Membranous glomerulonephritis pathophysiology
|
Membranous glomerulonephritis Microchapters |
|
Differentiating Membranous glomerulonephritis from other Diseases |
|---|
|
Diagnosis |
|
Treatment |
|
Case Studies |
|
Membranous glomerulonephritis pathophysiology On the Web |
|
American Roentgen Ray Society Images of Membranous glomerulonephritis pathophysiology |
|
Directions to Hospitals Treating Membranous glomerulonephritis |
|
Risk calculators and risk factors for Membranous glomerulonephritis pathophysiology |
- Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]; Associate Editor(s)-in-Chief: Syed Ahsan Hussain, M.D.[2]
Overview
It is thought that MGN is mediated by genetic factors like PLA2R, enviromental factors, drug (captopril) side effects, infections like hepatitis B and hepatitis C.
Pathophysiology
- The membranous glomerulonephritis is a result of multiple changes.[1][2][3][4][5][6][7][8]
- MGN is caused by immune complex formation in the glomerulus. The immune complexes are formed by binding of antibodies to antigens in the glomerular basement membrane. The antigens may be part of the basement membrane, or deposited from elsewhere by the systemic circulation
- The immune complex serves as an activator that triggers a response from the complement system and form a membrane attack complex which stimulates release of proteases and oxidants by the mesangial and epithelial cells, damaging the capillary walls and causing them to become "leaky".
Phospholipase A2 receptor
- The M-type PLA2R is the major antigen in human idiopathic MN. It is expressed in glomerular podocytes.[3][4][5][6][7][8]
- There was no colocalization of PLA2R in secondary MN biopsies.
- PLA2R antigen detected within immune deposits by immunofluorescence of the biopsy specimen.
- Detection of the immune complex specificity is 100 percent.
Thrombospondin type-1
- THSD7A has been found in patients with idiopathic MN who are negative for anti-PLA2R antibodies.[3][4][5][6][7][8]
Neutral endopeptidase
- Anti-neutral endopeptidase antibodies caused MN in the neonates.
- It resolves months after birth.
| HLA susceptibility 1 | Environmental factors | ||||||||||||||||||||||||||||
| Variant of PLA2R1 on podocyte surface | |||||||||||||||||||||||||||||
| Innate immunity activation and inflammation dendritic cell sense epitope of PLA2R1 and present them for adaptive immunity | |||||||||||||||||||||||||||||
| Production of auto-immune antibody IgG4/IgG1 which attach them self to epitope on podocyte surface | |||||||||||||||||||||||||||||
| In Situ formation and shedding of subepithelial immune complex | |||||||||||||||||||||||||||||
| Complement activationMANNAN PATHWAY and T cell activation | |||||||||||||||||||||||||||||
| Which lead to cytokine release oxygen derivative release membrane attack complex C5-C9 | |||||||||||||||||||||||||||||
| Podocyte injury by apoptosis altered lectin cytoskeleton loss of silt pore integrity loss of glomerular permeability proteinuria | |||||||||||||||||||||||||||||
Genetics
- Single-nucleotide polymorphisms (SNPs) at two loci that are highly associated with idiopathic MN.
- The two loci are within the genes for the PLA2R on chromosome 2q24.
- The human leukocyte antigen (HLA) complex class II alpha chain 1A (HLA-DQA1) on chromosome 6p21.
- The PLA2R has been identified as a major antigen in idiopathic MN.[3][4][5][6][7][8]
Associated Conditions
Consitions associated with MN include:
- Hepatitis B
- Hepatitis C
- Congenital Syphilis
- Systemic Lupus Erythematosis
- Malignancy
- Lung
- Breast
- Colon
- Stomach
- Kidney
- Leukemia
- Lymphomas (Hodgkin’s and non-Hodgkin’s)
Gross Pathology
- There is no significant gross pathological changes.
Microscopic Pathology
Microscopic pathologic findings characteristic of MN include:[1][2]
- Early biopsies may be normal
- Later: uniform diffuse capillary wall thickening without hypercellularity, No mesangial sclerosis and inflammatory cells
- Proximal convoluted tubules: hyaline droplets, reflecting protein reabsorption
- Membrane thickening and narrow capillary lumina.
- Mesangial sclerosis and glomerulosclerosis
Immunofluorescence
- Granular diffuse peripheral deposits, usually IgG and C3, also C5b-C9 and occasionally IgM or IgA
- C4d immunostaining may be diagnostic.
