Alirocumab: Difference between revisions
Tarek Nafee (talk | contribs) |
Tarek Nafee (talk | contribs) No edit summary |
||
| Line 158: | Line 158: | ||
* Moderate to high CV risk with LDL>100mg/dl | * Moderate to high CV risk with LDL>100mg/dl | ||
* Very high CV risk with LDL>70mg/dl | * Very high CV risk with LDL>70mg/dl | ||
* Washout of lipid lowering therapy for 2 weeks prior | |||
|% change in LDL at week 24 in ITT population compared to ezetimibe | |% change in LDL at week 24 in ITT population compared to ezetimibe | ||
|% change in LDL at week 12 and 24 in "on-treatment" population compared to ezetimibe | |% change in LDL at week 12 and 24 in "on-treatment" population compared to ezetimibe | ||
|All Adverse events during the study and the optional open label study extension | |All Adverse events during the study and the optional open label study extension | ||
|2:2:1 | |2:2:1 | ||
|75mg SQ Alirocumab | |75mg SQ Alirocumab Q2W | ||
|Ezetimibe 10mg PO QD | |Ezetimibe 10mg PO QD | ||
| Line 186: | Line 187: | ||
|103 | |103 | ||
| | | | ||
* Patients > 18 years | |||
* 10-year risk of fatal CV event ≥ 1% and <5%. | |||
* LDL>100mg/dl | |||
* No Hx of established CHD or CHD risk equivalents | |||
* Not receiving lipid lowering therapy for 4 weeks prior | |||
|% change in LDL at week 24 in ITT population | |||
|% change in LDL at week 12 in ITT population | |||
|AE reporting up to 10 weeks after last dose | |||
|1:1 | |||
|75mg SQ Alirocumab Q2W | |||
increased to 150 mg if LDL ≥100 mg/dl by week 12 | |||
(70mg/dl was utilized instead of 100mg/dl due to admin error) | |||
|Ezetimibe 10mg PO QD | |||
|24 weeks | |||
|8 weeks | |||
|<nowiki>-47.2% vs. -15.6% (Δ:31.6%; 95% CI: 23.0-40.2; p<0.0001)</nowiki> | |||
| | | | ||
* Substantial drop in first 4 weeks of treatment and sustained for 24 weeks. | |||
* On-treatment analysis demonstrated similar results (numerically larger difference) | |||
* More AEs in ezetemibe arm compared to | |||
* Disposable autoinjector resulted in low rate of injection related AEs | |||
|- | |- | ||
|FHI | |FHI | ||
| Line 202: | Line 218: | ||
|III | |III | ||
|486 | |486 | ||
| rowspan="2" | | |||
* Adult patients | |||
* HeFH patients | |||
* LDL ≥ 70 mg/dL if no hx of CVD | |||
* LDL ≥ 100 mg/dL if hx of CVD | |||
* Not receiving lipid lowering therapy for 4 weeks prior | |||
| rowspan="2" |% change in LDL at week 24 in ITT population | |||
| | | | ||
| | | | ||
| | | rowspan="2" |2:1 | ||
| | | rowspan="2" |75mg SQ Alirocumab Q2W | ||
| | |||
| | increased to 150 mg if LDL ≥70mg/dl by week 12 | ||
| | | rowspan="2" |Placebo | ||
| | | rowspan="2" |78 weeks | ||
| | | rowspan="2" |8 weeks | ||
| | |<nowiki>-48.8% vs. 9.1% (Δ:57.9%; 95% CI: 52.6-63.3; p<0.0001)</nowiki> | ||
| | | | ||
|- | |- | ||
| Line 221: | Line 244: | ||
| | | | ||
| | | | ||
| | |<nowiki>-48.7% vs. 2.8% (Δ:51.4%; 95% CI: 44.8-58.1; p<0.0001)</nowiki> | ||
| | | | ||
|- | |- | ||
| Line 243: | Line 259: | ||
|% change in LDL at week 24 in ITT population | |% change in LDL at week 24 in ITT population | ||
|% change in LDL at week 12 and 24 in "on-treatment" population | |% change in LDL at week 12 and 24 in "on-treatment" population | ||
|Adverse Events up to 10 weeks | |Adverse Events up to 10 weeks after last dose | ||
|2:1 | |2:1 | ||
|Single 150mg SQ Alirocumab | |Single 150mg SQ Alirocumab Q2W | ||
|Matched | |Matched | ||
Revision as of 20:18, 14 December 2016
For a review of all PCSK9 inhibitors please click here Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]; Associate Editor(s)-in-Chief: Tarek Nafee, M.D. [2]
Overview
Alirocumab (REGN727 and SAR236553) is an investigational human monoclonal antibody that inhibits PCSK9 for the treatment of hypercholesterolemia.
