Tuberculin
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]; Associate Editor(s)-in-Chief: Kiran Singh, M.D. [2]
Disclaimer
WikiDoc MAKES NO GUARANTEE OF VALIDITY. WikiDoc is not a professional health care provider, nor is it a suitable replacement for a licensed healthcare provider. WikiDoc is intended to be an educational tool, not a tool for any form of healthcare delivery. The educational content on WikiDoc drug pages is based upon the FDA package insert, National Library of Medicine content and practice guidelines / consensus statements. WikiDoc does not promote the administration of any medication or device that is not consistent with its labeling. Please read our full disclaimer here.
Overview
Tuberculin is a diagnostic agent that is FDA approved for the diagnosis of tuberculosis infection. Common adverse reactions include erythema at injection site, syncope,hypersensitivity reaction.
Adult Indications and Dosage
FDA-Labeled Indications and Dosage (Adult)
Indications
- TUBERSOL, Tuberculin Purified Protein Derivative (Mantoux), is indicated to aid diagnosis of tuberculosis infection (TB) in persons at increased risk of developing active disease.
- The Centers for Disease Control and Prevention (CDC) have published guidelines regarding populations that would benefit from tuberculin skin testing (TST). Current recommendations can be accessed at:
- Previous BCG vaccination is not a contraindication to tuberculin testing. The skin-test results of BCG vaccinated persons can be used to support or exclude the diagnosis of TB infection. However, an FDA-approved interferon gamma release assay is preferred over tuberculin skin test for persons 5 years of age and older who were previously vaccinated with BCG.
Dosage
- Five (5) tuberculin units (TU) per test dose of 0.1 mL is the standard strength used for intradermal (Mantoux) testing.
Off-Label Use and Dosage (Adult)
Guideline-Supported Use
- There is limited information regarding Off-Label Guideline-Supported Use of Tuberculin in adult patients.
Non–Guideline-Supported Use
- There is limited information regarding Off-Label Non–Guideline-Supported Use of Tuberculin in adult patients.
Pediatric Indications and Dosage
FDA-Labeled Indications and Dosage (Pediatric)
- There is limited information regarding FDA-Labeled Use of Tuberculin in pediatric patients.
Off-Label Use and Dosage (Pediatric)
Guideline-Supported Use
- There is limited information regarding Off-Label Guideline-Supported Use of Tuberculin in pediatric patients.
Non–Guideline-Supported Use
- There is limited information regarding Off-Label Non–Guideline-Supported Use of Tuberculin in pediatric patients.
Contraindications
- Allergy to any component of TUBERSOL or an anaphylactic or other allergic reaction to a previous test of tuberculin PPD is a contraindication to the use of TUBERSOL.
- TUBERSOL should not be administered to:
- Persons who have had a severe reaction (e.g., necrosis, blistering, anaphylactic shock or ulcerations) to a previous TST,
- Persons with documented active tuberculosis or a clear history of treatment for TB infection or disease,
- Persons with extensive burns or eczema.
Warnings
Hypersensitivity
- Allergic reactions may occur following the use of tubersol even in persons with no prior history of hypersensitivity to the product components.Epinephrine injection (1:1,000) and other appropriate agents used for the control of immediate allergic reactions must be immediately available.
Syncope
- Syncope (fainting) can occur in association with administration of injectable medicines, including TUBERSOL. Procedures should be in place to avoid falling injury and to restore cerebral perfusion following syncope.
Adverse Reactions
Clinical Trials Experience
- Induration at the tubersol injection site is the expected reaction for a positive skin test.
- The information pertaining to adverse events has been compiled from historical clinical studies and post-marketing experience with TUBERSOL.
General disorders and administration site conditions
Injection site pain, injection site pruritus and injection site discomfort.
- Injection site erythema or injection site rash (without induration) occurring within 12 hours of testing. These reactions do not indicate TB infection.
- Injection site hemorrhage and injection site hematoma up to three days after the administration of the test.
- Injection site vesicles, injection site ulcer or injection site necrosis in highly sensitive persons.
- Injection site scar as a result of strongly positive reactions.
