor and hemostatic. Formerly, it was also used to prolong the action of local anesthetics. It is the ketone form of epinephrine (adrenaline). Contrary to epinephrine, adrenalone mainly acts on alpha-1 adrenergic receptors, but has little affinity for beta receptors. The drug is largely obsolete, being superseded by other hemostatics such as thrombin, fibrinogen, and vasopressin analogues.[1]
Contraindications and interactions
Adrenalone does not stop bleeding from large blood vessels. It is not approved for systemic use. Combination with antithrombotics is not useful because they contravene the action of adrenalone.[1]
Side effects
Vasoconstriction by adrenalone may lead to local necrosis.[1]
Pregnancy and lactation
Adrenalone passes into breast milk, but adverse effects are unlikely because of its very low systemic resorption.[1]
Chemical properties
Adrenalone is a derivative of epinephrine, having the alcohol function replaced with a ketone. As a consequence, it is not optically active any more.
Solubility in water, ethanol and diethyl ether is low. The substance is typically used in form of the hydrochloride, a white crystalline powder which tastes bitter and slightly acidic, and is soluble in water (1:8) and 94% ethanol (1:45). The melting point of the hydrochloride is 243 °C (469.4 °F).[1]
Pharmacology
After local application, only traces of adrenalone are found in the blood, which is partly a consequence of the vasoconstriction caused by the drug via alpha-1 adrenergic receptors. In an (unspecified) pharmacological model, hypertensive (blood pressure increasing) action has been found to be about 0.5% that of epinephrine at equivalent plasma concentrations. Therefore, systemic effects are unlikely.
Like epinephrine, adrenalone is metabolised by catechol-O-methyl transferase (COMT), yielding 3O-methyladrenalone, which in turn is N-demethylized by monoamine oxidase (MAO). Alternatively, it can first undergo metabolization by MAO and then by COMT; in both cases, the resulting 3O-methyl-N-demethyladrenalone is conjugated to sulfate or glucuronide and excreted by the kidney. No reduction to epinephrine has been observed in vivo.[1]
References
- Pages with script errors
- Pages with non-numeric formatnum arguments
- CS1 maint: Unrecognized language
- Articles with changed CASNo identifier
- Articles with changed EBI identifier
- E number from Wikidata
- ECHA InfoCard ID from Wikidata
- Chemical articles with unknown parameter in Infobox drug
- Chemical pages without DrugBank identifier
- Drugs with no legal status
- Drugboxes which contain changes to verified fields
- Drug
- Catecholamines
- Aromatic ketones