Mechanism of Action
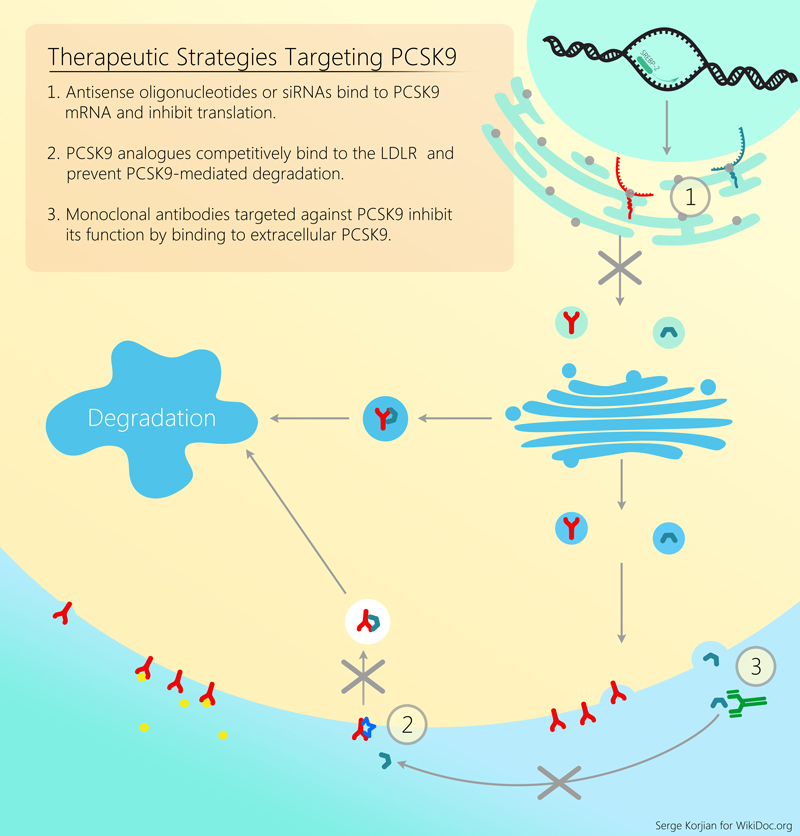
Alirocumab is a human monoclonal antibody that binds proprotein convertase subtilisin/kexin type 9 (PCSK9). PCSK9 is a protein that attaches to surface LDL receptors and triggers their degradation. This reduces the ability of hepatocytes to uptake LDL cholesterol and subsequently leads to increased levels of circulating LDL. By binding PCSK9, alirocumab inhibits LDL receptor destruction and increases the endocytosis of LDL from the circulation. Pre-clinical studies, and early clinical trials have shown the efficacy and safety of alirocumab in decreasing LDL cholesterol as an add-on agent or as monotherapy.
Approved Indications
In July 2015, the FDA approved the drug Praluent (alirocumab) for patients who have heterozygous familial hypercholesterolemia (FH) and high-risk patients who have had a stroke or myocardial infarction in the past and cannot take statins because of negative side effects. The EU is expected to make a decision by the end of September 2015.
