Abdominal aortic aneurysm overview
|
Abdominal Aortic Aneurysm Microchapters |
|
Differentiating Abdominal Aortic Aneurysm from other Diseases |
|---|
|
Diagnosis |
|
Treatment |
|
Case Studies |
|
Abdominal aortic aneurysm overview On the Web |
|
Directions to Hospitals Treating Abdominal aortic aneurysm overview |
|
Risk calculators and risk factors for Abdominal aortic aneurysm overview |
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]; Associate Editor-In-Chief: Cafer Zorkun, M.D., Ph.D. [2]
Synonyms and keywords: Abdominal aneurysm, aortic; aortic aneurysm, abdominal; AAA; triple A
Overview
An abdominal aortic aneurysm is a localized dilatation of the abdominal aorta, that exceeds the normal diameter of the abdominal aorta by more than 50%. The normal diameter of an aorta depends on the patient's age, sex, height, weight, race, body surface area, and baseline blood pressure. On average, the normal diameter of the infrarenal aorta is 2 cm, and therefore a true AAA measures 3.0 cm or more. Aortic ectasia is a mild generalized dilatation (<50% of the normal diameter of ≤ 2.9 cm) that is due to age-related degenerative changes in the vessel walls.
Historical Perspective
The etymology of the word aneurysm comes from the Greek word for "dilatation". Abdominal aortic aneurysm as a medical condition has been recognized since ancient times, but was not been successfully treated until the early part of the 20th century. In 1923, Rudolph Matas (who also proposed the concept of endoaneurysmorrhaphy), performed the first successful aortic ligation on a human. In 1949, Albert Einstein was operated on by Rudolf Nissen by wrapping the aorta with polyethene cellophane, which induced fibrosis and restricted the growth of the aneurysm. In 1951, Charles Dubost performed the first AAA repair using a homograft. In 1953, Blakemore and Voorhees repaired a ruptured AAA using a Vinyon-N graft (nylon). In 1962, Javid and Creech reported the technique of endoaneurysmorrhaphy. In 1980, Parodi et al described an endovascular repair.
Definition
The normal diameter of one's aorta depends on the patient's age, sex, height, weight, race, body surface area, and baseline blood pressure. On average, the normal diameter of the infrarenal aorta (where abdominal aortic aneurysms are located) is 2 cm. Therefore an abdominal aortic aneurysm is defined as a dilation of 3.0 cm or more.
An abdominal aortic aneurysm is to be distinguished from aortic ectasia which is defined as a mild generalized dilatation (<50% of the normal diameter of ≤ 2.9 cm) that is due to age-related degenerative changes in vessel wall.
Anatomy
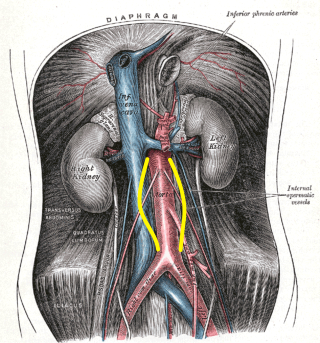
The aorta below the renal arteries, also known as the infrarenal aorta, is the location of 90% of abdominal aortic aneurysms. Other possible locations are suprarenal and pararenal. The aneurysm can extend to include one or both of the iliac arteries.
Classification
Aneurysms are usually classified by their shape:
Fusiform Aneurysms
Most common type seen in the infrarenal aorta and are diffuse and circumferential.
Saccular Aneurysms
Involve only a portion of the circumference, with a characteristic outpouching of the vessel wall.
Pathophysiology
The underlying pathophysiology of abdominal aortic aneurysm involves genetic influences, smoking, hypertension, hemodynamic influences and underlying atherosclerosis. In rare instances infection, arteritis, and connective tissue disorders may play a role.
Causes
The most common cause of an AAA is atherosclerosis.
Differentiating Abdominal Aortic Aneurysm from other Diseases
An abdominal aortic aneurysm should be differentiated from other causes of abdominal pain such as acute cholecystitis, gastrointestinal bleeding, perforated peptic ulcer, ischemic bowel, nephrolithiasis, pyelonephritis, appendicitis, cholelithiasis, large bowel obstruction, small bowel obstruction, pancreatitis, musculoskeletal pain, myocardial infarction, and urinary tract infection.
