Apheresis
|
WikiDoc Resources for Apheresis |
|
Articles |
|---|
|
Most recent articles on Apheresis |
|
Media |
|
Evidence Based Medicine |
|
Clinical Trials |
|
Ongoing Trials on Apheresis at Clinical Trials.gov Clinical Trials on Apheresis at Google
|
|
Guidelines / Policies / Govt |
|
US National Guidelines Clearinghouse on Apheresis
|
|
Books |
|
News |
|
Commentary |
|
Definitions |
|
Patient Resources / Community |
|
Patient resources on Apheresis Discussion groups on Apheresis Directions to Hospitals Treating Apheresis Risk calculators and risk factors for Apheresis
|
|
Healthcare Provider Resources |
|
Causes & Risk Factors for Apheresis |
|
Continuing Medical Education (CME) |
|
International |
|
|
|
Business |
|
Experimental / Informatics |
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]
Apheresis (Greek: "to take away") is a medical technology in which the blood of a donor or patient is passed through an apparatus that separates out one particular constituent and returns the remainder to the circulation. It is thus an extracorporeal therapy.
Method
Depending on the substance that is being removed, different processes are employed in apheresis. If separation by weight is required, centrifugation would be the method of choice. Other methods involve absorption onto beads coated with an absorbent material. The centrifugation method can be divided into two basic categories:
A. Continuous flow centrifugation (CFC) historically required two venipunctures as the "continuous" means the blood is collected, spun, and returned simultaneously. Newer systems can use a single venipuncture. The main advantage of this system is the low extracorporeal volume (calculated by volume of the apheresis chamber, the donor's hematocrit, and total blood volume of the donor) used in the procedure, which may be advantageous in the elderly and for children.
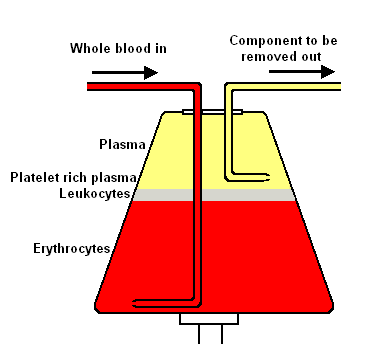
B. Intermittent flow centrifugation works in cycles, taking blood, spinning/processing it and then giving back the unnecessary parts to the donor in a bolus. The main advantage is a single venipuncture site. To stop the blood from coagulating, anticoagulant is automatically mixed with the blood as it is pumped from the body into the apheresis machine. The centrifugation process itself has four variables that can be controlled to selectively remove desired components. The first is spin speed and bowl diameter, the second is "sit time" in centrifuge, the third is solutes added, and the fourth is not as easily controllable: plasma volume and cellular content of the donor. The end product in most cases is the classic sedimented blood sample with the RBC's at the bottom, the "buffy coat" of platelets and WBC's (lymphocytes/granulocytes (PMN's, basophils, eosinophils)/monocytes) in the middle and the plasma on top.
It is important to remember that, when the apheresis system is used for therapy, the system is removing relatively small amounts of fluid (not more than 10.5 mL/kg body weight), but that fluid must be replaced to keep correct intravascular volume. The fluid replaced is different at different institutions. If a crystalloid like normal saline is used, the infusion amount should be triple what is removed as the three to one ratio of NS for plasma is needed to keep up oncotic pressure. Some institutions use normal serum albumin, but it is costly and can be difficult to find. Some advocate using FFP, but the dangers including citrate toxicity (from the anticoagulant), ABO incompatibility, bacterial infection, and cellular antigens make this choice less than desirable.
Types of apheresis
There are numerous types of apheresis. Blood taken from a healthy donor can be separated into its component parts, where the needed component is collected and the "unused" components are returned to the donor. Fluid replacement is usually not needed in these type of collections. There are large categories of component collections:
- Plasmapheresis - blood plasma. Plasmapheresis is useful in collecting FFP (fresh frozen plasma) of a particular ABO group. Commercial uses aside from FFP for this procedure include immune globulin products, plasma derivatives, and collection of rare WBC and RBC antibodies. Consult AABB standards for procedure criteria. FFP may be stored one year at -18 C and seven years at -65 C. After thawing, FFP can be stored for 24 hours at 1-6 C.