Contraindications
Investigational Indications
Known Adverse Effects
Major Trials
Synopsis
| Trial Name | Year | NCT Identifier | Phase | N | Study Population | Primary Endpoint | Secondary Endpoints | Safety Endpoints | Randomization | Intervention
Arm |
Control
Arm(s) |
Study Duration | Follow up | Main Study Findings | Other Findings |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| NCT 01288469 | II | 92 | |||||||||||||
| NCT 01266876 | II | 77 | |||||||||||||
| NCT 01288443 | II | 183 | |||||||||||||
| COMBO I | 2012-2013 | NCT 01644175 | III | 316 |
|
% change in LDL at week 24 in ITT population | % change in LDL at different timepoints in "on-treatment" population | AE reporting | 2:1 | 75mg SQ Alirocumab Q2W
increased to 150 mg if LDL ≥70mg/dl by week 12 |
Matched
Placebo |
52 weeks | 8 weeks | -48.2% vs. -2.3% (Δ:45.9%; 95% CI: 39.3-52.5; p<0.0001) |
|
| COMBO II | 2012-2013 | NCT 01644188 | III | 720 |
|
% change in LDL at week 24 in ITT population | % change in LDL at week 12(ITT or OT), 24 (OT), 52 (ITT or OT) | AE reporting | 2:1 | 75mg SQ Alirocumab Q2W
increased to 150 mg if LDL ≥70mg/dl by week 12 |
Ezetimibe 10mg PO QD | 104 weeks | 8 weeks | -50.6% vs. -20.7% (Δ:29.8%; 95% CI: 25.3-34.4; p<0.0001) |
|
| ALTERNATIVE | 2012-2013 | NCT 01709513 | III | 314 |
|
% change in LDL at week 24 in ITT population compared to ezetimibe | % change in LDL at week 12 and 24 in "on-treatment" population compared to ezetimibe | All Adverse events during the study and the optional open label study extension | 2:2:1 | 75mg SQ Alirocumab Q2W | Ezetimibe 10mg PO QD
or Atorvastatin 20mg PO QD |
24 weeks | 8 week follow up with option to extend to 3 year open label study | -45.0% vs. -14.6% (Δ:30.4%; 95% CI: 24.2-36.6; p<0.0001) |
|
| MONO | 2012-2013 | NCT 01644474 | III | 103 |
|
% change in LDL at week 24 in ITT population | % change in LDL at week 12 in ITT population | AE reporting up to 10 weeks after last dose | 1:1 | 75mg SQ Alirocumab Q2W
increased to 150 mg if LDL ≥100 mg/dl by week 12 (70mg/dl was utilized instead of 100mg/dl due to admin error) |
Ezetimibe 10mg PO QD | 24 weeks | 8 weeks | -47.2% vs. -15.6% (Δ:31.6%; 95% CI: 23.0-40.2; p<0.0001) |
|
| FHI | 2012-2014 | NCT 01623115 | III | 486 |
|
% change in LDL at week 24 in ITT population | 2:1 | 75mg SQ Alirocumab Q2W
increased to 150 mg if LDL ≥70mg/dl by week 12 |
Placebo | 78 weeks | 8 weeks | -48.8% vs. 9.1% (Δ:57.9%; 95% CI: 52.6-63.3; p<0.0001) | |||
| FHII | 2012-2014 | NCT 01709500 | III | 249 | -48.7% vs. 2.8% (Δ:51.4%; 95% CI: 44.8-58.1; p<0.0001) | ||||||||||
| Long Term | 2012-2014 | NCT 01507831 | III | 2314 |
|
% change in LDL at week 24 in ITT population | % change in LDL at week 12 and 24 in "on-treatment" population | Adverse Events up to 10 weeks after last dose | 2:1 | Single 150mg SQ Alirocumab Q2W | Matched
Placebo |
78 weeks | 8 weeks | -61.0% vs. 0.8% (Δ:61.9%; 95% CI: 59.4-64.3; p<0.001) | Similar rates of any Adverse event between the groups. |
| OPTIONS I | 2012-2014 | NCT 01730040 | III | 355 | |||||||||||
| OPTIONS II | 2012-2014 | NCT 01730053 | III | 305 | |||||||||||
| CHOICE I | 2013-2014 | NCT 01926782 | III | 803 | |||||||||||
| CHOICE II | 2013-2014 | NCT 02023879 | III | 233 | |||||||||||
| HIGH FH | 2012-2015 | NCT 01617655 | III | 107 | |||||||||||
| JAPAN | 2014-2015 | NCT 02107898 | III | 216 | |||||||||||
| ESCAPE | 2015 | NCT 02326220 | III | 62 |
Phase II Trials
Safety and efficacy of atorvastatin with or without alirocumab in primary hypercholesterolemia (NCT 01288469)
