Doxycycline (oral)
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]; Associate Editor(s)-in-Chief: Kiran Singh, M.D. [2]
Disclaimer
WikiDoc MAKES NO GUARANTEE OF VALIDITY. WikiDoc is not a professional health care provider, nor is it a suitable replacement for a licensed healthcare provider. WikiDoc is intended to be an educational tool, not a tool for any form of healthcare delivery. The educational content on WikiDoc drug pages is based upon the FDA package insert, National Library of Medicine content and practice guidelines / consensus statements. WikiDoc does not promote the administration of any medication or device that is not consistent with its labeling. Please read our full disclaimer here.
Black Box Warning
|
Title
See full prescribing information for complete Boxed Warning.
ConditionName:
|
Overview
Doxycycline (oral) is a {{{drugClass}}} that is FDA approved for the treatment of {{{indication}}}. There is a Black Box Warning for this drug as shown here. Common adverse reactions include .
Adult Indications and Dosage
FDA-Labeled Indications and Dosage (Adult)
Indication
- ORACEA is indicated for the treatment of only inflammatory lesions (papules and pustules) of rosacea in adult patients. No meaningful effect was demonstrated for generalized erythema (redness) of rosacea.
Limitations of Use
- This formulation of doxycycline has not been evaluated in the treatment or prevention of infections. ORACEA should not be used for treating bacterial infections, providing antibacterial prophylaxis, or reducing the numbers or eliminating microorganisms associated with any bacterial disease.
- To reduce the development of drug-resistant bacteria as well as to maintain the effectiveness of other antibacterial drugs, ORACEA should be used only as indicated.
- Efficacy of ORACEA beyond 16 weeks and safety beyond 9 months have not been established.
- ORACEA has not been evaluated for the treatment of the erythematous, telangiectatic, or ocular components of rosacea.
Dosage
General Dosing Information
- One ORACEA Capsule (40 mg) should be taken once daily in the morning on an empty stomach, preferably at least one hour prior to or two hours after meals.
- Administration of adequate amounts of fluid along with the capsules is recommended to wash down the capsule to reduce the risk of esophageal irritation and ulceration.
Important Considerations for Dosing Regimen
- The dosage of ORACEA differs from that of doxycycline used to treat infections. Exceeding the recommended dosage may result in an increased incidence of side effects including the development of resistant organisms.
Dosage Forms And Strengths
- 40 mg beige opaque capsule imprinted with “GLD 40”
Off-Label Use and Dosage (Adult)
Guideline-Supported Use
Condition1
- Developed by:
- Class of Recommendation:
- Strength of Evidence:
- Dosing Information
- Dosage
Condition2
There is limited information regarding Off-Label Guideline-Supported Use of Doxycycline (oral) in adult patients.
Non–Guideline-Supported Use
Condition1
- Dosing Information
- Dosage
Condition2
There is limited information regarding Off-Label Non–Guideline-Supported Use of Doxycycline (oral) in adult patients.
Pediatric Indications and Dosage
FDA-Labeled Indications and Dosage (Pediatric)
Condition1
- Dosing Information
- Dosage
Condition2
There is limited information regarding FDA-Labeled Use of Doxycycline (oral) in pediatric patients.
Off-Label Use and Dosage (Pediatric)
Guideline-Supported Use
Condition1
- Developed by:
- Class of Recommendation:
- Strength of Evidence:
- Dosing Information
- Dosage
Condition2
There is limited information regarding Off-Label Guideline-Supported Use of Doxycycline (oral) in pediatric patients.
Non–Guideline-Supported Use
Condition1
- Dosing Information
- Dosage
Condition2
There is limited information regarding Off-Label Non–Guideline-Supported Use of Doxycycline (oral) in pediatric patients.
Contraindications
- This drug is contraindicated in persons who have shown hypersensitivity to doxycycline or any of the other tetracyclines.
Warnings
|
Title
See full prescribing information for complete Boxed Warning.
ConditionName:
|
Teratogenic Effects
- ORACEA should not be used during pregnancy.
