Hereditary nonpolyposis colorectal cancer pathophysiology: Difference between revisions
No edit summary |
|||
| Line 4: | Line 4: | ||
==Overview== | ==Overview== | ||
Hereditary nonpolyposis colorectal cancer is an autosomal dominant genetic disease characterized by an early onset of [[colon cancer]], [[endometrial cancer]], and other malignant tumors.<ref name="pmid26474631">{{cite journal |vauthors=Si JW, Wang L, Ba XJ, Zhang X, Dong Y, Zhang JX, Li WT, Li T |title=[Clinicopathological screening of Lynch syndrome: a report of 2 cases and literature review] |language=Chinese |journal=Beijing Da Xue Xue Bao |volume=47 |issue=5 |pages=858–64 |year=2015 |pmid=26474631 |doi= |url=}}</ref> Development of hereditary nonpolyposis colorectal cancer is the result of multiple [[genetic mutation]]s. The most common genetic mutation involved in the pathogenesis of hereditary nonpolyposis colorectal cancer is a mutation in the APC gene. Other genes involved in the pathogenesis of hereditary nonpolyposis colorectal cancer include: MSH-2, MLH-1, MSH-6, PMS-2 ,PMS-1, TGF-BR2, and MLH-3. This syndrome occurs most commonly in the proximal colon (60% to 80%). [[Endometrial cancer]] is the most common sentinel cancer among female patients with hereditary nonpolyposis colorectal cancer.<ref name="pmid26474631">{{cite journal |vauthors=Si JW, Wang L, Ba XJ, Zhang X, Dong Y, Zhang JX, Li WT, Li T |title=[Clinicopathological screening of Lynch syndrome: a report of 2 cases and literature review] |language=Chinese |journal=Beijing Da Xue Xue Bao |volume=47 |issue=5 |pages=858–64 |year=2015 |pmid=26474631 |doi= |url=}}</ref> | Hereditary nonpolyposis colorectal cancer is an autosomal dominant genetic disease characterized by an early onset of [[colon cancer]], [[endometrial cancer]], and other malignant tumors.<ref name="pmid26474631">{{cite journal |vauthors=Si JW, Wang L, Ba XJ, Zhang X, Dong Y, Zhang JX, Li WT, Li T |title=[Clinicopathological screening of Lynch syndrome: a report of 2 cases and literature review] |language=Chinese |journal=Beijing Da Xue Xue Bao |volume=47 |issue=5 |pages=858–64 |year=2015 |pmid=26474631 |doi= |url=}}</ref> Development of hereditary nonpolyposis colorectal cancer is the result of multiple [[genetic mutation]]s. The most common genetic mutation involved in the pathogenesis of hereditary nonpolyposis colorectal cancer is a mutation in the APC gene. Other genes involved in the pathogenesis of hereditary nonpolyposis colorectal cancer include: MSH-2, MLH-1, MSH-6, PMS-2 , PMS-1, TGF-BR2, and MLH-3. This syndrome occurs most commonly in the proximal colon (60% to 80%). [[Endometrial cancer]] is the most common sentinel cancer among female patients with hereditary nonpolyposis colorectal cancer.<ref name="pmid26474631">{{cite journal |vauthors=Si JW, Wang L, Ba XJ, Zhang X, Dong Y, Zhang JX, Li WT, Li T |title=[Clinicopathological screening of Lynch syndrome: a report of 2 cases and literature review] |language=Chinese |journal=Beijing Da Xue Xue Bao |volume=47 |issue=5 |pages=858–64 |year=2015 |pmid=26474631 |doi= |url=}}</ref> | ||
==Pathogenesis== | ==Pathogenesis== | ||
Revision as of 15:37, 9 February 2016
|
Hereditary Nonpolyposis Colorectal Cancer Microchapters |
|
Differentiating Hereditary Nonpolyposis Colorectal Cancer from other Diseases |
|---|
|
Diagnosis |
|
Treatment |
|
Case Studies |
|
Hereditary nonpolyposis colorectal cancer pathophysiology On the Web |
|
American Roentgen Ray Society Images of Hereditary nonpolyposis colorectal cancer pathophysiology |
|
FDA on Hereditary nonpolyposis colorectal cancer pathophysiology |
|
CDC on Hereditary nonpolyposis colorectal cancer pathophysiology |
|
Hereditary nonpolyposis colorectal cancer pathophysiology in the news |
|
Blogs on Hereditary nonpolyposis colorectal cancer pathophysiology |
|
Directions to Hospitals Treating Hereditary nonpolyposis colorectal cancer |
|
Risk calculators and risk factors for Hereditary nonpolyposis colorectal cancer pathophysiology |
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]Associate Editor(s)-in-Chief: Maria Fernanda Villarreal, M.D. [2]
Overview
Hereditary nonpolyposis colorectal cancer is an autosomal dominant genetic disease characterized by an early onset of colon cancer, endometrial cancer, and other malignant tumors.[1] Development of hereditary nonpolyposis colorectal cancer is the result of multiple genetic mutations. The most common genetic mutation involved in the pathogenesis of hereditary nonpolyposis colorectal cancer is a mutation in the APC gene. Other genes involved in the pathogenesis of hereditary nonpolyposis colorectal cancer include: MSH-2, MLH-1, MSH-6, PMS-2 , PMS-1, TGF-BR2, and MLH-3. This syndrome occurs most commonly in the proximal colon (60% to 80%). Endometrial cancer is the most common sentinel cancer among female patients with hereditary nonpolyposis colorectal cancer.[1]
Pathogenesis
- Hereditary nonpolyposis colorectal cancer results from a defect in DNA mismatch repair mechanism that leads to microsatellite instability (MSI-H), which is a hallmark of hereditary nonpolyposis colorectal cancer.