Epidemiology and Demographics
Abdominal aortic aneurysm is the 13th leading cause of death in the US. Abdominal aortic aneurysms are more common in developed countries. Elderly, caucasian males who are smokers are at higher risk for developing an abdominal aortic aneurysm.
Risk Factors
Initial Development
Smoking
The most significant modifiable risk factor for the development of an abdominal aortic aneurysm is smoking which increases the risk of aneurysm development 8 fold.
Age
Advanced age and family history are the strongest non-modifiable risk factors for the development of an abdominal aortic aneurysm.
Hypercholesterolemia
Hypercholesterolemia and hypertension are risk factors as well.
Caucasian Race
Caucasian race is associated with a higher incidence of AAA when compared to non Caucasian race.
Expansion
An increased rate of expansion of abdominal aortic aneurysm is related to systolic hypertension, wide pulse pressure, and ongoing smoking.
Rupture
The risk of abdominal aortic aneurysm rupture is proportional to the size and rate of growth of the aneurysm. Abdominal aortic aneurysms greater than 5 cm diameter or those that grow faster than 1 cm per year have a significantly increased risk of rupture and are indications for elective operative repair. Advanced age, female gender, hypertension, active smoking, outpouchings, and mural thrombus are also risk factors for abdominal aortic aneurysm rupture.
Screening
Approximately 16% of large abdominal aortic aneurysms (diameter > 5.5 cm) rupture, causing 9,000 AAA-related deaths in the United States per year[1]. Several studies have shown that screening can drastically reduce the aneurysm rupture rate by 45-49% for men older than 60, and reduce AAA-related mortality by 21-68%. In a landmark study randomizing 67,800 men, (The Multicenter Aneurysm Screening Study) aneurysm-related mortality was 53% lower in the screening group as compared with control patients[2][3]. The United States Preventive Services Task Force (USPSTF) recommends one-time screening for abdominal aortic aneurysm (AAA) by ultrasonography in any man aged 65 to 75 who has ever smoked. The USPSTF makes no recommendation for or against screening for AAA in men aged 65 to 75 who have never smoked. The USPSTF recommends against routine screening for AAA in women.
Natural History, Complications and Prognosis
Abdominal aortic aneurysms expand slowly over time. An aortic aneurysm can progress to a ruptured abdominal aortic aneurysm, which is a medical emergency associated with an extremely high mortality. Serious complications can also occur as a result of an aortic dissection, such as myocardial infarction, shock, stroke, kidney failure, and arterial emboli. A ruptured AAA carries an overall mortality rate approaching 75%, which is much higher than the mortality rate of 2-6% in those patients who underwent elective surgical repair. Approximately 16% of large AAAs (diameter >5.5 cm) rupture, causing 9,000 AAA-related deaths in the United States per year[1]. Several studies have shown that screening can drastically reduce the aneurysm rupture rate by 45-49% for men older than 60, and reduce AAA-related mortality by 21-68%. The outcome is usually good if an experienced surgeon repairs the aneurysm before it ruptures. Rupture of an abdominal aortic aneurysm is associated with a 60% to 90% mortality before the patient reaches a hospital. It is associated with a 30% to 80% operative mortality among those patients who survive long enough to undergo surgery.
History and Symptoms
Given that smoking increases the risk of abdominal aortic aneurysm development, a smoking history should be obtained in elderly men. If a male over the age of 65 has a history of smoking, they should undergo ultrasonography to screen for an abdominal aortic aneurysm. Abdominal aortic aneurysms are usually asymptomatic until they expand or rupture. Spontaneous abdominal pain in a patient with a pulsatile epigastric mass or a known AAA may signal rupture into the retroperitoneum or leakage within the aneurysm wall. If a patient does develop symptoms, the risk of rupture is quite high, which is why symptoms are considered an indication for surgery. Pain is the most common symptomatic manifestation of AAA.
Physical Examination
Physical examination has a low sensitivity in the detection of small abdominal aortic aneurysms (29-61% for abdominal aortic aneurysms 3.0-3.9 cm in diameter) but has a sensitivity of 76-82% to detect those abdominal aortic aneurysms that are 5.0 cm or larger that may warrant repair. It is easier to detect a pulsatile mass in thin patients and those who do not have tense abdomens. Contrary to popular belief, gentle palpation of abdominal aortic aneurysms is safe, and does not precipitate rupture.