- Plateletpheresis (thrombapheresis, thrombocytapheresis) - blood platelets. Plateletpheresis, like it sounds, is the collection of platelets by apheresis; while returning the RBC's, WBC's, and component plasma. The yield is normally the equivalent of between six and ten random platelet concentrates. Quality control demands the platelets from apheresis be equal to or greater than 3.0 x 10^11 in number and have a pH of equal to or greater than 6.2 in 90% of the products tested and must be used within five days. ABO compatibility is a good idea.
- Leukapheresis - leukocytes (white blood cells). Leukopheresis is the removal of PMN's, basophils, eosinophils for transfusion into patients whose PMN's are ineffective or traditional therapy has failed. There is limited data to suggest the benefit of granulocyte infusion. The complications of this procedure are the difficulty in collection and short shelf life (24 hours at 20 to 24 C). Since the "buffy coat" layer sits directly atop the RBC layer, HES, a sedimenting agent, is employed to improve yield while minimizing RBC collection. Quality control demands the resultant concentrate be 1.0 x 10^10 granulocytes in 75% of the units tested and that the product be irradiated to avoid graft-versus-host disease (inactivate lymphocytes). Irradiation does not affect PMN function. Since there is usually a small amount of RBC's collected, ABO compatibility should be employed when feasible.
- Stem cell harvesting - circulating bone marrow cells are harvested to use in bone marrow transplantation.
- LDL apheresis - removal of low density lipoprotein in patients with familial hypercholesterolemia.
- Photopheresis
- Automated Red Cell Collection (2RBC) - removal of two units of red blood cells. Erythrocytaphersis is the collection of RBC's, either two standard units of RBC's or one unit plus either plasma or platelets. The advantage to the donor is the use of smaller needles and saline compensation. The advantage to the blood bankers is the on-line separation into standardized RBC masses with the subsequent reduction in testing, data entry and staffing.
- Immunoadsorbtion with Staphylococcal protein A-agarose column - removal of allo- and autoantibodies (in autoimmune diseases, transplant rejection, hemophilia) by directing plasma through protein A-agarose columns. Protein A is a cell wall component produced by several strains of Staphylococcus aureus which binds to the Fc region of IgG.

Uses
Donation
Blood components can be separated from a collected bag of whole blood or from a donor's blood flow before collected to a blood bag. Various blood components are obtained by apheresis from donors. This includes platelets and blood plasma.
Intravenous immunoglobulin (IVIG) is a blood product administered intravenously. It contains the pooled IgG immunoglobulins (antibodies extracted from the plasma of thousands of blood donors). IVIG is given as a protein replacement therapy for immune deficient patients which have decreased or abolished antibody production capabilities. IVIG is administered to maintain adequate antibodies levels to prevent infections and confers a passive immunity. IVIG effects last between 2 weeks and 3 months. It is mainly used as treatment in three major categories:
Immune deficiencies - Immune deficiencies such as X-linked agammaglobulinemia, hypogammaglobulinemia (Primary immune deficiencies), and acquired compromised immunity conditions (secondary immune deficiencies), featuring low antibody levels. Inflammatory and autoimmune diseases. Acute infections.
Therapy
- Please refer to the individual apheresis methods for use in diseases
The various apheresis techniques may be used whenever the removed constituent is causing severe symptoms of disease. Generally, apheresis has to be performed fairly often, and is an invasive process. It is therefore only employed if other means to control a particular disease have failed, or the symptoms are of such a nature that waiting for medication to become effective would cause suffering or risk of complications.
See also
External links
- Apheresis News
- American Society for Apheresis
- WebPath Apheresis page.
- WebPath Blood Donation and Processing
- Donating Platelet Apheresis: Facts and the FAQ
- Baxter: Automated Component Collection
- Haemonetics: PCS2 System
- Haemonetics: MCS+ 9000 Dystem
- Gambro BCT: Trima Automated Blood Collection System
- Gambro BCT: COBE Spectra Apheresis System
- Gambro BCT: Spectra Optia Apheresis System