This randomized, double-blind, placebo-controlled phase 2 trial of 92 patients with LDL-C≥100 mg/dL after treatment with 10 mg of atorvastatin for at least 7 weeks randomized patients to 8 weeks of therapy with either 80 mg of atorvastatin daily plus 150 mg SC alirocumab once every 2 weeks, 10 mg of atorvastatin daily plus 150 mg alirocumab once every 2 weeks, or 80 mg of atorvastatin daily plus SC placebo once every 2 weeks. The trial demonstrated a significant 73.2% reduction from baseline serum LDL-C cholesterol with 80 mg of atorvastatin plus alirocumab compared with 17.3% with the 80 mg atorvastatin plus placebo. Ninety percent of the patients who received alirocumab reached LDL-C concentrations lower than 70 mg/dL compared with 17% of those receiving atorvastatin alone. There were no significant safety signals and the drug was well tolerated.[2]
Safety and efficacy in heterozygous familial hypercholesterolaemia with ongoing stable-dose statin with or without ezetimibe therapy (NCT 01266876)
This randomized, double-blind, placebo-controlled phase 2 trial of 77 adults with heterozygous familial hypercholesterolaemia and LDL-C concentrations of ≥100 mg/dL or higher on stable diet and statin dose, with or without ezetimibe therapy were randomized (1:1:1:1:1) to 5 different treatment arms for 12 weeks: 150 mg SC alirocumab every 4 weeks, 200 mg SC alirocumab every 4 weeks, 300 mg SC alirocumab every 4 weeks, 150 mg SC alirocumab every 2 weeks, or SC placebo. Randomization was stratified by baseline use of ezetimibe. Alirocumab demonstrated a significant reduction in LDL-C at week 12 (28.9%, 31.5%, and 42.5% for the 150, 200, and 300 mg every 4 weeks respectively, and 67.9% for the 150 mg every 2 weeks dose, compared with 10.7% in the placebo arm) with no significant safety signal. There were no increases >3 x ULN in hepatic transaminases or creatine kinase (CK).[3]
Safety and efficacy in primary hypercholesterolemia with ongoing stable atorvastatin therapy (NCT 01288443)
This randomized, double-blind, placebo-controlled phase 2 trial of 183 patients with LDL-C ≥100 mg/dL on stable-dose atorvastatin for 6 or more weeks randomized patients to a 12 week treatment course in either one of 6 arms (1:1:1:1:1:1): subcutaneous (SC) placebo every 2 weeks, 50 mg SC alirocumab every 2 weeks, 100 mg SC alirocumab every 2 weeks, 150 mg SC alirocumab every 2 weeks, 200 mg SC alirocumab every 4 weeks alternating with placebo, or 300 mg SC alirocumab every 4 weeks alternating with placebo. Alirocumab demonstrated a significant dose-related reduction in serum LDL-C (40%, 64%, and 72% with 50, 100, and 150 mg respectively, and 43% and 48% with 200 and 300 mg respectively compared with 5% in placebo) with no major safety signals. [4]
Phase III Trials
ODYSSEY - COMBO II
COMBO II was a randomized, double-blind, double-dummy, trial that randomized 720 patients with elevated CV risk and LDL-C despite maximal statins use to either SC alirocumab 75 mg every 2 weeks (and PO placebo) or PO ezetimibe 10 mg daily (and SC placebo). Alirocumab was generally well tolerated, with no reported safety signals. Alirocumab treatment was associated with a significantly higher reduction in mean LDL-C values from baselines at week 24 (50.6 ± 1.4% for alirocumab vs. 20.7 ± 1.9% for ezetimibe; P < 0.0001). Patients on alirocumab were more likely to achieve LDL-C <1.8 mmol/L (77.0% vs. 45.6%; P < 0.0001). At week 24, mean LDL-C levels were 1.3 ± 0.04 mmol/L among patients receiving alirocumab, and 2.1 ± 0.05 mmol/L among patients receiving ezetimibe.[5]
ODYSSEY - FH I & FH II
ODYSSEY FHI and FH II were multicenter, double-blind, placebo-controlled trials that enrolled a total of 735 heterozygous familial hypercholesterolemia patients and randomized them to either SC alirocumab 75-150 mg every 2 weeks or matching placebo for a total of 78 weeks, on top of a background of lipid lowering therapy.The primary endpoint was the prrcent change in LDL-C from baseline to week 24. Alirocumab administration was associated with a 57.9% reduction compared to placebo in the FH I population (P<0.0001), and a 51.4% reduction compared to placebo in the FH II population (P<0.0001). Alirocumab was well tolerated and there were no safety concerns.