Immune system disorders
- Hypersensitivity, including anaphylaxis/anaphylactic reactions, angiodema, urticaria
Respiratory, thoracic and mediastinal disorders
Skin and subcutaneous tissue disorders
Nervous system disorders
- Presyncope, syncope (including syncope associated with tonic-clonic movements and other seizure-like activity) sometimes resulting in transient loss of consciousness with injury
Postmarketing Experience
There is limited information regarding Postmarketing Experience of Tuberculin in the drug label.
Drug Interactions
- Reactivity to the test may be depressed or suppressed in persons who are receiving corticosteroids or immunosuppressive agents.
- Reactivity to TUBERSOL may be temporarily depressed by certain live virus vaccines (measles, mumps, rubella, oral polio, yellow fever, and varicella). If a parenteral live attenuated virus vaccine has been administered recently, tuberculin testing should be delayed for >1 month after vaccination.
- When tuberculin screening is required at the same time as a measles-containing vaccine or other parenteral live attenuated virus vaccine, simultaneous administration of TUBERSOL and the vaccine at separate sites is the preferred option.
Use in Specific Populations
Pregnancy
Pregnancy Category (FDA): PREGNANCY CATEGORY C
- Animal reproduction studies have not been conducted with TUBERSOL. It is also not known whether TUBERSOL can cause fetal harm when administered to a pregnant woman or can affect reproduction capacity. TUBERSOL should be given to a pregnant woman only if clearly needed.
- Australian Drug Evaluation Committee (ADEC) Pregnancy Category
There is no Australian Drug Evaluation Committee (ADEC) guidance on usage of Tuberculin in women who are pregnant.
Labor and Delivery
There is no FDA guidance on use of Tuberculin during labor and delivery.
Nursing Mothers
- It is not known whether TUBERSOL is excreted in human milk. Because many drugs are excreted in human milk, caution should be exercised when TUBERSOL is administered to a nursing woman.
Pediatric Use
- There is no age contraindication to tuberculin skin testing of infants. Because their immune systems are immature, many infants <6 weeks of age who are infected with M. tuberculosis do not react to tuberculin tests.
Geriatic Use
- Clinical studies of TUBERSOL did not include sufficient numbers of subjects aged 65 and over to determine whether they respond differently from younger subjects.
Gender
There is no FDA guidance on the use of Tuberculin with respect to specific gender populations.
Race
There is no FDA guidance on the use of Tuberculin with respect to specific racial populations.
Renal Impairment
There is no FDA guidance on the use of Tuberculin in patients with renal impairment.
Hepatic Impairment
There is no FDA guidance on the use of Tuberculin in patients with hepatic impairment.
Females of Reproductive Potential and Males
There is no FDA guidance on the use of Tuberculin in women of reproductive potentials and males.
Immunocompromised Patients
There is no FDA guidance one the use of Tuberculin in patients who are immunocompromised.
Administration and Monitoring
Administration
- Intradermal injection
- TUBERSOL is indicated for intradermal injection only. Do not inject intravenously, intramuscularly, or subcutaneously. If subcutaneous injection occurs, the test cannot be interpreted.
- Inspect for extraneous particulate matter and/or discoloration before use. If these conditions exist, do not administer the product.
- Use a separate syringe and needle for each injection.
- The following procedure is recommended for performing the Mantoux test:
- The preferred site of the test is the volar aspect of the forearm. Avoid areas on the skin that are red or swollen. Avoid visible veins.
- Clean the skin site with a suitable germicide and allow the site to dry prior to injection of the antigen.
- Administer the test dose (0.1 mL) of TUBERSOL with a 1 mL syringe calibrated in tenths and fitted with a short, one-quarter to one-half inch, 26 or 27 gauge needle.
- Wipe the stopper of the vial with a suitable germicide and allow to dry before needle insertion. Then insert the needle gently through the stopper and draw 0.1 mL of TUBERSOL into the syringe. Avoid injection of excess air with removal of each dose so as not to over pressurize the vial and possibly cause seepage at the puncture site.
