Baeyer-Villiger oxidation
Overview
The Baeyer-Villiger oxidation is an organic reaction in which a ketone is oxidized to an ester by treatment with peroxy acids or hydrogen peroxide. [1] [2] Key features of the Baeyer-Villiger oxidation are its stereospecificity and predictable regiochemistry.[3] It is named after the German chemist Johann Friedrich Wilhelm Adolf von Baeyer (1835-1917) and the Swiss chemist Victor Villiger (1868-1934).
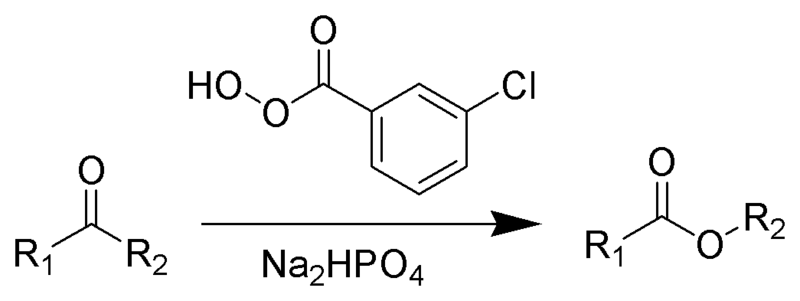
Reagents typically used to carry out this rearrangement are meta-chloroperoxybenzoic acid (mCPBA), peroxyacetic acid, or peroxytrifluoroacetic acid [4]. The original reagent in the 1899 publication is Caro's acid discovered just a year earlier. [5] Disodium hydrogen phosphate is added as a buffering agent to prevent transesterification.
Mechanism
The reaction mechanism of this oxidative cleavage involves first addition of the peroxy acid to the carbonyl forming a tetrahedral intermediate also called the Criegee intermediate for its similarity with rearrangement of that name. The transition state for this step is envisioned as a hydrogen relay involving three peroxy acid molecules with linear O-H-O interactions [6]. Next is a concerted migration of one of the adjacent carbons to oxygen with loss of a carboxylic acid. For unsymmetrical ketones, the migrating group is the one that gives the most stable cation. If the migrating carbon is chiral, the stereochemistry is retained.
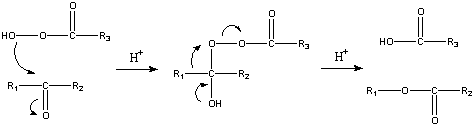
- Cation stability: H > tertiary alkyl > cyclohexyl > secondary alkyl, aryl > primary alkyl > methyl
In the transition state for this migration step the R-C-O-O dihedral angle should be 180° in order to maximise the interaction between the filled R-C sigma bond and the antibonding O-O sigma bond. This step is also (at least in silico) assisted by two or three peroxyacid units enabling the hydroxyl proton to shuttle to its new position[6].
Thus, cyclic ketones produce lactones and aldehydes produce first a formate and then a carboxylic acids although sometimes a alcohol is formed when the formate is hydrolytically unstable.
Biocatalytic BV oxydation
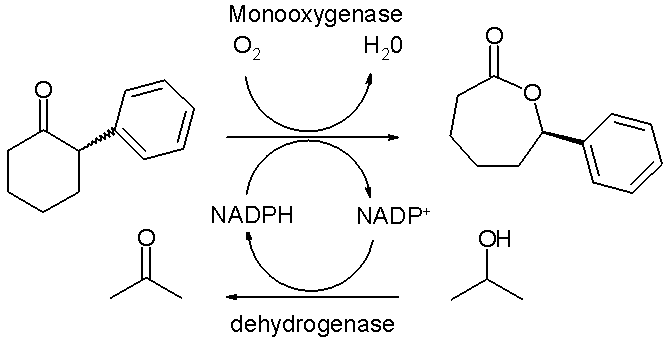
The Baeyer-Villiger oxidation can also be performed by biocatalysis with a so-called Baeyer-Villiger monooxygenase or BVMO. Though largely an experimental technique it shows the promise of enantioselectivity and green chemistry for this reaction type. Current stumbling blocks are confinement to water as the reaction medium, substrate specificity, dependence on the stoichiometric use and costs of cofactors such as NADPH and the costs associated with BVMO's themselves because lengthy purification steps are required. In vivo oxidations with metabolically active microbial cells introduce complications on their own.
In one study [7] the enzyme purification issue is addressed and a special thermally stable monooxygenase is isolated from a specific E-coli strain. This enzyme converts racemic 2-phenylcyclohexanone with oxygen to the corresponding (R)-lactone with 50% chemical yield and 94% enantiomeric excess with in a biphasic system of water and hexane. The NADPH cofactor is regenerated in each catalytic cycle by action of a second dehydrogenase enzyme which consumes isopropanol as a sacrificial catalyst. The solubility of the organic reactant and product is low in the aqueous phase thus averting inhibition. On the other hand the catalytic turnover number for this reaction is much larger than can be obtained with classical organic asymmetric catalysts.
References
- ↑ Baeyer, A. (1899). "Einwirkung des Caro'schen Reagens auf Ketone". Ber. 32 (3): 3625–3633. doi:10.1002/cber.189903203151. Unknown parameter
|coauthors=ignored (help) - ↑ Baeyer, A. (1900). "Ueber die Einwirkung des Caro'schen Reagens auf Ketone". Ber. 33 (1): 858–864. doi:10.1002/cber.190003301153. Unknown parameter
|coauthors=ignored (help) - ↑ Crudden, C. M.; Chen, A. C.; Calhoun, L. A. (2000). "A Demonstration of the Primary Stereoelectronic Effect in the Baeyer-Villiger Oxidation of α-Fluorocyclohexanones". Angew. Chem. Int. Ed. 39 (16): 2851–2855. doi:10.1002/1521-3773(20000818)39:16%3C2851::AID-ANIE2851%3E3.0.CO;2-Y.
- ↑ Burton, J.W.; Clark, J.S.; Derrer, S.; Stork, T.C.; Bendall, J.G.; Holmes, A.B. (1997). "Synthesis of Medium Ring Ethers. 5. The Synthesis of (+)-Laurencin" (Abstract). J. Am. Chem. Soc. 119 (32): 7483–7498. doi:10.1021/ja9709132.
- ↑ 100 Years of Baeyer-Villiger Oxidations Michael Renz, Bernard Meunier European Journal of Organic Chemistry Volume 1999, Issue 4 , Pages 737 - 750 doi:10.1002/(SICI)1099-0690(199904)1999:4<737::AID-EJOC737>3.0.CO;2-B
- ↑ 6.0 6.1 The Role of Hydrogen Bonds in Baeyer-Villiger Reactions Shinichi Yamabe and Shoko Yamazaki J. Org. Chem.; 2007; 72(8) pp 3031 - 3041; (Article) doi:10.1021/jo0626562
- ↑ Frank Schulz; François Leca; Frank Hollmann; Manfred T Reetz (2005). "Towards practical biocatalytic Baeyer-Villiger reactions: applying a thermostable enzyme in the gram-scale synthesis of optically active lactones in a two-liquid-phase system" (PDF). Beilstein Journal of Organic Chemistry. 1 (10). doi:10.1186/1860-5397-1-10. Check date values in:
|date=(help)
See also
de:Baeyer-Villiger-Oxidation it:Baeyer-Villiger nl:Baeyer-Villiger-reactie Template:Jb1