Niacin (extended-release tablet)
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [4]; Associate Editor(s)-in-Chief: Alberto Plate [5]
Disclaimer
WikiDoc MAKES NO GUARANTEE OF VALIDITY. WikiDoc is not a professional health care provider, nor is it a suitable replacement for a licensed healthcare provider. WikiDoc is intended to be an educational tool, not a tool for any form of healthcare delivery. The educational content on WikiDoc drug pages is based upon the FDA package insert, National Library of Medicine content and practice guidelines / consensus statements. WikiDoc does not promote the administration of any medication or device that is not consistent with its labeling. Please read our full disclaimer here.
Overview
Niacin (extended-release tablet) is an antihyperlipidemic, nutriceutical, nutritive agent, Vitamin B that is FDA approved for the treatment of hyperlipidemia, prevention of recurrence of myocardial infarction, reduce atheroesclerotic plaque in CAD and hypertriglyceridemia in patients with risk for pancreatitis. Common adverse reactions include flushing, diarrhea, nausea, vomiting, increased cough, and pruritus.
Adult Indications and Dosage
FDA-Labeled Indications and Dosage (Adult)
There is limited information regarding Niacin (extended-release tablet) FDA-Labeled Indications and Dosage (Adult) in the drug label.
Off-Label Use and Dosage (Adult)
Guideline-Supported Use
There is limited information regarding Off-Label Guideline-Supported Use of Niacin in adult patients.
Non–Guideline-Supported Use
There is limited information regarding Off-Label Non–Guideline-Supported Use of Niacin in adult patients.
Pediatric Indications and Dosage
FDA-Labeled Indications and Dosage (Pediatric)
There is limited information regarding Niacin (extended-release tablet) FDA-Labeled Indications and Dosage (Pediatric) in the drug label.
Off-Label Use and Dosage (Pediatric)
Guideline-Supported Use
There is limited information regarding Off-Label Guideline-Supported Use of Niacin in pediatric patients.
Non–Guideline-Supported Use
There is limited information regarding Off-Label Non–Guideline-Supported Use of Niacin in pediatric patients.
Contraindications
Niacin extended-release tablet is contraindicated in the following conditions:
- Active liver disease or unexplained persistent elevations in hepatic transaminases.
- Patients with active peptic ulcer disease
- Patients with arterial bleeding
- Hypersensitivity to niacin or any component of this medication.
Warnings
Niacin extended-release tablet preparations should not be substituted for equivalent doses of immediate-release (crystalline) niacin. For patients switching from immediate-release niacin to niacin extended-release tablets, therapy with niacin extended-release tablets should be initiated with low doses (i.e., 500 mg at bedtime) and the niacin extended-release tablets dose should then be titrated to the desired therapeutic response.
Caution should also be used when niacin extended-release tablet is used in patients with unstable angina or in the acute phase of an MI, particularly when such patients are also receiving vasoactive drugs such as nitrates, calcium channel blockers, or adrenergic blocking agents.
Niacin is rapidly metabolized by the liver, and excreted through the kidneys. Niacin extended-release tablet is contraindicated in patients with significant or unexplained hepatic impairment and should be used with caution in patients with renal impairment. Patients with a past history of jaundice, hepatobiliary disease, or peptic ulcer should be observed closely during niacin extended-release tablets therapy.