- Doxycycline, like other tetracycline-class antibiotics, can cause fetal harm when administered to a pregnant woman. If any tetracycline is used during pregnancy or if the patient becomes pregnant while taking these drugs, the patient should be informed of the potential hazard to the fetus and treatment stopped immediately.
- The use of drugs of the tetracycline-class during tooth development (last half of pregnancy, infancy, and childhood up to the age of 8 years) may cause permanent discoloration of the teeth (yellow-gray-brown). This adverse reaction is more common during long-term use of the drug but has been observed following repeated short-term courses. Enamel hypoplasia has also been reported. Tetracycline drugs, therefore, should not be used during tooth development unless other drugs are not likely to be effective or are contraindicated.
- All tetracyclines form a stable calcium complex in any bone-forming tissue. A decrease in fibula growth rate has been observed in premature human infants given oral tetracycline in doses of 25 mg/kg every 6 hours. This reaction was shown to be reversible when the drug was discontinued.
- Results of animal studies indicate that tetracyclines cross the placenta, are found in fetal tissues, and can cause retardation of skeletal development on the developing fetus. Evidence of embryotoxicity has been noted in animals treated early in pregnancy.
Pseudomembranous Colitis
- Clostridium difficile associated diarrhea (CDAD) has been reported with nearly all antibacterial agents, including doxycycline, and may range in severity from mild to fatal colitis.
- Treatment with antibacterial agents alters the normal flora of the colon leading to overgrowth of C. difficile.
- C. difficile produces toxins A and B which contribute to the development of CDAD. Hypertoxin producing strains of C. difficile cause increased morbidity and mortality, as these infections can be refractory to antimicrobial therapy and may require colectomy. CDAD must be considered in all patients who present with diarrhea following antibiotic use. Careful medical history is necessary since CDAD has been reported to occur over two months after the administration of antibacterial agents.
- If CDAD is suspected or confirmed, ongoing antibiotic use not directed against C. difficile may need to be discontinued. Appropriate fluid and electrolyte management, protein supplementation, antibiotic treatment of C. difficile, and surgical evaluation should be instituted as clinically indicated.
Metabolic Effects
- The anti-anabolic action of the tetracyclines may cause an increase in BUN. While this is not a problem in those with normal renal function, in patients with significantly impaired function, higher serum levels of tetracycline-class antibiotics may lead to azotemia, hyperphosphatemia, and acidosis. If renal impairment exists, even usual oral or parenteral doses may lead to excessive systemic accumulations of the drug and possible liver toxicity. Under such conditions, lower than usual total doses are indicated, and if therapy is prolonged, serum level determinations of the drug may be advisable.
Photosensitivity
- Photosensitivity manifested by an exaggerated sunburn reaction has been observed in some individuals taking tetracyclines. Although this was not observed during the duration of the clinical studies with ORACEA, patients should minimize or avoid exposure to natural or artificial sunlight (tanning beds or UVA/B treatment) while using ORACEA. If patients need to be outdoors while using ORACEA, they should wear loose-fitting clothes that protect skin from sun exposure and discuss other sun protection measures with their physician.
Autoimmune Syndromes
- Tetracyclines have been associated with the development of autoimmune syndromes. Symptoms may be manifested by fever, rash , arthralgia, and malaise. In symptomatic patients, liver function tests, ANA, CBC, and other appropriate tests should be performed to evaluate the patients. Use of all tetracycline-class drugs should be discontinued immediately.
Tissue Hyperpigmentation
- Tetracycline-class drugs are known to cause hyperpigmentation. Tetracycline therapy may induce hyperpigmentation in many organs, including nails, bone, skin, eyes, thyroid, visceral tissue, oral cavity (teeth, mucosa, alveolar bone), sclerae and heart valves. Skin and oral pigmentation has been reported to occur independently of time or amount of drug administration, whereas other pigmentation has been reported to occur upon prolonged administration. Skin pigmentation includes diffuse pigmentation as well as over sites of scars or injury.