Stages
- Stage I: LM - normal for slightly thickened BM,
- slight GMB vacuolization; IF - fine granular IgG, C3; EM - scattered small subepithelial electron dense deposits, no foot process effacement or spikes
- Stage II: LM - moderately thickened BM with spikes and vacuolization; IF - moderate sized, granular IgG, C3; EM - diffuse spikes due to subepithelial deposits, diffuse foot process effacement
- Stage III: LM - markedly thickened GBM, residual spikes and vacuoles, chain like appearance; IF - coarsely granular IgG, C3; EM - intramembranous deposits, spikes, neomembrane formation and diffuse foot process effacement
- Stage IV: LM - markedly thickened GBM, few spikes, vacuoles and glomerulosclerosis; IF - focal IgG, C3; EM - sclerotic GBM, few deposits and lacunae
GBM means glomerular basement membrane, IF means immunofluorenscence, EM means electrone microscopy [1][2]
| stage | Glomerular Basement Membrane | Immunofluorescence | Electron Microscopy |
|---|---|---|---|
| Stage 1 | Normal or slightly thickned BM | Fine granular IgG, C3 | Scattered small subepithelial electron dense deposits no foot effacement |
| Stage 2 | Moderately thickened BM with spikes and vacuolization | Granular IgG, C3 | Diffuse spikes due to subepithelial deposits, diffuse foot process effacement |
| Stage 3 | Moderately thickened BM residual spikes and vacuoles | Chain like appearance IF, coarsely granular IgG, C3 | Intramembraneous deposits, spikes, neomembrane formation and diffuse foot process effacement |
| Stage 4 | Markedly thick GBM, few spikes, vacoules and glomerulosclerosis | Focal IgG, C3 | Sclerotic GBM, few deposits and lacunae |
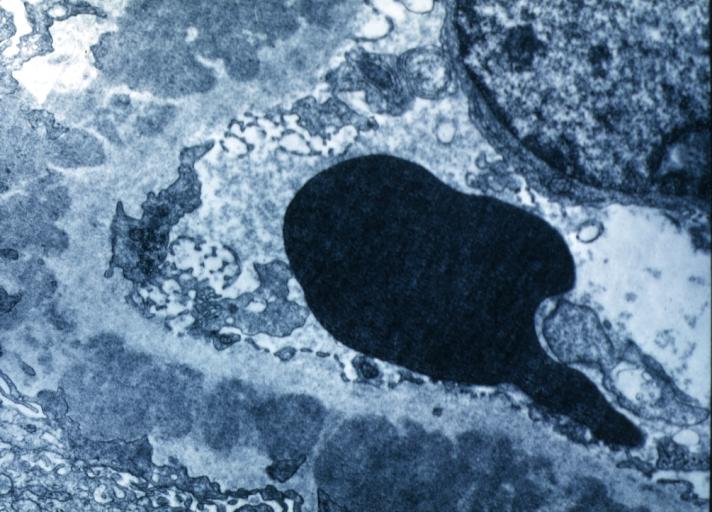
Electron microscopy
- There are electron-dense deposits in the epithelial aspect (external) of the GBM, between this one and the epithelial cell: subepithelials or epimembranous.
- Subepithelial deposits that nestle against the glomerular basement membrane seems to be the cause of the thickening.
- Spikes are demonstrated as irregular projections of the GBM among the subepithelial deposits.
- The podocytes lose their foot processes.
References
- ↑ 1.0 1.1 1.2 Cybulsky AV, Quigg RJ, Salant DJ (October 2005). "Experimental membranous nephropathy redux". Am. J. Physiol. Renal Physiol. 289 (4): F660–71. doi:10.1152/ajprenal.00437.2004. PMC 1325222. PMID 16159900.
- ↑ 2.0 2.1 2.2 Nangaku M, Shankland SJ, Couser WG (May 2005). "Cellular response to injury in membranous nephropathy". J. Am. Soc. Nephrol. 16 (5): 1195–204. doi:10.1681/ASN.2004121098. PMID 15800119.
- ↑ 3.0 3.1 3.2 3.3 3.4 Cunningham PN, Quigg RJ (May 2005). "Contrasting roles of complement activation and its regulation in membranous nephropathy". J. Am. Soc. Nephrol. 16 (5): 1214–22. doi:10.1681/ASN.2005010096. PMID 15800113.
- ↑ 4.0 4.1 4.2 4.3 4.4 Beck LH, Bonegio RG, Lambeau G, Beck DM, Powell DW, Cummins TD, Klein JB, Salant DJ (July 2009). "M-type phospholipase A2 receptor as target antigen in idiopathic membranous nephropathy". N. Engl. J. Med. 361 (1): 11–21. doi:10.1056/NEJMoa0810457. PMC 2762083. PMID 19571279.
- ↑ 5.0 5.1 5.2 5.3 5.4 Kanigicherla D, Gummadova J, McKenzie EA, Roberts SA, Harris S, Nikam M, Poulton K, McWilliam L, Short CD, Venning M, Brenchley PE (May 2013). "Anti-PLA2R antibodies measured by ELISA predict long-term outcome in a prevalent population of patients with idiopathic membranous nephropathy". Kidney Int. 83 (5): 940–8. doi:10.1038/ki.2012.486. PMID 23364522.
- ↑ 6.0 6.1 6.2 6.3 6.4 Debiec H, Ronco P (February 2011). "PLA2R autoantibodies and PLA2R glomerular deposits in membranous nephropathy". N. Engl. J. Med. 364 (7): 689–90. doi:10.1056/NEJMc1011678. PMID 21323563.