Abdominal X Ray
Plain abdominal radiographs are obtained in patients with abdominal complaints before the diagnosis of AAA has been made. It is very difficult to evaluate patients with AAA using plain radiographs, because aortic wall calcification is seen in less than half of the cases with AAA. However, plain radiographs may be helpful when an aneurysmal aorta appears normal on angiogram, due to thrombus within the sac.
Abdominal Ultrasound
Abdominal ultrasound is the standard imaging modality to detect and follow the progression of an abdominal aortic aneurysm. When performed by an expert, it has a sensitivity and specificity close to 100% and 96%, respectively. Abdominal ultrasound can also detect mural thrombus, iliac artery aneurysms, and free peritoneal blood. Patients should fast before examination to optimize image quality.
Computed Tomography (CT)
Although CT scanning provides detailed anatomic information and is valuable in planning abdominal aortic aneurysm repair, it is not used as a screening tool given the potential nephrotoxicity associated with the dye load, the cost, and the exposure to radiation.
Magnetic Resonance Imaging (MRI)
MRI might be a better soft tissue visualizer than ultrasonography and CT, but its limitations limit its usage as a screening tool and as a diagnostic tool during emergencies like ruptured AAA. However its advantages makes it a good alternative in AAA patients with poor renal function and in elective preoperative evaluation of unruptured AAA[4].
Other Imaging Findings
Angiography is an alternative less often used imaging method for visualization of an abdominal aortic aneurysm. Angiography is helpful in determining anatomy of aorta precisely. It can be used preoperatively in case of suspected suprarenal or thoracic aortic aneurysm, femoral or popliteal aneurysm, renal artery stenosis, unexplained renal insufficiency, occlusive ilio-femoral disease, or visceral ischemia.
Medical Therapy
Risk factor modification, such as smoking cessation, management of hypertension, and lipid lowering are essential in reducing the risk of development and the rate of progression of abdominal aortic aneurysms.
Surgery
Indications for operative repair of an abdominal aortic aneurysm include the presence of symptoms such as back pain, the presence of a rupture or contained rupture, an abdominal aortic aneurysm diameter greater than 5.5 cm, a rate of expansion of greater than 1 cm in one year, and an inflammatory or infectious etiology. Open surgical repair is favored in young patients as an elective procedure, the presence of an expanding aneurysm, symptomatic aneurysm, or ruptured aneurysm. An endovascular repair (EVAR) is favored in older patients and high risk patients or those unfit for open repair. Risk factors for postoperative morbidity and mortality include extensive atheromatous disease, mural calcification, thrombosis, juxtarenal extension of the aneurysm and the presence of inflammatory changes.
Prevention
Smoking cessation and blood pressure management are two strategies to reverse modifiable risk factors for the development, progression, and rupture of an abdominal aortic aneurysm.
References
- ↑ 1.0 1.1 Gillum RF (1995). "Epidemiology of aortic aneurysm in the United States". Journal of Clinical Epidemiology. 48 (11): 1289–98. PMID 7490591. Retrieved 2012-10-27. Unknown parameter
|month=ignored (help) - ↑ Ashton HA, Buxton MJ, Day NE, Kim LG, Marteau TM, Scott RA, Thompson SG, Walker NM (2002). "The Multicentre Aneurysm Screening Study (MASS) into the effect of abdominal aortic aneurysm screening on mortality in men: a randomised controlled trial". Lancet. 360 (9345): 1531–9. PMID 12443589. Retrieved 2012-10-27. Unknown parameter
|month=ignored (help) - ↑ "Multicentre aneurysm screening study (MASS): cost effectiveness analysis of screening for abdominal aortic aneurysms based on four year results from randomised controlled trial". BMJ (Clinical Research Ed.). 325 (7373): 1135. 2002. PMC 133450. PMID 12433761. Retrieved 2012-10-27. Unknown parameter
|month=ignored (help) - ↑ Thurnher, SA.; Dorffner, R.; Thurnher, MM.; Winkelbauer, FW.; Kretschmer, G.; Polterauer, P.; Lammer, J. (1997). "Evaluation of abdominal aortic aneurysm for stent-graft placement: comparison of gadolinium-enhanced MR angiography versus helical CT angiography and digital subtraction angiography". Radiology. 205 (2): 341–52. PMID 9356613. Unknown parameter
|month=ignored (help)