Pseudotumor cerebri
- Pseudotumor cerebri (benign intracranial hypertension) in adults has been associated with the use of tetracyclines. The usual clinical manifestations are headache and blurred vision. Bulging fontanels have been associated with the use of tetracyclines in infants. While both of these conditions and related symptoms usually resolve after discontinuation of the tetracycline, the possibility for permanent sequelae exists. Patients should be questioned for visual disturbances prior to initiation of treatment with tetracyclines and should be routinely checked for papiledema while on treatment.
Development of Drug Resistant Bacteria
- Bacterial resistance to tetracyclines may develop in patients using ORACEA. Because of the potential for drug-resistant bacteria to develop during the use of ORACEA, it should only be used as indicated.
Superinfection
- As with other antibiotic preparations, use of ORACEA may result in overgrowth of non-susceptible microorganisms, including fungi. If superinfection occurs, ORACEA should be discontinued and appropriate therapy instituted. Although not observed in clinical trials with ORACEA, the use of tetracyclines may increase the incidence of vaginal candidiasis. ORACEA should be used with caution in patients with a history of or predisposition to Candida overgrowth.
Laboratory Monitoring
- Periodic laboratory evaluations of organ systems, including hematopoietic, renal and hepatic studies should be performed. Appropriate tests for autoimmune syndromes should be performed as indicated.
Adverse Reactions
Clinical Trials Experience
- Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
- Adverse Reactions in Clinical Trials of ORACEA: In controlled clinical trials of adult subjects with mild to moderate rosacea, 537 subjects received ORACEA or placebo over a 16-week period. The following table summarizes selected adverse reactions that occurred in the clinical trials at a rate of >1% for the active arm:
Adverse Reactions for Tetracyclines: The following adverse reactions have been observed in patients receiving tetracyclines at higher, antimicrobial doses:
- Gastrointestinal: anorexia, nausea, vomiting, diarrhea, glossitis, dysphagia, enterocolitis, and inflammatory lesions (with vaginal candidiasis) in the anogenital region. Hepatotoxicity has been reported rarely. Rare instances of esophagitis and esophageal ulcerations have been reported in patients receiving the capsule forms of the drugs in the tetracycline-class. Most of the patients experiencing esophagitis and/or esophageal ulceration took their medication immediately before lying down.
- Skin: maculopapular and erythematous rashes. Exfoliative dermatitis has been reported but is uncommon. Photosensitivity is discussed above .
- Renal toxicity: Rise in BUN has been reported and is apparently dose-related.
- Hypersensitivity reactions: urticaria , angioneurotic edema, anaphylaxis, anaphylactoid purpura, serum sickness, pericarditis, and exacerbation of systemic lupus erythematosus.
- Blood: Hemolytic anemia, thrombocytopenia, neutropenia, and eosinophilia have been reported.
Postmarketing Experience
- Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure. The following adverse reactions have been identified during postapproval use of ORACEA.
• Nervous system: Pseudotumor ceribri (benign intracranial hypertension), headache
Drug Interactions
Anticoagulants
- Because tetracyclines have been shown to depress plasma prothrombin activity, patients who are on anticoagulant therapy may require downward adjustment of their anticoagulant dosage.
Penicillin
- Since bacteriostatic drugs may interfere with the bactericidal action of penicillin, it is advisable to avoid giving tetracycline-class drugs in conjunction with penicillin.
Methoxyflurane
- The concurrent use of tetracycline and methoxyflurane has been reported to result in fatal renal toxicity.
Antacids and Iron Preparations
- Absorption of tetracyclines is impaired by bismuth subsalicylate, proton pump inhibitors, antacids containing aluminum, calcium or magnesium and iron-containing preparations.
Low Dose Oral Contraceptives
- Doxycycline may interfere with the effectiveness of low dose oral contraceptives. To avoid contraceptive failure, females are advised to use a second form of contraceptive during treatment with doxycycline.