Mortality and Coronary Heart Disease Morbidity
The Atherothrombosis Intervention in Metabolic Syndrome with Low HDL/High Triglycerides: Impact on Global Health Outcomes (AIM-HIGH) trial was a randomized placebo-controlled trial of 3414 patients with stable, previously diagnosed cardiovascular disease. Mean baseline lipid levels were LDL-C 74 mg/dL, HDL-C 35 mg/dL, non-HDL-C 111 mg/dL and median triglyceride level of 163 to 177 mg/dL. Ninety-four percent of patients were on background statin therapy prior to entering the trial. All participants received simvastatin, 40 to 80 mg per day, plus ezetimibe 10 mg per day if needed, to maintain an LDL-C level of 40 to 80 mg/dL, and were randomized to receive niacin extended-release tablets 1500 to 2000 mg/day (n=1718) or matching placebo (IR Niacin, 100 - 150 mg, n=1696). On-treatment lipid changes at two years for LDL-C were -12.0% for the simvastatin plus niacin extended-release tablets group and -5.5% for the simvastatin plus placebo group. HDL-C increased by 25.0% to 42 mg/dL in the simvastatin plus niacin extended-release tablets group and by 9.8% to 38 mg/dL in the simvastatin plus placebo group (P<0.001). Triglyceride levels decreased by 28.6% in the simvastatin plus niacin extended-release tablets group and by 8.1% in the simvastatin plus placebo group. The primary outcome was an ITT composite of the first study occurrence of coronary heart disease death, nonfatal myocardial infarction, ischemic stroke, hospitalization for acute coronary syndrome or symptom-driven coronary or cerebral revascularization procedures. The trial was stopped after a mean follow-up period of 3 years owing to a lack of efficacy. The primary outcome occurred in 282 patients in the simvastatin plus niacin extended-release tablets group (16.4%) and in 274 patients in the simvastatin plus placebo group (16.2%) (HR 1.02 [95% CI, 0.87 to 1.21], P=0.79. In an ITT analysis, there were 42 cases of first occurrence of ischemic stroke reported, 27 (1.6%) in the simvastatin plus niacin extended-release tablets group and 15 (0.9%) in the simvastatin plus placebo group, a non-statistically significant result (HR 1.79, [95%CI = 0.95 to 3.36], p=0.071). The on-treatment ischemic stroke events were 19 for the simvastatin plus niacin extended-release tablets group and 15 for the simvastatin plus placebo group.
Skeletal Muscle
Cases of rhabdomyolysis have been associated with concomitant administration of lipid-altering doses (≥1 g/day) of niacin and statins. Physicians contemplating combined therapy with statins and niacin extended-release tablets should carefully weigh the potential benefits and risks and should carefully monitor patients for any signs and symptoms of muscle pain, tenderness, or weakness, particularly during the initial months of therapy and during any periods of upward dosage titration of either drug. Periodic serum creatine phosphokinase (CPK) and potassium determinations should be considered in such situations, but there is no assurance that such monitoring will prevent the occurrence of severe myopathy.
The risk for myopathy and rhabdomyolysis are increased when lovastatin or simvastatin are coadministered with niacin extended-release tablets, particularly in elderly patients and patients with diabetes, renal failure, or uncontrolled hypothyroidism.
Liver Dysfunction
Cases of severe hepatic toxicity, including fulminant hepatic necrosis, have occurred in patients who have substituted sustained-release (modified-release, timed-release) niacin products for immediate-release (crystalline) niacin at equivalent doses.
Niacin extended-release tablets should be used with caution in patients who consume substantial quantities of alcohol and/or have a past history of liver disease. Active liver diseases or unexplained transaminase elevations are contraindications to the use of niacin extended-release tablets.
Niacin preparations have been associated with abnormal liver tests. In three placebo-controlled clinical trials involving titration to final daily niacin extended-release tablets doses ranging from 500 to 3000 mg, 245 patients received niacin extended-release tablets for a mean duration of 17 weeks. No patient with normal serum transaminase levels (AST, ALT) at baseline experienced elevations to more than 3 times the upper limit of normal (ULN) during treatment with niacin extended-release tablets. In these studies, fewer than 1% (2/245) of niacin extended-release tablets patients discontinued due to transaminase elevations greater than 2 times the ULN.
In three safety and efficacy studies with a combination tablet of niacin extended-release tablets and lovastatin involving titration to final daily doses (expressed as mg of niacin/ mg of lovastatin) 500 mg/10 mg to 2500 mg/40 mg, ten of 1028 patients (1.0%) experienced reversible elevations in AST/ALT] to more than 3 times the ULN. Three of ten elevations occurred at doses outside the recommended dosing limit of 2000 mg/40 mg; no patient receiving 1000 mg/20 mg had 3-fold elevations in AST/ALT.
Niacin extended-release and simvastatin can cause abnormal liver tests. In a simvastatin- controlled, 24 week study with a fixed dose combination of niacin extended-release tablets and simvastatin in 641 patients, there were no persistent increases (more than 3x the ULN) in serum transaminases. In three placebo-controlled clinical studies of extended-release niacin there were no patients with normal serum transaminase levels at baseline who experienced elevations to more than 3x the ULN. Persistent increases (more than 3x the ULN) in serum transaminases have occurred in approximately 1% of patients who received simvastatin in clinical studies. When drug treatment was interrupted or discontinued in these patients, the transaminases levels usually fell slowly to pretreatment levels. The increases were not associated with jaundice or other clinical signs or symptoms. There was no evidence of hypersensitivity.