- ↑ 7.0 7.1 7.2 7.3 7.4 Hoxha E, Kneißler U, Stege G, Zahner G, Thiele I, Panzer U, Harendza S, Helmchen UM, Stahl RA (October 2012). "Enhanced expression of the M-type phospholipase A2 receptor in glomeruli correlates with serum receptor antibodies in primary membranous nephropathy". Kidney Int. 82 (7): 797–804. doi:10.1038/ki.2012.209. PMID 22673885.
- ↑ 8.0 8.1 8.2 8.3 8.4 Svobodova B, Honsova E, Ronco P, Tesar V, Debiec H (July 2013). "Kidney biopsy is a sensitive tool for retrospective diagnosis of PLA2R-related membranous nephropathy". Nephrol. Dial. Transplant. 28 (7): 1839–44. doi:10.1093/ndt/gfs439. PMID 23223223.
-
Membranous Glomerulonephritis: Electron micrography. An excellent example to show thickened basement membrane and immune complexes.
-
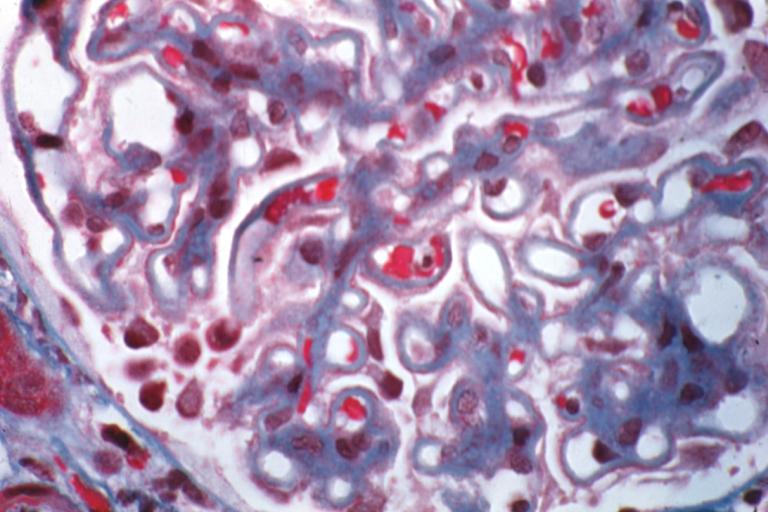
Membranous Glomerulonephritis: Micro trichrome high mag excellent to show thickened capillary basement membranes
-
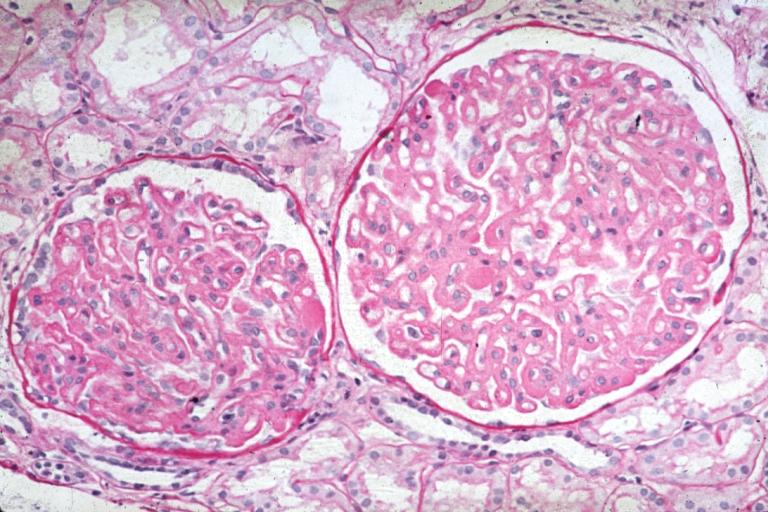
Membranous Glomerulonephritis: Micro PAS high mag excellent example of this lesion
-
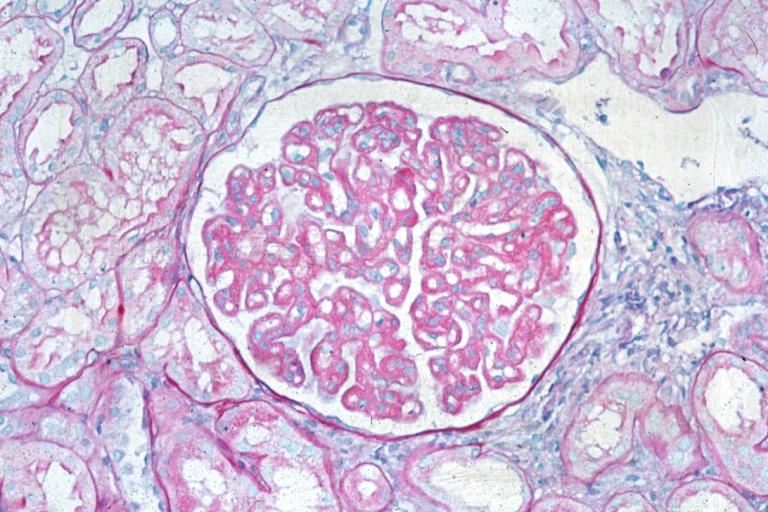
Membranous Glomerulonephritis: Micro PAS med mag