Oral Retinoids
- There have been reports of pseudotumor cerebri (benign intracranial hypertension) associated with the concomitant use of isotretinoin and tetracyclines. Since both oral retinoids, including isotretinoin and acitretin, and the tetracyclines, primarily minocycline, can cause increased intracranial pressure, the concurrent use of an oral retinoid and a tetracycline should be avoided.
Barbiturates and Anti-epileptics
- Barbiturates, carbamazepine, and phenytoin decrease the half-life of doxycycline.
Drug/Laboratory Test Interactions
- False elevations of urinary catecholamine levels may occur due to interference with the fluorescence test
Use in Specific Populations
Pregnancy
- Teratogenic Effects: Pregnancy Category D. Results from animal studies indicate that doxycycline crosses the placenta and is found in fetal tissues.
Pregnancy Category (AUS):
There is no Australian Drug Evaluation Committee (ADEC) guidance on usage of doxycycline in women who are pregnant.
Labor and Delivery
There is no FDA guidance on use of doxycycline during labor and delivery.
Nursing Mothers
- Tetracyclines are excreted in human milk. Because of the potential for serious adverse reactions in infants from doxycycline, ORACEA should not be used in mothers who breastfeed.
Pediatric Use
- ORACEA should not be used in infants and children less than 8 years of age. ORACEA has not been studied in children of any age with regard to safety or efficacy, therefore use in children is not recommended.
Geriatic Use
- Clinical studies of ORACEA did not include sufficient numbers of subjects aged 65 and over to determine whether they respond differently from younger subjects. Other reported clinical experience has not identified differences in responses between elderly and younger patients. In general, dose selection for an elderly patient should be cautious, usually starting at the low end of the dosing range, reflecting the greater frequency of decreased hepatic, renal, or cardiac function, and concomitant disease or other drug therapy.
Gender
There is no FDA guidance on the use of doxycycline with respect to specific gender populations.
Race
There is no FDA guidance on the use of doxycycline with respect to specific racial populations.
Renal Impairment
There is no FDA guidance on the use of doxycycline in patients with renal impairment.
Hepatic Impairment
There is no FDA guidance on the use of doxycycline in patients with hepatic impairment.
Females of Reproductive Potential and Males
There is no FDA guidance on the use of doxycycline in women of reproductive potentials and males.
Immunocompromised Patients
There is no FDA guidance one the use of doxycycline in patients who are immunocompromised.
Administration and Monitoring
Administration
- Oral
- Intravenous
Monitoring
There is limited information regarding Monitoring of Doxycycline (oral) in the drug label.
- Description
IV Compatibility
There is limited information regarding IV Compatibility of Doxycycline (oral) in the drug label.
Overdosage
- In case of overdosage, discontinue medication, treat symptomatically, and institute supportive measures. Dialysis does not alter serum half-life and thus would not be of benefit in treating cases of overdose.
Pharmacology
There is limited information regarding Doxycycline (oral) Pharmacology in the drug label.
Mechanism of Action
- The mechanism of action of ORACEA in the treatment of inflammatory lesions of rosacea is unknown.
Structure
- ORACEA (doxycycline, USP) Capsules 40 mg are hard gelatin capsule shells filled with two types of doxycycline beads (30 mg immediate release and 10 mg delayed release) that together provide a dose of 40 mg of anhydrous doxycycline (C22H24N2O8).
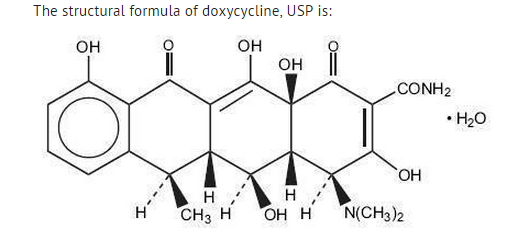
- with an empirical formula of C22H24N2O8•H2O and a molecular weight of 462.46. The chemical designation for doxycycline is 2-Naphthacenecar-boxamide,4-(dimethylamino)-1,4,4a,5,5a,6,11,12a-octahydro-3,5,10,12,12a-pentahydroxy-6-methyl-1,11-dioxo-, [4S-(4α, 4aα, 5α, 5aα, 6α,12aα)]-, monohydrate. It is very slightly soluble in water.