In the placebo-controlled clinical trials and the long-term extension study, elevations in transaminases did not appear to be related to treatment duration; elevations in AST levels did appear to be dose related. Transaminase elevations were reversible upon discontinuation of niacin extended-release tablets.
Liver function tests should be performed on all patients during therapy with niacin extended-release tablets. Serum transaminase levels, including AST and ALT (SGOT and SGPT), should be monitored before treatment begins, every 6 to 12 weeks for the first year, and periodically thereafter (e.g., at approximately 6-month intervals). Special attention should be paid to patients who develop elevated serum transaminase levels, and in these patients, measurements should be repeated promptly and then performed more frequently. If the transaminase levels show evidence of progression, particularly if they rise to 3 times ULN and are persistent, or if they are associated with symptoms of nausea, fever, and/or malaise, the drug should be discontinued.
Laboratory Abnormalities
- Increase in Blood Glucose: Niacin treatment can increase fasting blood glucose. Frequent monitoring of blood glucose should be performed to ascertain that the drug is producing no adverse effects. Diabetic patients may experience a dose-related increase in glucose intolerance. Diabetic or potentially diabetic patients should be observed closely during treatment with niacin extended-release tablets, particularly during the first few months of use or dose adjustment; adjustment of diet and/or hypoglycemic therapy may be necessary.
- Reduction in platelet count: Niacin extended-release tablet has been associated with small but statistically significant dose-related reductions in platelet count (mean of -11% with 2000 mg). Caution should be observed when niacin extended-release tablet is administered concomitantly with anticoagulants; platelet counts should be monitored closely in such patients.
- Increase in Prothrombin Time (PT): Niacin extended-release tablet has been associated with small but statistically significant increases in prothrombin time (mean of approximately +4%); accordingly, patients undergoing surgery should be carefully evaluated. Caution should be observed when niacin extended-release tablet is administered concomitantly with anticoagulants; prothrombin time should be monitored closely in such patients.
- Increase in Uric Acid: Elevated uric acid levels have occurred with niacin therapy, therefore use with caution in patients predisposed to gout.
- Decrease in Phosphorus: In placebo-controlled trials, niacin extended-release tablet has been associated with small but statistically significant, dose-related reductions in phosphorus levels (mean of -13% with 2000 mg). Although these reductions were transient, phosphorus levels should be monitored periodically in patients at risk for hypophosphatemia.
Adverse Reactions
Clinical Trials Experience
There is limited information regarding Niacin (extended-release tablet) Clinical Trials Experience in the drug label.
Postmarketing Experience
There is limited information regarding Niacin (extended-release tablet) Postmarketing Experience in the drug label.
Drug Interactions
Statins
Caution should be used when prescribing niacin (≥ 1 gm/day) with statins as these drugs can increase risk of myopathy/rhabdomyolysis. Combination therapy with niacin extended-release tablets and lovastatin or niacin extended-release tablets and simvastatin should not exceed doses of 2000 mg niacin extended-release tablets and 40 mg lovastatin or simvastatin daily.
Bile Acid Sequestrants
An in vitro study results suggest that the bile acid-binding resins have high niacin binding capacity. Therefore, 4 to 6 hours, or as great an interval as possible, should elapse between the ingestion of bile acid-binding resins and the administration of niacin extended-release tablets.
Aspirin
Concomitant aspirin may decrease the metabolic clearance of nicotinic acid. The clinical relevance of this finding is unclear.
Antihypertensive Therapy
Niacin may potentiate the effects of ganglionic blocking agents and vasoactive drugs resulting in postural hypotension.
Other
Vitamins or other nutritional supplements containing large doses of niacin or related compounds such as nicotinamide may potentiate the adverse effects of niacin extended-release tablets.
Laboratory Test Interactions
Niacin may produce false elevations in some fluorometric determinations of plasma or urinary catecholamines. Niacin may also give false-positive reactions with cupric sulfate solution (Benedict's reagent) in urine glucose tests.