- Insert the point of the needle into the most superficial layers of the skin with the needle bevel pointing upward and administer the dose by slow intradermal injection. If the intradermal injection is performed properly, a definite pale bleb will rise at the needle point, about 10 mm (⅜") in diameter. This bleb will disperse within minutes. Do not dress the site.
- A drop of blood may appear at the administration site following injection. Blot the site lightly to remove the blood but avoid squeezing out the injected tuberculin test fluid.
- In the event of an improperly performed injection (ie, no bleb formed), repeat the test immediately at another site, at least 2 inches from the first site and circle the second injection site as an indication that this is the site to be read.
- Inform the patient of the need to return for the reading of the test by a trained health professional. Self-reading may be inaccurate and is strongly discouraged.
Monitoring
INTERPRETATION OF THE TEST
- The skin test should be read by a trained health professional 48 to 72 hours after administration of TUBERSOL. Skin test sensitivity is indicated by induration only; redness should not be measured.
- Measure the diameter of induration transversely to the long axis of the forearm and record the measurement in millimetres (including 0 mm). (8) The tip of a ballpoint pen, gently pushed at a 45° angle toward the site of injection, will stop at the edge of induration.
- Also record presence and size (if present) of necrosis and edema, although these are not used in the interpretation of the test.
Positive Reactions
- Tuberculin reactivity may indicate latent infection, prior infection and/or disease with M. tuberculosis and does not necessarily indicate the presence of active tuberculous disease. Persons showing positive tuberculin reactions should be considered positive by current public health guidelines and referred for further medical evaluation.The repeated testing of uninfected persons does not sensitize them to TUBERSOL.
- The significance of induration measurements in diagnosing latent TB infection must be considered in terms of the patient's history and the risk of developing active TB disease as indicated in Table 1.
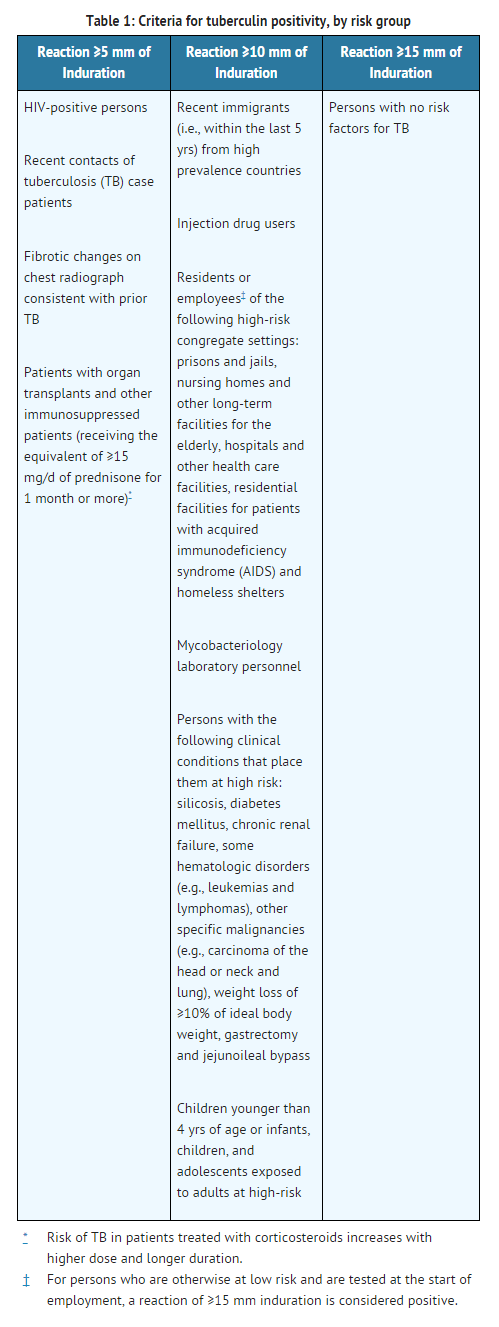
- A TST conversion is defined as an increase of ≥ 10 mm of induration within a 2-year period, regardless of age.
- The possibility should be considered that the skin test sensitivity may also be due to a previous contact with atypical mycobacteria or previous BCG vaccination.