- Inert ingredients in the formulation are: hypromellose, iron oxide red, iron oxide yellow, methacrylic acid copolymer, polyethylene glycol, Polysorbate 80, sugar spheres, talc, titanium dioxide, and triethyl citrate. Active ingredients: Each capsule contains doxycycline, USP in an amount equivalent to 40 mg of anhydrous doxycycline.
Pharmacodynamics
There is limited information regarding pharmacodynamics of doxycycline in the drug label.
Pharmacokinetics
- ORACEA capsules are not bioequivalent to other doxycycline products. The pharmacokinetics of doxycycline following oral administration of ORACEA was investigated in 2 volunteer studies involving 61 adults. Pharmacokinetic parameters for ORACEA following single oral doses and at steady-state in healthy subjects are presented in Table 2.
- Absorption: In a single-dose food-effect study involving administration of ORACEA to healthy volunteers, concomitant administration with a 1000 calorie, high-fat, high-protein meal that included dairy products, resulted in a decrease in the rate and extent of absorption (Cmax and AUC) by about 45% and 22%, respectively, compared to dosing under fasted conditions. This decrease in systemic exposure can be clinically significant, and therefore if ORACEA is taken close to meal times, it is recommended that it be taken at least one hour prior to or two hours after meals.
- Distribution: Doxycycline is greater than 90% bound to plasma proteins.
- Metabolism: Major metabolites of doxycycline have not been identified. However, enzyme inducers such as barbiturates, carbamazepine, and phenytoin decrease the half-life of doxycycline.
- Excretion: Doxycycline is excreted in the urine and feces as unchanged drug. It is reported that between 29% and 55.4% of an administered dose can be accounted for in the urine by 72 hours. Terminal half-life averaged 21.2 hours in subjects receiving a single dose of ORACEA.
Special Populations
- Geriatric: Doxycycline pharmacokinetics have not been evaluated in geriatric patients.
- Pediatric: Doxycycline pharmacokinetics have not been evaluated in pediatric patients.
- Gender: The pharmacokinetics of ORACEA were compared in 16 male and 14 female subjects under fed and fasted conditions. While female subjects had a higher Cmax and AUC than male subjects, these differences were thought to be due to differences in body weight/lean body mass.
- Race: Differences in doxycycline pharmacokinetics among racial groups have not been evaluated.
- Renal Insufficiency: Studies have shown no significant difference in serum half-life of doxycycline in patients with normal and severely impaired renal function. Hemodialysis does not alter the serum half-life of doxycycline.
- Hepatic Insufficiency: Doxycycline pharmacokinetics have not been evaluated in patients with hepatic insufficiency.
- Gastric Insufficiency:In a study in healthy volunteers (N=24) the bioavailability of doxycycline is reported to be reduced at high pH. This reduced bioavailability may be clinically significant in patients with gastrectomy, gastric bypass surgery or who are otherwise deemed achlorhydric.
Drug Interactions:
Nonclinical Toxicology
There is limited information regarding Nonclinical Toxicology of Doxycycline (oral) in the drug label.
Clinical Studies
- The safety and efficacy of ORACEA in the treatment of only inflammatory lesions (papules and pustules) of rosacea was evaluated in two randomized, placebo-controlled, multi-centered, double-blind, 16-week Phase 3 trials involving 537 subjects (total of 269 subjects on ORACEA from the two trials) with rosacea (10 to 40 papules and pustules and two or fewer nodules). Pregnant and nursing women, subjects <18 years of age, and subjects with ocular rosacea and/or blepharitis/meibomianitis who require ophthalmologic treatment were excluded from trial. Mean baseline lesion counts were 20 and 21 for ORACEA and placebo subject groups respectively.
- At Week 16, subjects in the ORACEA group were evaluated using co-primary endpoints of mean reduction in lesion counts and a dichotomized static Investigators Global Assessment of Clear or Almost Clear (defined as 1 to 2 small papules or pustules) when compared to the placebo group in both Phase 3 trials.