Use in Specific Populations
Pregnancy
Pregnancy Category (FDA): C Animal reproduction studies have not been conducted with niacin or with niacin extended-release tablets. It is also not known whether niacin at doses typically used for lipid disorders can cause fetal harm when administered to pregnant women or whether it can affect reproductive capacity. If a woman receiving niacin for primary hyperlipidemia becomes pregnant, the drug should be discontinued. If a woman being treated with niacin for hypertriglyceridemia conceives, the benefits and risks of continued therapy should be assessed on an individual basis.
All statins are contraindicated in pregnant and nursing women. When niacin extended-release tablet is administered with a statin in a woman of childbearing potential, refer to the pregnancy category and product labeling for the statin.
Pregnancy Category (AUS):
There is no Australian Drug Evaluation Committee (ADEC) guidance on usage of Niacin (extended-release tablet) in women who are pregnant.
Labor and Delivery
There is no FDA guidance on use of Niacin (extended-release tablet) during labor and delivery.
Nursing Mothers
Niacin is excreted into human milk but the actual infant dose or infant dose as a percent of the maternal dose is not known. Because of the potential for serious adverse reactions in nursing infants from lipid-altering doses of nicotinic acid, a decision should be made whether to discontinue nursing or to discontinue the drug, taking into account the importance of the drug to the mother. No studies have been conducted with niacin extended-release tablets in nursing mothers.
Pediatric Use
Safety and effectiveness of niacin therapy in pediatric patients (≤16 years) have not been established.
Geriatic Use
Of 979 patients in clinical studies of niacin extended-release tablets, 21% of the patients were age 65 and over. No overall differences in safety and effectiveness were observed between these patients and younger patients, and other reported clinical experience has not identified differences in responses between the elderly and younger patients, but greater sensitivity of some older individuals cannot be ruled out.
Gender
Data from the clinical trials suggest that women have a greater hypolipidemic response than men at equivalent doses of niacin extended-release tablets.
Race
There is no FDA guidance on the use of Niacin (extended-release tablet) with respect to specific racial populations.
Renal Impairment
No studies have been performed in this population. Niacin extended-release tablets should be used with caution in patients with renal impairment.
Hepatic Impairment
No studies have been performed in this population. Niacin extended-release tablets should be used with caution in patients with a past history of liver disease and/or who consume substantial quantities of alcohol. Active liver disease, unexplained transaminase elevations and significant or unexplained hepatic dysfunction are contraindications to the use of niacin extended-release tablets
Females of Reproductive Potential and Males
There is no FDA guidance on the use of Niacin (extended-release tablet) in women of reproductive potentials and males.
Immunocompromised Patients
There is no FDA guidance one the use of Niacin (extended-release tablet) in patients who are immunocompromised.
Administration and Monitoring
Administration
There is limited information regarding Niacin (extended-release tablet) Administration in the drug label.
Monitoring
There is limited information regarding Niacin (extended-release tablet) Monitoring in the drug label.
IV Compatibility
There is limited information regarding the compatibility of Niacin (extended-release tablet) and IV administrations.
Overdosage
There is limited information regarding Niacin (extended-release tablet) overdosage. If you suspect drug poisoning or overdose, please contact the National Poison Help hotline (1-800-222-1222) immediately.
Pharmacology
Mechanism of Action
The mechanism by which niacin alters lipid profiles has not been well defined. It may involve several actions including partial inhibition of release of free fatty acids from adipose tissue, and increased lipoprotein lipase activity, which may increase the rate of chylomicron triglyceride removal from plasma. Niacin decreases the rate of hepatic synthesis of VLDL and LDL, and does not appear to affect fecal excretion of fats, sterols, or bile acids.
