Corticosteroid
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]
Corticosteroids are a class of steroid hormones that are produced in the adrenal cortex. Corticosteroids are involved in a wide range of physiologic systems such as stress response, immune response and regulation of inflammation, carbohydrate metabolism, protein catabolism, blood electrolyte levels, and behavior.
- Glucocorticoids such as cortisol control carbohydrate, fat and protein metabolism and are anti-inflammatory by preventing phospholipid release, decreasing eosinophil action and a number of other mechanisms.
- Mineralocorticoids such as aldosterone control electrolyte and water levels, mainly by promoting sodium retention in the kidney.
Some common natural hormones are corticosterone (C21H30O4), cortisone (C21H28O5, 17-hydroxy-11-dehydrocorticosterone) and aldosterone.
Synthesis
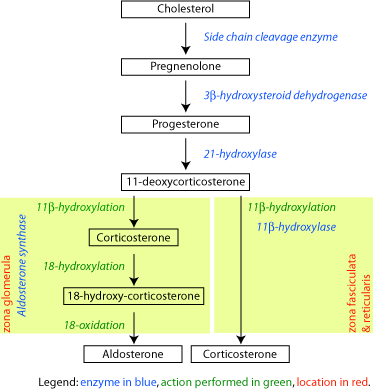
The corticosteroids are synthesized from cholesterol within the adrenal cortex. Most steroidogenic reactions are catalysed by enzymes of the cytochrome P450 family. They are located within the mitochondria and require adrenodoxin as a cofactor (except 21-hydroxylase and 17α-hydroxylase).
Aldosterone and corticosterone share the first part of their biosynthetic pathway. The last part is either mediated by the aldosterone synthase (for aldosterone) or by the 11β-hydroxylase (for corticosterone). These enzymes are nearly identical (they share 11β-hydroxylation and 18-hydroxylation functions). But aldosterone synthase is also able to perform a 18-oxidation. Moreover, aldosterone synthase is found within the zona glomerulosa at the outer edge of the adrenal cortex; 11β-hydroxylase is found in the zona fasciculata and reticularis.
Uses
Synthetic drugs with corticosteroid-like effect are used in a variety of conditions, ranging from brain tumors to skin diseases. Dexamethasone and its derivatives are almost pure glucocorticoids, while prednisone and its derivatives have some mineralocorticoid action in addition to the glucocorticoid effect. Fludrocortisone (Florinef®) is a synthetic mineralocorticoid. Hydrocortisone (cortisol) is available for replacement therapy, e.g. in adrenal insufficiency and congenital adrenal hyperplasia.
Synthetic glucocorticoids are used in the treatment of joint pain or inflammation (arthritis), temporal arteritis, dermatitis, allergic reactions, asthma, hepatitis, systemic lupus erythematosus, inflammatory bowel disease (ulcerative colitis and Crohn's disease), sarcoidosis and for glucocorticoid replacement in Addison's disease or other forms of adrenal insufficiency. Topical formulations for treatment of skin, eye diseases (uveitis) or inflammatory bowel disease are available. Corticosteroids are also used supportively to prevent nausea, often in combination with 5-HT3 antagonists (e.g. ondansetron).
Typical undesired effects of glucocorticoids present quite uniformly as drug-induced Cushing's syndrome. Typical mineralocorticoid side effects are hypertension (abnormally high blood pressure), hypokalemia (low potassium levels in the blood), hypernatremia (high sodium levels in the blood) without causing peripheral edema, metabolic alkalosis and connective tissue weakness (Werner, 2005).
Clinical and experimental evidence indicates that corticosteroids can cause permanent eye damage by inducing central serous retinopathy (CSR, also known as central serous chorioretinopathy, CSC). A variety of steroid medications, from anti-allergy nasal sprays (Nasonex, Flonase) to topical skin creams, to eye drops (Tobradex), to Prednisone have been implicated in the development of CSR.
History
Tadeusz Reichstein together with Edward Calvin Kendall and Philip Showalter Hench were awarded the Nobel Prize for Physiology and Medicine in 1950 for their work on hormones of the adrenal cortex which culminated in the isolation of cortisone.
Corticosteroids have been used as a drug treatment for some time. Lewis Sarett of Merck & Co. was the first to synthesize cortisone, using a complicated 36-step process that started with deoxycholic acid, which was extracted from ox bile. The low efficiency of converting deoxycholic acid into cortisone led to a cost of US $200 per gram. Russell Marker, at Syntex, discovered a much cheaper and more convenient starting material, diosgenin from wild Mexican yams. His conversion of diosgenin into progesterone by a four-step process now known as Marker degradation was an important step in mass production of all steroidal hormones, including cortisone and chemicals used in hormonal contraception. In 1952, D.H. Peterson and H.C. Murray of Upjohn Co. developed a process that used Rhizopus mold to oxidize progesterone into a compound that was readily converted to cortisone. The ability to cheaply synthesize large quantities of cortisone from the diosgenin in yams resulted in a rapid drop in price to US $6 per gram, falling to $0.46 per gram by 1980. Percy Julian's research also aided progress in the field. The exact nature of cortisone's anti-inflammatory nature remained a mystery for years after however, until the leukocyte adhesion cascade and the role of phospholipase A2 in the production of prostaglandins and leukotrienes was fully understood in the early 1980s. These steroids help people with muscle problems.
References
Werner R (2005). A massage therapist's guide to Pathology. 3rd edition. Lippincott Williams & Wilkins, Pennsylvania, USA.
See also
Template:Corticosteroids Template:Major Drug Groups
cs:Kortikosteroidy
de:Kortikoid
it:Corticosteroide
lt:Kortikosteroidai
nl:Corticosteroïde
no:Kortikosteroid
sl:Kortikosteroid