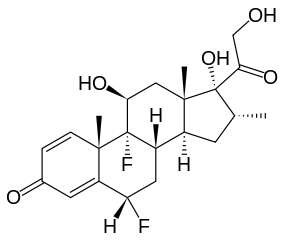
Flumetasone
 | |
| Clinical data | |
|---|---|
| Synonyms | (6S,8S,9S,10S,11S,13S,14S,16R,17R)-6,9-difluoro-11,17-dihydroxy-17-(2-hydroxyacetyl)-10,13,16-trimethyl-6,7,8,11,12,14,15,16-octahydrocyclopenta[a]phenanthren-3-one |
| AHFS/Drugs.com | Micromedex Detailed Consumer Information |
| Routes of administration | Topical |
| ATC code |
|
| Pharmacokinetic data | |
| Metabolism | Hepatic, CYP3A4-mediated |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| UNII | |
| KEGG | |
| ChEMBL | |
| E number | {{#property:P628}} |
| ECHA InfoCard | {{#property:P2566}}Lua error in Module:EditAtWikidata at line 36: attempt to index field 'wikibase' (a nil value). |
| Chemical and physical data | |
| Formula | C22H28F2O5 |
| Molar mass | 410.452 g/mol |
| | |
|
WikiDoc Resources for Flumetasone |
|
Articles |
|---|
|
Most recent articles on Flumetasone Most cited articles on Flumetasone |
|
Media |
|
Powerpoint slides on Flumetasone |
|
Evidence Based Medicine |
|
Clinical Trials |
|
Ongoing Trials on Flumetasone at Clinical Trials.gov Clinical Trials on Flumetasone at Google
|
|
Guidelines / Policies / Govt |
|
US National Guidelines Clearinghouse on Flumetasone
|
|
Books |
|
News |
|
Commentary |
|
Definitions |
|
Patient Resources / Community |
|
Patient resources on Flumetasone Discussion groups on Flumetasone Patient Handouts on Flumetasone Directions to Hospitals Treating Flumetasone Risk calculators and risk factors for Flumetasone
|
|
Healthcare Provider Resources |
|
Causes & Risk Factors for Flumetasone |
|
Continuing Medical Education (CME) |
|
International |
|
|
|
Business |
|
Experimental / Informatics |
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]
Overview
Flumethasone (usually as the pivalic acid ester flumethasone pivalate, trade name Locacorten or Locorten) is a corticosteroid for topical use. It is available in combination with clioquinol, under the trade name Locacorten-Vioform (in some countries Locorten-Vioform), for the treatment of otitis externa and otomycosis.
Flumethasone shows fully 420 times the potency of cortisone in an animal model for anti-inflammatory activity.
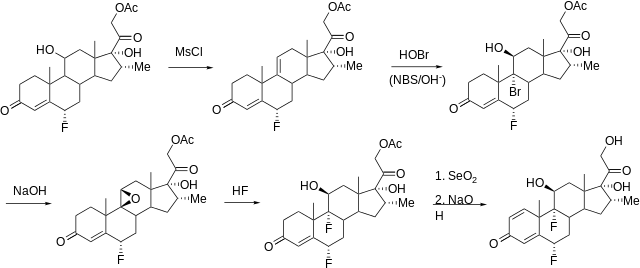
Synthesis
See also
References
- Pages with script errors
- Template:drugs.com link with non-standard subpage
- Articles with changed EBI identifier
- E number from Wikidata
- ECHA InfoCard ID from Wikidata
- Chemical pages without ChemSpiderID
- Chemical pages without DrugBank identifier
- Articles without InChI source
- Drugs with no legal status
- Drugboxes which contain changes to verified fields
- Drugboxes which contain changes to watched fields
- Glucocorticoids
- Organofluorides
- Drug