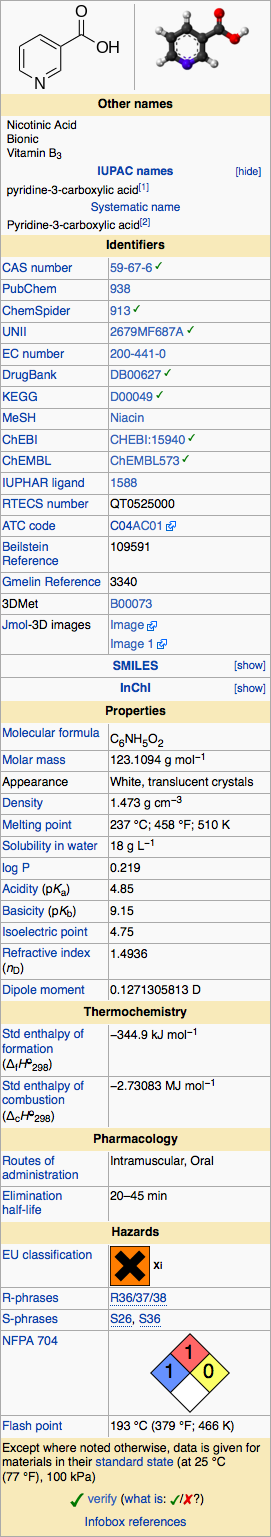
Structure
Niacin extended-release tablets USP (film-coated), contain niacin, which at therapeutic doses is an antihyperlipidemic agent. Niacin (nicotinic acid, or 3-pyridinecarboxylic acid) is a white, crystalline powder, very soluble in water, with the following structural formula:
Pharmacodynamics
Niacin functions in the body after conversion to nicotinamide adenine dinucleotide (NAD) in the NAD coenzyme system. Niacin (but not nicotinamide) in gram doses reduces total cholesterol (TC), low density lipoprotein cholesterol (LDL-C), and triglycerides (TG), and increases high-density lipoprotein cholesterol (HDL-C). The magnitude of individual lipid and lipoprotein responses may be influenced by the severity and type of underlying lipid abnormality. The increase in HDL-C is associated with an increase in apolipoprotein A-I (Apo A-I) and a shift in the distribution of HDL subfractions. These shifts include an increase in the HDL2:HDL3 ratio, and an elevation in lipoprotein A-I (Lp A-I, an HDL-C particle containing only Apo A-I). Niacin treatment also decreases serum levels of apolipoprotein B-100 (Apo B), the major protein component of the very low-density lipoprotein (VLDL) and LDL fractions, and of Lp(a), a variant form of LDL independently associated with coronary risk. In addition, preliminary reports suggest that niacin causes favorable LDL particle size transformations, although the clinical relevance of this effect requires further investigation. The effect of niacin-induced changes in lipids/proteins on cardiovascular morbidity or mortality in individuals without preexisting coronary disease has not been established.
A variety of clinical studies have demonstrated that elevated levels of TC, LDL-C, and Apo B promote human atherosclerosis. Similarly, decreased levels of HDL-C are associated with the development of atherosclerosis. Epidemiological investigations have established that cardiovascular morbidity and mortality vary directly with the level of Total-C and LDL-C, and inversely with the level of HDL-C.
Like LDL, cholesterol-enriched triglyceride-rich lipoproteins, including VLDL, intermediate-density lipoprotein (IDL), and their remnants, can also promote atherosclerosis. Elevated plasma TG are frequently found in a triad with low HDL-C levels and small LDL particles, as well as in association with non-lipid metabolic risk factors for coronary heart disease (CHD). As such, total plasma TG has not consistently been shown to be an independent risk factor for CHD. Furthermore, the independent effect of raising HDL-C or lowering TG on the risk of coronary and cardiovascular morbidity and mortality has not been determined.
Pharmacokinetics
Absorption
Due to extensive and saturable first-pass metabolism, niacin concentrations in the general circulation are dose dependent and highly variable. Time to reach the maximum niacin plasma concentrations was about 5 hours following niacin extended-release tablets. To reduce the risk of gastrointestinal (GI) upset, administration of niacin extended-release tablets with a low-fat meal or snack is recommended.
Single-dose bioavailability studies have demonstrated that the 500 mg and 1000 mg tablet strengths are dosage form equivalent but the 500 mg and the 750 mg tablet strengths are not dosage form equivalent.
