Elvitegravir cobicistat emtricitabine tenofovir description
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]; Associate Editor(s)-in-Chief: Mohamed Moubarak, M.D. [2]
Description
STRIBILD is a fixed-dose combination tablet containing elvitegravir, cobicistat, emtricitabine, and tenofovir DF for oral administration.
Elvitegravir is an HIV-1 integrase strand transfer inhibitor. Cobicistat is a mechanism-based inhibitor of cytochrome P450 (CYP) enzymes of the CYP3A family. Emtricitabine is a synthetic nucleoside analog of cytidine. EMTRIVA is the brand name for emtricitabine. Tenofovir DF is converted in vivo to tenofovir, an acyclic nucleoside phosphonate (nucleotide) analog of adenosine 5'-monophosphate. VIREAD is the brand name for tenofovir DF.
Each tablet contains 150 mg of elvitegravir, 150 mg of cobicistat, 200 mg of emtricitabine, and 300 mg of tenofovir DF (equivalent to 245 mg of tenofovir disoproxil). The tablets include the following inactive ingredients: lactose monohydrate, microcrystalline cellulose, silicon dioxide, croscarmellose sodium, hydroxypropyl cellulose, sodium lauryl sulfate, and magnesium stearate. The tablets are film-coated with a coating material containing indigo carmine (FD&C Blue #2) aluminum lake, polyethylene glycol, polyvinyl alcohol, talc, titanium dioxide, and yellow iron oxide.
Elvitegravir: The chemical name of elvitegravir is 6-(3-Chloro-2-fluorobenzyl)-1-[(2S)-1-hydroxy-3-methylbutan-2-yl]-7-methoxy-4-oxo-1,4-dihydroquinoline-3-carboxylic acid.
It has a molecular formula of C23H23ClFNO5 and a molecular weight of 447.9. It has the following structural formula:
Elvitegravir is a white to pale yellow powder with a solubility of less than 0.3 micrograms per mL in water at 20 °C.
Cobicistat: The chemical name for cobicistat is 1,3-thiazol-5-ylmethyl [(2R,5R)-5-{[(2S)-2-[(methyl{[2-(propan-2-yl)-1,3-thiazol-4-yl]methyl}carbamoyl)amino]-4-(morpholin-4-yl)butanoyl]amino}-1,6-diphenylhexan-2-yl]carbamate.
It has a molecular formula of C40H53N7O5S2 and a molecular weight of 776.0. It has the following structural formula:
Cobicistat is adsorbed onto silicon dioxide. Cobicistat on silicon dioxide is a white to pale yellow solid with a solubility of 0.1 mg per mL in water at 20 °C.
Emtricitabine: The chemical name of emtricitabine is 5-fluoro-1-[(2R,5S)-2-(hydroxymethyl)-1,3-oxathiolan-5-yl]cytosine. Emtricitabine is the (-)enantiomer of a thio analog of cytidine, which differs from other cytidine analogs in that it has a fluorine in the 5-position.
It has a molecular formula of C8H10FN3O3S and a molecular weight of 247.25. It has the following structural formula:
Emtricitabine is a white to off-white crystalline powder with a solubility of approximately 112 mg per mL in water at 25 °C.
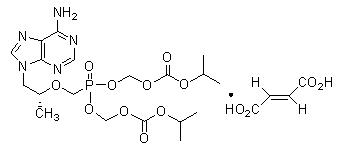
Tenofovir Disoproxil Fumarate: Tenofovir DF is a fumaric acid salt of the bis-isopropoxycarbonyloxymethyl ester derivative of tenofovir. The chemical name of tenofovir DF is 9-[(R)-2-[[bis[[(isopropoxycarbonyl)oxy]-methoxy]phosphinyl]methoxy]propyl]adenine fumarate (1:1). It has a molecular formula of C19H30N5O10P ∙ C4H4O4 and a molecular weight of 635.51. It has the following structural formula:
Tenofovir DF is a white to off-white crystalline powder with a solubility of 13.4 mg per mL in water at 25 °C. All dosages are expressed in terms of tenofovir DF except where otherwise noted.[1]
References
Adapted from the FDA Package Insert.