- Most cases result in changes in the lengths of dinucleotide repeats of the nucleobases cytosine and adenine.[2]
- In hereditary nonpolyposis colorectal cancer, colon malignancies usually occur from a single to a few adenomas, and mostly occurs in the proximal colon.
Genetics
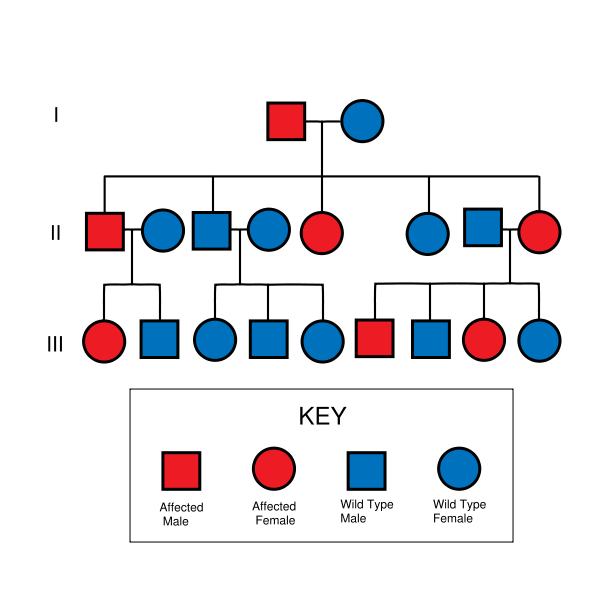
- Hereditary nonpolyposis colorectal cancer is inherited in an autosomal dominant manner.
- The majority of patients with hereditary nonpolyposis colorectal cancer inherit the condition from a parent.
- Families found to have a deleterious mutation in an hereditary nonpolyposis colorectal cancer gene should be considered to have hereditary nonpolyposis colorectal cancer regardless of the extent of the family history.
- However, due to incomplete penetrance, variable age of cancer diagnosis, cancer risk reduction, or early death, not all patients with an hereditary nonpolyposis colorectal cancer gene mutation have a positive family history of the disease.
- Some patients develop hereditary nonpolyposis colorectal cancer de-novo in a new generation, without inheriting the gene. These patients are often only identified after they develop colon cancer at an early age.
- Patients with hereditary nonpolyposis colorectal cancer have a 50% chance to pass the gene on to each child.
