Captodiame
 | |
| Clinical data | |
|---|---|
| AHFS/Drugs.com | International Drug Names |
| Routes of administration | Oral |
| ATC code | |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Excretion | Renal |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| E number | {{#property:P628}} |
| ECHA InfoCard | {{#property:P2566}}Lua error in Module:EditAtWikidata at line 36: attempt to index field 'wikibase' (a nil value). |
| Chemical and physical data | |
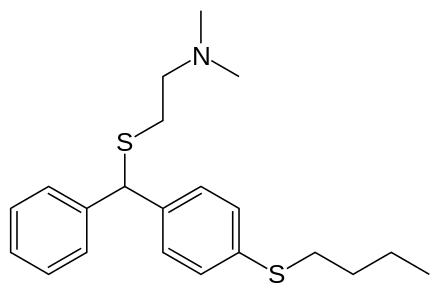
| Formula | C21H29NS2 |
| Molar mass | 359.594 g/mol |
| 3D model (JSmol) | |
| |
| |
| | |
|
WikiDoc Resources for Captodiame |
|
Articles |
|---|
|
Most recent articles on Captodiame |
|
Media |
|
Evidence Based Medicine |
|
Clinical Trials |
|
Ongoing Trials on Captodiame at Clinical Trials.gov Clinical Trials on Captodiame at Google
|
|
Guidelines / Policies / Govt |
|
US National Guidelines Clearinghouse on Captodiame
|
|
Books |
|
News |
|
Commentary |
|
Definitions |
|
Patient Resources / Community |
|
Patient resources on Captodiame Discussion groups on Captodiame Patient Handouts on Captodiame Directions to Hospitals Treating Captodiame Risk calculators and risk factors for Captodiame
|
|
Healthcare Provider Resources |
|
Causes & Risk Factors for Captodiame |
|
Continuing Medical Education (CME) |
|
International |
|
|
|
Business |
|
Experimental / Informatics |
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]
Overview
Captodiame (INN), also known as captodiamine, is an antihistamine sold under the trade names Covatine, Covatix, and Suvren which is used as a sedative and anxiolytic. It is a derivative of diphenhydramine.[1]
A 2004 study suggested captodiame may be helpful in preventing benzodiazepine withdrawal syndrome in people discontinuing benzodiazepine treatment.[1]
In addition to its actions as an antihistamine, captodiamine has been found to act as a 5-HT2C receptor antagonist and σ1 receptor and D3 receptor agonist.[2] It produces antidepressant-like effects in rats.[2] However, captodiamine is unique among antidepressant-like drugs in that it increases brain-derived neurotrophic factor (BDNF) levels in the hypothalamus but not in the frontal cortex or hippocampus.[2] This unique action may be related to its ability to attenuate stress-induced anhedonia and corticotropin-releasing factor (CRF) signaling in the hypothalamus.[2]
See also
References
- ↑ 1.0 1.1 Mercier-Guyon C, Chabannes JP, Saviuc P (2004). "The role of captodiamine in the withdrawal from long-term benzodiazepine treatment". Curr Med Res Opin. 20 (9): 1347–55. doi:10.1185/030079904125004457. PMID 15383182. Free full text with registration
- ↑ 2.0 2.1 2.2 2.3 Ring RM, Regan CM (October 2013). "Captodiamine, a putative antidepressant, enhances hypothalamic BDNF expression in vivo by synergistic 5-HT2c receptor antagonism and sigma-1 receptor agonism". J. Psychopharmacol. (Oxford). 27 (10): 930–9. doi:10.1177/0269881113497614. PMID 23863923.
- Pages with script errors
- CS1 maint: Multiple names: authors list
- Template:drugs.com link with non-standard subpage
- Drugs with non-standard legal status
- Articles with changed DrugBank identifier
- E number from Wikidata
- ECHA InfoCard ID from Wikidata
- Chemical articles with unknown parameter in Infobox drug
- Articles without EBI source
- Drugboxes which contain changes to verified fields
- Drug
- Anticholinergics
- Antihistamines
- Anxiolytics
- Dopamine agonists
- Sedatives
- Sigma agonists
- Thioethers