Metabolism
The pharmacokinetic profile of niacin is complicated due to extensive first-pass metabolism that is dose-rate specific and, at the doses used to treat dyslipidemia, saturable. In humans, one pathway is through a simple conjugation step with glycine to form nicotinuric acid (NUA). NUA is then excreted in the urine, although there may be a small amount of reversible metabolism back to niacin. The other pathway results in the formation of nicotinamide adenine dinucleotide (NAD). It is unclear whether nicotinamide is formed as a precursor to, or following the synthesis of, NAD. Nicotinamide is further metabolized to at least N-methylnicotinamide (MNA) and nicotinamide-N-oxide (NNO). MNA is further metabolized to two other compounds, N-methyl-2-pyridone-5-carboxamide (2PY) and N-methyl-4-pyridone-5-carboxamide (4PY). The formation of 2PY appears to predominate over 4PY in humans. At the doses used to treat hyperlipidemia, these metabolic pathways are saturable, which explains the nonlinear relationship between niacin dose and plasma concentrations following multiple-dose niacin extended-release tablets administration.
Nicotinamide does not have hypolipidemic activity; the activity of the other metabolites is unknown.
Elimination
Following single and multiple doses, approximately 60 to 76% of the niacin dose administered as niacin extended-release tablet was recovered in urine as niacin and metabolites; up to 12% was recovered as unchanged niacin after multiple dosing. The ratio of metabolites recovered in the urine was dependent on the dose administered.
Nonclinical Toxicology
There is limited information regarding Niacin (extended-release tablet) Nonclinical Toxicology in the drug label.
Clinical Studies
There is limited information regarding Niacin (extended-release tablet) Clinical Studies in the drug label.
How Supplied
There is limited information regarding Niacin (extended-release tablet) How Supplied in the drug label.
Storage
There is limited information regarding Niacin (extended-release tablet) Storage in the drug label.
Images
Drug Images
{{#ask: Page Name::Niacin (extended-release tablet) |?Pill Name |?Drug Name |?Pill Ingred |?Pill Imprint |?Pill Dosage |?Pill Color |?Pill Shape |?Pill Size (mm) |?Pill Scoring |?NDC |?Drug Author |format=template |template=DrugPageImages |mainlabel=- |sort=Pill Name }}
Package and Label Display Panel
{{#ask: Label Page::Niacin (extended-release tablet) |?Label Name |format=template |template=DrugLabelImages |mainlabel=- |sort=Label Page }}
Patient Counseling Information
There is limited information regarding Niacin (extended-release tablet) Patient Counseling Information in the drug label.
Precautions with Alcohol
Alcohol-Niacin interaction has not been established. Talk to your doctor about the effects of taking alcohol with this medication.
Brand Names
There is limited information regarding Niacin (extended-release tablet) Brand Names in the drug label.
Look-Alike Drug Names
There is limited information regarding Niacin (extended-release tablet) Look-Alike Drug Names in the drug label.
Drug Shortage Status
Price
References
The contents of this FDA label are provided by the National Library of Medicine.
For patient information click here
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [6]
Overview
Niacin, also known as nicotinic acid or vitamin B3, is a water-soluble vitamin discovered by Conrad Elvehjem in 1937. Its derivatives, NADH, NAD, NAD+, and NADP play essential roles in energy metabolism in the living cell and DNA repair (an enzymatic process in a living cell). [1] The designation vitamin B3 also includes the corresponding amide nicotinamide (or "niacinamide"), whose chemical formula is C6H6N2O.
Other functions of niacin include removing toxic chemicals from the body,[2] and assisting in the production of steroid hormones made by the adrenal gland, such as sex hormones and stress-related hormones.
History
Niacin was first discovered from the oxidation of nicotine to form nicotinic acid. When the properties of nicotinic acid were discovered, it was thought prudent to choose a name to dissociate it from nicotine, in order to avoid the perception that vitamins or niacin-rich food contains nicotine. The resulting name 'niacin' was derived from nicotinic acid + vitamin.
Niacin is also referred to as Vitamin B3 because it was the third of the B vitamins to be discovered. It has historically been referred to as "vitamin PP", a name derived from the term "pellagra-preventing factor".
Dietary needs
The recommended daily allowance of niacin is 2-12 mg a day for children, 14 mg a day for women, 16 mg a day for men, and 18 mg a day for pregnant or breast-feeding women.[3]
Severe deficiency of niacin in the diet causes the disease pellagra, whereas mild deficiency slows down the metabolism, causing decreased tolerance to cold.