Negative Reactions
- An individual who does not show a positive reaction to 5 TU on the first test, but is suspected of being TB positive, may be retested with 5 TU.Any individual who does not show a positive reaction to an initial injection of 5 TU, or a second test with 5 TU may be considered as tuberculin negative.
False Positive Reactions
- False positive tuberculin reactions can occur in individuals who have been infected with other mycobacteria, including vaccination with BCG.However, a diagnosis of M. tuberculosis infection and the use of preventive therapy should be considered for any BCG-vaccinated person who has a positive TST reaction, especially if the person has been, or is, at increased risk of acquiring TB infection.
False-Negative Reactions
- Not all infected persons will have a delayed hypersensitivity reaction to a tuberculin test.
- In those who are elderly or those who are being tested for the first time, reactions may develop slowly and may not peak until after 72 hours.
- Since tuberculin sensitivity may take up to 8 weeks to develop following exposure to M. tuberculosis, persons who have a negative tuberculin test <8 weeks following possible TB exposure should be retested ≥8-10 weeks following the last known or suspected exposure.
Altered Immune Status
- Impaired or attenuated cell mediated immunity (CMI) can potentially cause a false negative tuberculin reaction. Many factors have been reported to cause a decreased ability to respond to the tuberculin test in the presence of tuberculous infection including viral infections (e.g., measles, mumps, chickenpox and HIV), live virus vaccinations (e.g., measles, mumps, rubella, oral polio and yellow fever), overwhelming tuberculosis, other bacterial infections, leukemia, sarcoidosis, fungal infections, metabolic derangements, low protein states, diseases affecting lymphoid organs, drugs (corticosteroids and many other immunosuppressive agents), and malignancy or stress.A TST should be deferred for patients with major viral infections or live-virus vaccination in the past month. Persons with the common cold may be tuberculin tested.
- Because TST results in HIV-infected individuals are less reliable as CD4 counts decline, screening should be completed as early as possible after HIV-infection occurs.
IV Compatibility
- There is limited information regarding IV Compatibility of Tuberculin in the drug label.
Overdosage
- There is limited information regarding Chronic Overdose of Tuberculin in the drug label.
Pharmacology
Tuberculin
| |
| Systematic (IUPAC) name | |
| ? | |
| Identifiers | |
| CAS number | ? |
| ATC code | V04 |
| PubChem | ? |
| Chemical data | |
| Formula | ? |
| Mol. mass | ? |
| Pharmacokinetic data | |
| Bioavailability | ? |
| Metabolism | ? |
| Half life | ? |
| Excretion | ? |
| Therapeutic considerations | |
| Pregnancy cat. |
C(US) |
| Legal status |
[[Prescription drug|Template:Unicode-only]](US) |
| Routes | Intradermal |
Mechanism of Action
- The sensitization following infection with mycobacteria occurs primarily in the regional lymph nodes. Small lymphocytes (T lymphocytes) proliferate in response to the antigenic stimulus to give rise to specifically sensitized lymphocytes. After 3-8 weeks, these lymphocytes enter the blood stream and circulate for years. (7) Subsequent restimulation of these sensitized lymphocytes with the same or a similar antigen, such as the intradermal injection of TUBERSOL, evokes a local reaction mediated by these cells.
- Characteristically, delayed hypersentitivity reactions to tuberculin begin at 5 to 6 hours, are maximal at 48 to 72 hours and subside over a period of days. The resultant immune response consists of induration due to cell infiltration and occasionally vesiculation and necrosis. Clinically, a delayed hypersensitivity reaction to tuberculin is a manifestation of previous infection with M tuberculosis or a variety of non-tuberculosis bacteria. In most cases sensitization is induced by natural mycobacterial infection or by vaccination with BCG Vaccine.
Structure
- TUBERSOL®, Tuberculin Purified Protein Derivative (Mantoux) (PPD) (1) for intradermal tuberculin testing is prepared from a large Master Batch Connaught Tuberculin (CT68) and is a cell-free purified protein fraction obtained from a human strain of Mycobacterium tuberculosis grown on a protein-free synthetic medium and inactivated.The use of a standard preparation derived from a single batch (CT68) has been adopted in order to eliminate batch to batch variation by the same manufacturer.
