Gatifloxacin labels and packages
Jump to navigation
Jump to search
| Gatifloxacin |
|---|
| GATIFLOXACIN®,ZYMAXID®,ZYMAR® FDA Package Insert |
| Description |
| Clinical Pharmacology |
| Microbiology |
| Indications and Usage |
| Contraindications |
| Warnings and Precautions |
| Adverse Reactions |
| Clinical Studies |
| Dosage and Administration |
| How Supplied |
| Labels and Packages |
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1];Associate Editor(s)-in-Chief: Abdurahman Khalil, M.D. [2]
For patient information, click here.
© 2012 Allergan, Inc. Irvine, CA 92612, U.S.A. ® marks owned by Allergan, Inc. Licensed from Kyorin Pharmaceuticals Co., Ltd. U.S. Patents 5,880,283 and 6,333,045
Made in the U.S.A.
72366US11C
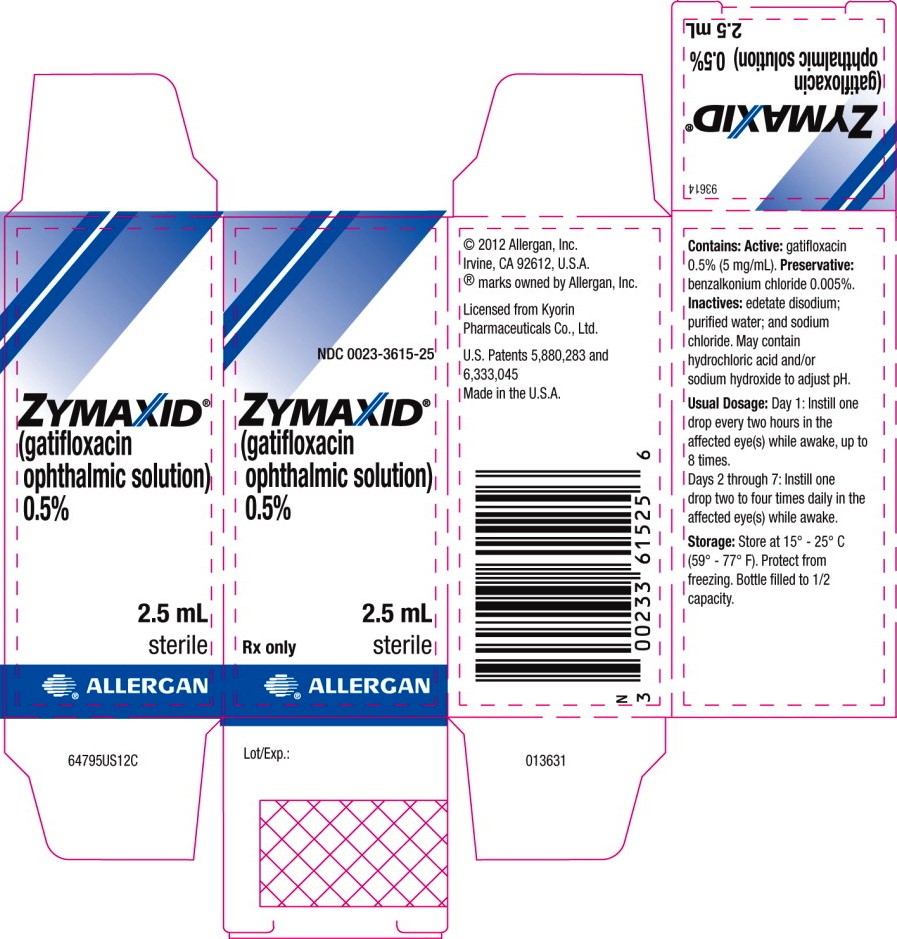
Principal Display Panel
Package Label – 2.5 mL Carton Label
NDC 0023-3615-25
Zymaxid®
(gatifloxacin ophthalmic solution) 0.5%
2.5 mL
Rx only sterile
ALLERGAN®
 |
References
http://dailymed.nlm.nih.gov/dailymed/lookup.cfm?setid=1160b16c-929a-4e85-9c0b-1d8c96a7678b