Dietary niacin deficiency tends to occur only in areas where people eat corn (maize), the only grain low in niacin, as a staple food, and that do not use lime during meal/flour production. Alkali lime releases the tryptophan from the corn in a process called nixtamalization so that it can be absorbed in the intestine, and converted to niacin.[2]
Pharmacological uses
Niacin, when taken in large doses, blocks the breakdown of fats in adipose tissue, thus altering blood lipid levels. Niacin is used in the treatment of hyperlipidemia because it reduces very-low-density lipoprotein (VLDL), a precursor of low-density lipoprotein (LDL) or "bad" cholesterol. Because niacin blocks breakdown of fats, it causes a decrease in free fatty acids in the blood and, as a consequence, decreased secretion of VLDL and cholesterol by the liver.[4]
By lowering VLDL levels, niacin also increases the level of high-density lipoprotein (HDL) or "good" cholesterol in blood, and therefore it is sometimes prescribed for patients with low HDL, who are also at high risk of a heart attack.[5][6] An extended release formulation of niacin for this indication is marketed by Abbott Laboratories under the trade name Niaspan.
Niacin is sometimes consumed in large quantities by people who wish to fool drug screening tests, particularly for lipid-soluble drugs such as marijuana.[7] It is believed to "promote metabolism" of the drug and cause it to be "flushed out." Scientific studies have shown it does not affect drug screenings, but can pose a risk of overdose, causing arrhythmias, metabolic acidosis, hyperglycemia, and other serious problems (see below).
In October 2008, Merck expanded the THRIVE Trial from 20,000 to 25,000 patients in order to expedite a second bid for FDA approval of its experimental cholesterol drug, MK-0524A. MK-0524A is a combination of niacin and laropiprant, which is aimed at limiting facial flushing associated with niacin. In April 2008 the FDA decided to withhold approval for the experimental drug, deciding to wait until the results of the THRIVE Trial could be analyzed.[8]
Toxicity
People taking pharmacological doses of niacin (1.5 - 6 g per day) often experience a syndrome of side-effects that can include one or more of the following:[9]
- dermatological complaints
- facial flushing and itching
- dry skin
- skin rashes including acanthosis nigricans
- gastrointestinal complaints
- dyspepsia (indigestion)
- liver toxicity
- hyperglycemia
- cardiac arrhythmias
- birth defects
Facial flushing is the most commonly-reported side-effect.[10] It lasts for about 15 to 30 minutes, and is sometimes accompanied by a prickly or itching sensation. This effect is mediated by prostaglandins and can be blocked by taking 300 mg of aspirin half an hour before taking niacin, or by taking one tablet of ibuprofen per day. Taking the niacin with meals also helps reduce this side-effect. After 1 to 2 weeks of a stable dose, most patients no longer flush. Slow- or "sustained"-release forms of niacin have been developed to lessen these side-effects.[11][12] [13] One study showed the incidence of flushing was 4.5x lower (1.9 vs. 8.6 episodes in the first month) with a sustained-release formulation.[14]
Doses above 2 g per day have been associated with liver damage, particularly with slow-release formulations. [15]
High-dose niacin may also elevate blood sugar, thereby worsening diabetes mellitus.[16] Hyperuricemia is another side-effect of taking high-dose niacin; thus niacin may worsen gout.
Niacin at doses used in lowering cholesterol has been associated with birth defects in laboratory animals and should not be taken by pregnant women.[17]
Niacin at extremely high doses can have life-threatening acute toxic reactions. One patient suffered vomiting after taking eleven 500-milligram niacin tablets over 36 hours, and another was unresponsive for several minutes after taking five 500-milligram tablets over two days.[18][19] Extremely high doses of niacin can also cause niacin maculopathy, a thickening of the macula and retina which leads to blurred vision and blindness.[20]
Inositol hexanicotinate
One popular form of dietary supplement is inositol hexanicotinate, usually sold as "flush-free" or "no-flush" niacin (although those terms are also used for regular sustained-release.) While this form of niacin does not cause the flushing associated with the nicotinic acid form, it is not clear whether it is pharmacologically equivalent in its positive effect.[21]
Biosynthesis
The liver can synthesize niacin from the essential amino acid tryptophan (see below), but the synthesis is extremely inefficient; 60 mg of tryptophan are required to make one milligram of niacin.[22]
The 5-membered aromatic heterocycle of the essential amino acid, tryptophan, is cleaved and rearranged with the alpha amino group of tryptophan into the 6-membered aromatic heterocycle of niacin by the following reaction:
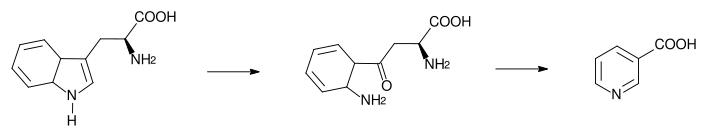
Biosynthesis: Tryptophan → kynurenine → niacin
Receptor
The receptor for niacin is a G-protein coupled receptor called HM74A.[23] It couples to Gi[24].
