Elvitegravir cobicistat emtricitabine tenofovir contraindications
Jump to navigation
Jump to search
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]; Associate Editor(s)-in-Chief: Mohamed Moubarak, M.D. [2]
Contraindications
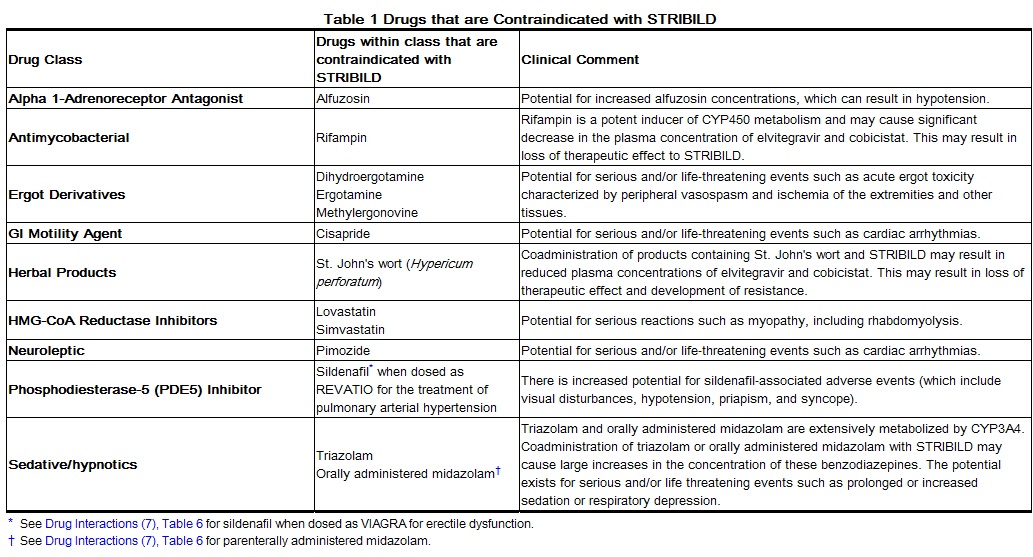
Coadministration of STRIBILD is contraindicated with drugs that are highly dependent on CYP3A for clearance and for which elevated plasma concentrations are associated with serious and/or life-threatening events. These drugs and other contraindicated drugs (which may lead to reduced efficacy of STRIBILD and possible resistance) are listed in Table 1.[1]
References
Adapted from the FDA Package Insert.