- TUBERSOL is a clear, colorless liquid.
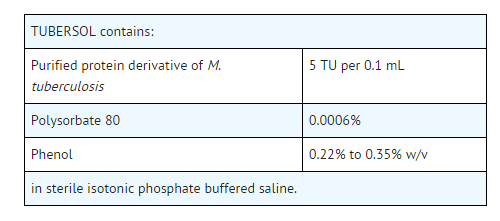
- Before release, each successive lot is tested for potency in comparison with the US Standard Tuberculin PPD-S.
- Independent studies conducted by the US Public Health Service in humans have determined the amount of CT68 in stabilized solution necessary to produce bio-equivalency with Tuberculin PPD-S (in phosphate buffer without polysorbate 80) using 5 US units (TU) Tuberculin PPD-S as the standard.
Pharmacodynamics
There is limited information regarding Pharmacodynamics of Tuberculin in the drug label.
Pharmacokinetics
There is limited information regarding Pharmacokinetics of Tuberculin in the drug label.
Nonclinical Toxicology
CARCINOGENESIS, MUTAGENESIS, IMPAIRMENT OF FERTILITY
- TUBERSOL has not been evaluated for its carcinogenic or mutagenic potentials or impairment of fertility.
Clinical Studies
There is limited information regarding Clinical Studies of Tuberculin in the drug label.
How Supplied
- TUBERSOL, Tuberculin Purified Protein Derivative (Mantoux), bioequivalent to 5 US units (TU) PPD-S per test dose (0.1 mL) is supplied in:
- 10-test vial, 1 mL. package of 1 vial, NDC No. 54868-2972-1
- 50-test vial, 5 mL.
- The stopper of the vial for this product does not contain natural latex rubber.
Storage
- Store at 2° to 8°C (35° to 46°F). (21) Do not freeze. Discard product if exposed to freezing.
- Protect from light. Tuberculin PPD solutions can be adversely affected by exposure to light. The product should be stored in the dark except when doses are actually being withdrawn from the vial.
- A vial of TUBERSOL which has been entered and in use for 30 days should be discarded.
- Do not use after expiration date.
Images
Drug Images
{{#ask: Page Name::Tuberculin |?Pill Name |?Drug Name |?Pill Ingred |?Pill Imprint |?Pill Dosage |?Pill Color |?Pill Shape |?Pill Size (mm) |?Pill Scoring |?NDC |?Drug Author |format=template |template=DrugPageImages |mainlabel=- |sort=Pill Name }}
Package and Label Display Panel


{{#ask: Label Page::Tuberculin |?Label Name |format=template |template=DrugLabelImages |mainlabel=- |sort=Label Page }}
Patient Counseling Information
There is limited information regarding Patient Counseling Information of Tuberculin in the drug label.
Precautions with Alcohol
- Alcohol-Tuberculin interaction has not been established. Talk to your doctor about the effects of taking alcohol with this medication.
Brand Names
- TUBERSOL ®[1]
Look-Alike Drug Names
- A® — B®[2]
Drug Shortage Status
Price
References
The contents of this FDA label are provided by the National Library of Medicine.
- ↑ "TUBERSOL - tuberculin purified protein derivative injection, solution".
- ↑ "http://www.ismp.org". External link in
|title=(help)
{{#subobject:
|Page Name=Tuberculin
|Pill Name=No image.jpg
|Drug Name=
|Pill Ingred=|+sep=;
|Pill Imprint=
|Pill Dosage={{{dosageValue}}} {{{dosageUnit}}}
|Pill Color=|+sep=;
|Pill Shape=
|Pill Size (mm)=
|Pill Scoring=
|Pill Image=
|Drug Author=
|NDC=
}}
{{#subobject:
|Label Page=Tuberculin |Label Name=Tuberculin11.png
}}
{{#subobject:
|Label Page=Tuberculin |Label Name=Tuberculin11.png
}}