- Development of hereditary nonpolyposis colorectal cancer is the result of multiple genetic mutations.[1]
- Most mutations (90%) that cause hereditary nonpolyposis colorectal cancer are found in the MLH1 or MSH2 genes. The other genes, account for the other 10% of mutations.[3]
- MSH2 mutations have an increased risk of urothelial carcinoma relative to MLH1 and MSH6 mutations.[4]
- The table below lists the genes involved in the pathogenesis of hereditary nonpolyposis colorectal cancer include:
| Genes implicated in hereditary nonpolyposis colorectal cancer | Frequency of mutations in hereditary nonpolyposis colorectal cancer families | Locus |
|---|---|---|
| MSH2 | approximately 60% | 2p22 |
| MLH1 | approximately 30% | 3p21 |
| MSH6 | 7-10% | 2p16 |
| PMS2 | relatively infrequent | 7p22 |
| PMS1 | case report | 2q31-q33 |
| TGFBR2 | case report | 3p22 |
| MLH3 | disputed | 14q24.3 |
Associated Conditions
- Colorectal carcinoma
- Endometrial carcinoma
- Non-endometrioid endometrial carcinoma[5][6]
- Endometrioid endometrial carcinoma[7]
- Stomach carcinoma (intestinal type)[8][9]
- Biliary tree carcinoma[8]
- Pancreatic carcinoma
- Urinary system carcinoma
Gross Pathology
- On gross pathology, there are no characteristic findings of hereditary nonpolyposis colorectal cancer.[1]
Microscopic Pathology
- On microscopic histopathological analysis, hereditary nonpolyposis colorectal cancer is more likely to be poorly differentiated, abundant in extracellular mucin, and distinguished by a lymphoid (peritumoral lymphocytes) host response to the tumor.[1]
- Three major groups of MSI-H cancers can be recognized by histopathological criteria, which include:
- Right-sided poorly differentiated cancers
- Right-sided mucinous cancers
- Adenocarcinomas in any location showing any measurable level of intraepithelial lymphocyte
Gallery
-
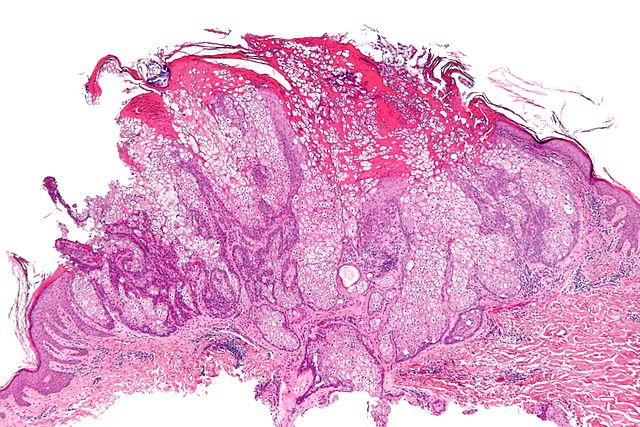
Micrograph of a sebaceous adenoma, as may be seen in Muir-Torre syndrome.
-
Hereditary nonpolyposis colorectal cancer is inherited in an autosomal dominant fashion.
References
- ↑ 1.0 1.1 1.2 1.3 1.4 Si JW, Wang L, Ba XJ, Zhang X, Dong Y, Zhang JX, Li WT, Li T (2015). "[Clinicopathological screening of Lynch syndrome: a report of 2 cases and literature review]". Beijing Da Xue Xue Bao (in Chinese). 47 (5): 858–64. PMID 26474631.
- ↑ Oki E, Oda S, Maehara Y, Sugimachi K (Mar 1999). "Mutated gene-specific phenotypes of dinucleotide repeat instability in human colorectal carcinoma cell lines deficient in DNA mismatch repair". Oncogene. 18 (12): 2143–7. doi:10.1038/sj.onc.1202583. PMID 10321739.
- ↑ Lynch Syndrome. Canadian Cancer Society http://www.cancer.ca/en/cancer-information/cancer-101/what-is-a-risk-factor/genetic-risk/lynch-syndrome/?region=ab#ixzz3t69IQ9M7 Accessed on December 01 2015
- ↑ van der Post, RS.; Kiemeney, LA.; Ligtenberg, MJ.; Witjes, JA.; Hulsbergen-van de Kaa, CA.; Bodmer, D.; Schaap, L.; Kets, CM.; van Krieken, JH. (2010). "Risk of urothelial bladder cancer in Lynch syndrome is increased, in particular among MSH2 mutation carriers". J Med Genet. 47 (7): 464–70. doi:10.1136/jmg.2010.076992. PMID 20591884. Unknown parameter
|month=ignored (help) - ↑ Okuda T, Sekizawa A, Purwosunu Y; et al. (2010). "Genetics of endometrial cancers". Obstet Gynecol Int. 2010: 984013. doi:10.1155/2010/984013. PMC 2852605. PMID 20396392.
- ↑ Garg, K.; Soslow, RA. (2009). "Lynch syndrome (hereditary non-polyposis colorectal cancer) and endometrial carcinoma". J Clin Pathol. 62 (8): 679–84. doi:10.1136/jcp.2009.064949. PMID 19638537. Unknown parameter
|month=ignored (help) - ↑ Lax, SF. (2002). "[Dualistic model of molecular pathogenesis in endometrial carcinoma]". Zentralbl Gynakol. 124 (1): 10–6. doi:10.1055/s-2002-20303. PMID 11873308. Unknown parameter
|month=ignored (help) - ↑ 8.0 8.1 Online Mendelian Inheritance in Man (OMIM) 120435
- ↑ Cristofaro, G.; Lynch, HT.; Caruso, ML.; Attolini, A.; DiMatteo, G.; Giorgio, P.; Senatore, S.; Argentieri, A.; Sbano, E. (1987). "New phenotypic aspects in a family with Lynch syndrome II". Cancer. 60 (1): 51–8. PMID 3581033. Unknown parameter
|month=ignored (help)