ODYSSEY - Long Term
Long term was a randomized, double-blind, placebo-controlled trial that enrolled 2341 patients at high risk for CV events with baseline LDL-C ≥70 mg/dL. Patients were randomized in 2:1 ratio to receive either 150mg SC alirocumab or equivalent placebo once every 2 weeks for 78 weeks. The primary efficacy end point was the percentage change in calculated LDL cholesterol level from baseline to week 24. Alirocumab treatment was associated with a 62% reduction in LDL-C compared to placebo (P<0.001). In an exploratory analysis, alirocumab was associated with a significant reduction in the rate of major adverse cardiovascular events (1.7% vs. 3.3%; hazard ratio, 0.52; 95% confidence interval, 0.31 to 0.90; nominal P=0.02). Alirocumab treatment was associated with a significantly higher incidence of myalgia (5.4% vs. 2.9%; P=0.006). [6]
Cost-Effectiveness
Doses are administered every two weeks with a cost of $40 a day or $14,600 a year, substantially higher than some generic statins, which can cost as little as $0.10 a day. Praluent is more expensive to manufacture than statins because it is made in live genetically engineered cells. Manufacturers argue that the drug is cost-effective because it will reduce medical costs of hospitalizations from stroke or myocardial infarction and that the price of the drug reflects its value. Praluent used in combination with statins can lower cholesterol 40-70% [6] compared to statins that lower LDL an average of 40% [7]. Still, further research into the actual ability of the drug to reduce risk and complications is ongoing. Reduced prices and plans through insurers should help make the drug accessible to patients with lower ability to pay.
Future Investigations
OUTCOMES***
DM DYSLIPIDEMIA
DM INSULIN
KT
References
- ↑ Urban, D.; Pöss, J.; Böhm, M.; Laufs, U. (2013). "Targeting the proprotein convertase subtilisin/kexin type 9 for the treatment of dyslipidemia and atherosclerosis". J Am Coll Cardiol. 62 (16): 1401–8. doi:10.1016/j.jacc.2013.07.056. PMID 23973703. Unknown parameter
|month=ignored (help) - ↑ Roth EM, McKenney JM, Hanotin C, Asset G, Stein EA (2012). "Atorvastatin with or without an antibody to PCSK9 in primary hypercholesterolemia". N Engl J Med. 367 (20): 1891–900. doi:10.1056/NEJMoa1201832. PMID 23113833.
- ↑ Stein EA, Gipe D, Bergeron J, Gaudet D, Weiss R, Dufour R; et al. (2012). "Effect of a monoclonal antibody to PCSK9, REGN727/SAR236553, to reduce low-density lipoprotein cholesterol in patients with heterozygous familial hypercholesterolaemia on stable statin dose with or without ezetimibe therapy: a phase 2 randomised controlled trial". Lancet. 380 (9836): 29–36. doi:10.1016/S0140-6736(12)60771-5. PMID 22633824.
- ↑ McKenney JM, Koren MJ, Kereiakes DJ, Hanotin C, Ferrand AC, Stein EA (2012). "Safety and efficacy of a monoclonal antibody to proprotein convertase subtilisin/kexin type 9 serine protease, SAR236553/REGN727, in patients with primary hypercholesterolemia receiving ongoing stable atorvastatin therapy". J Am Coll Cardiol. 59 (25): 2344–53. doi:10.1016/j.jacc.2012.03.007. PMID 22463922.
- ↑ Cannon CP, Cariou B, Blom D, McKenney JM, Lorenzato C, Pordy R; et al. (2015). "Efficacy and safety of alirocumab in high cardiovascular risk patients with inadequately controlled hypercholesterolaemia on maximally tolerated doses of statins: the ODYSSEY COMBO II randomized controlled trial". Eur Heart J. doi:10.1093/eurheartj/ehv028. PMID 25687353.
- ↑ 6.0 6.1 Robinson JG, Farnier M, Krempf M, Bergeron J, Luc G, Averna M; et al. (2015). "Efficacy and Safety of Alirocumab in Reducing Lipids and Cardiovascular Events". N Engl J Med. doi:10.1056/NEJMoa1501031. PMID 25773378.
- ↑ Anand SS (2003). "Quantifying effect of statins on low density lipoprotein cholesterol, ischaemic heart disease, and stroke: systematic review and meta-analysis. Law MR, Wald NJ, Rudnicka AR. BMJ 2003; 326: 1407-408". Vasc Med. 8 (4): 289–90. PMID 15125495.