Aminolevulinic acid synthase
| aminolevulinate, delta-, synthase 1 | |
|---|---|
| Identifiers | |
| Symbol | ALAS1 |
| Alt. symbols | ALAS3, ALAS |
| Entrez | 211 |
| HUGO | 396 |
| OMIM | 125290 |
| RefSeq | NM_000688 |
| UniProt | P13196 |
| Other data | |
| EC number | 2.3.1.37 |
| Locus | Chr. 3 p21 |
| aminolevulinate, delta-, synthase 2 | |
|---|---|
| Identifiers | |
| Symbol | ALAS2 |
| Alt. symbols | ASB |
| Entrez | 212 |
| HUGO | 397 |
| OMIM | 301300 |
| RefSeq | NM_000032 |
| UniProt | P22557 |
| Other data | |
| EC number | 2.3.1.37 |
| Locus | Chr. X p11.21 |
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]
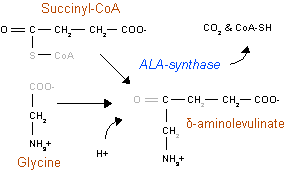
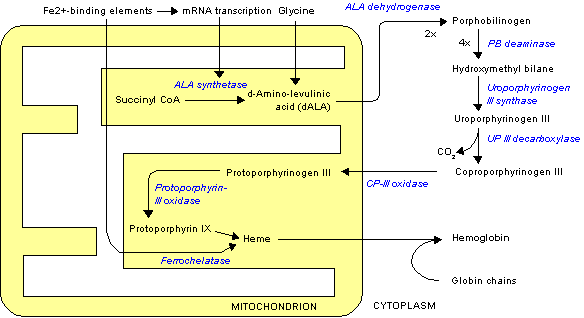
The rate-limiting enzyme in porphyrin and heme biosynthesis is ALA synthase, the enzyme (EC 2.3.1.37) that catalyses glycine and succinyl-CoA into D-Aminolevulinic acid. In humans, transcription of ALA synthase is tightly controlled by the presence of Fe2+-binding elements, to prevent accumulation of porphyrin intermediates in the absence of iron. There are two forms of ALA synthase in the body. One form is expressed in red blood cell precursor cells, whereas the other is expressed throughout the body. The red blood cell form is coded by a gene on chromosome x, whereas the other form is coded by a gene on chromsome 3. The disease X-linked sideroblastic anemia is caused by mutations in the ALA synthase gene on chromosome X, whereas no diseases are known to be caused by mutations in the other gene.
ALA synthase removes the carboxyl group from glycine and the CoA from the succinate, forming δ-aminolevulinic acid (dALA), so called because the amino group is on the fourth carbon atom in the molecule.
External links
- NIH
- Abu-Farha M, Niles J, Willmore W (2005). "Erythroid-specific 5-aminolevulinate synthase protein is stabilized by low oxygen and proteasomal inhibition". Biochem Cell Biol. 83 (5): 620–30. PMID 16234850.