- Subjects treated with ORACEA did not demonstrate significant improvement in erythema when compared to those treated with placebo.
How Supplied
- ORACEA (beige opaque capsule imprinted with “GLD 40”) containing doxycycline, USP in an amount equivalent to 40 mg of anhydrous doxycycline. Bottle of 30 (NDC 0299-3822-30).
Storage
- All products are to be stored at controlled room temperatures of 15°C - 30°C (59°F - 86°F) and dispensed in tight, light-resistant containers (USP).
- Keep out of reach of children
Images
Drug Images
{{#ask: Page Name::Doxycycline (oral) |?Pill Name |?Drug Name |?Pill Ingred |?Pill Imprint |?Pill Dosage |?Pill Color |?Pill Shape |?Pill Size (mm) |?Pill Scoring |?NDC |?Drug Author |format=template |template=DrugPageImages |mainlabel=- |sort=Pill Name }}
Package and Label Display Panel
{{#ask: Label Page::Doxycycline (oral) |?Label Name |format=template |template=DrugLabelImages |mainlabel=- |sort=Label Page }}
Patient Counseling Information
Patients taking ORACEA Capsules 40 mg should receive the following information and instructions:
- It is recommended that ORACEA not be used by individuals of either gender who are attempting to conceive a child
- It is recommended that ORACEA not be used by pregnant or breast feeding women.
- Patients should be advised that pseudomembranous colitis can occur with doxycycline therapy. If patients develop watery or bloody stools, they should seek medical attention.
- Patients should be advised that pseudotumor cerebri can occur with doxycycline therapy. If patients experience headache or blurred vision they should seek medical attention.
- Photosensitivity manifested by an exaggerated sunburn reaction has been observed in some individuals taking tetracyclines, including doxycycline. Patients should minimize or avoid exposure to natural or artificial sunlight (tanning beds or UVA/B treatment) while using doxycycline. If patients need to be outdoors while using doxycycline, they should wear loose-fitting clothes that protect skin from sun exposure and discuss other sun protection measures with their physician. Treatment should be discontinued at the first evidence of sunburn.
- Concurrent use of doxycycline may render oral contraceptives less effective.
- Autoimmune syndromes, including drug-induced lupus-like syndrome, autoimmune hepatitis, vasculitis and serum sickness have been observed with tetracycline-class drugs, including doxycycline. Symptoms may be manifested by arthralgia, fever, rash and malaise. Patients who experience such symptoms should be cautioned to stop the drug immediately and seek medical help.
- Patients should be counseled about discoloration of skin, scars, teeth or gums that can arise from doxycycline therapy.
- Take ORACEA exactly as directed. Increasing doses beyond 40 mg every morning may increase the likelihood that bacteria will develop resistance and will not be treatable by other antibacterial drugs in the future.
Precautions with Alcohol
- Alcohol- doxycycline interaction has not been established. Talk to your doctor about the effects of taking alcohol with this medication.
Brand Names
- ®[1]
Look-Alike Drug Names
- A® — B®[2]
Drug Shortage Status
Price
References
The contents of this FDA label are provided by the National Library of Medicine.
- ↑ Empty citation (help)
- ↑ "http://www.ismp.org". External link in
|title=(help)
{{#subobject:
|Page Name=Doxycycline (oral)
|Pill Name=No image.jpg
|Drug Name=
|Pill Ingred=|+sep=;
|Pill Imprint=
|Pill Dosage={{{dosageValue}}} {{{dosageUnit}}}
|Pill Color=|+sep=;
|Pill Shape=
|Pill Size (mm)=
|Pill Scoring=
|Pill Image=
|Drug Author=
|NDC=
}}
{{#subobject:
|Label Page=Doxycycline (oral) |Label Name=Doxycycline (oral)11.png
}}
{{#subobject:
|Label Page=Doxycycline (oral) |Label Name=Doxycycline (oral)11.png
}}