Food sources
| Animal products: | Fruits and vegetables: | Seeds: | Fungi: |
|---|---|---|---|
|
References
- ↑ Northwestern University Nutrition
- ↑ 2.0 2.1 Vitamin B3 University of Maryland Medical Center.
- ↑ Template:Pauling
- ↑ T. Katzung, Basic and Clinical Pharmacology, 9th ed. p. 570.
- ↑ Postgraduate Medicine
- ↑ Canner PL, Berge KG, Wenger NK, et al. Fifteen year mortality in Coronary Drug Project patients: long-term benefit with niacin. J Am Coll Cardiol. 1986;8(6):1245-1255.
- ↑ Niacin abuse in the attempt to alter urine drug tests. Pharmacist's Letter/Prescriber's Letter 2007;23(6):230606.
- ↑ http://biz.yahoo.com/ap/081017/merck_cholesterol_study.html?.v=2
- ↑ J.G. Hardman et al., eds., Goodman and Gilman's Pharmacological Basis of Therapeutics, 10th ed., p.991.
- ↑ NIH Medline Plus: Niacin. http://www.nlm.nih.gov/medlineplus/ency/article/002409.htm.
- ↑ J.G. Hardman et al., eds., Goodman and Gilman's Pharmacological Basis of Therapeutics, 10th ed., p.991.
- ↑ T. Katzung, Basic and Clinical Pharmacology, 9th ed. p. 570.
- ↑ Options for therapeutic intervention: How effective are the different agents? European Heart Journal Supplements Vol 8 Suppl F Pp. F47-F53 [1]
- ↑ Chapman M, Assmann G, Fruchart J, Sheperd J, Sirtori C. Raising high-density lipoprotein cholesterol with reduction of cardiovascular risk: the role of nicotinic acid - a position paper developed by the European Consensus Panel on HDL-C. Cur Med Res Opin. 2004 Aug;20(8):1253-68. PMID 15324528
- ↑ J.G. Hardman et al., eds., Goodman and Gilman's Pharmacological Basis of Therapeutics, 10th ed., p.992.
- ↑ J.G. Hardman et al., eds., Goodman and Gilman's Pharmacological Basis of Therapeutics, 10th ed., p.991.
- ↑ J.G. Hardman et al., eds., Goodman and Gilman's Pharmacological Basis of Therapeutics, 10th ed., p.992.
- ↑ Hazards: Niacin to Pass a Drug Test Can Have Dangerous Results, By ERIC NAGOURNEY, New York Times, April 17, 2007[2]
- ↑ Mittal MK, Florin T, Perrone J, Delgado JH, Osterhoudt KC. Toxicity From the Use of Niacin to Beat Urine Drug Screening. Ann Emerg Med. 2007 Apr 4. PMID 17418450[3]
- ↑ JD Gass, Nictonic Acid Maculopathy, Am. J. Opthamology, 1973;76:500-10
- ↑ No-Flush Niacin for the Treatment of Hyperlipidemia
- ↑ Oxidization Reactions of Niacin from the Linus Pauling Institute at Oregon State University Linus Pauling Institute.
- ↑ medscape.com - The Metabolic Syndrome: Etiology, Controversies, and Emerging ...
- ↑ Variations in human HM74 (GPR109B) and HM74A (GPR109A) niacin receptors Christian Zellner 1 *, Clive R. Pullinger 1, Bradley E. Aouizerat 2, Philip H. Frost 1, Pui-Yan Kwok 1, Mary J. Malloy 1, John P. Kane
External links
Template:Peripheral vasodilators Template:Lipid modifying agents